- Methamphetamine
-
"Meth" and "Tik" redirect here. For other uses, see Meth (disambiguation) and Tik (disambiguation).This article is about the psychostimulant drug, methamphetamine, in both racemic and dextrorotatory forms. For the CNS inactive OTC nasal decongestant, see levomethamphetamine.
Methamphetamine 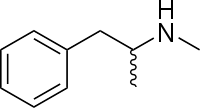
Systematic (IUPAC) name N-methyl-1-phenylpropan-2-amine Clinical data Pregnancy cat. C(US) Legal status Controlled (S8) (AU) Schedule I (CA) Schedule II (US) Class A(NZ)
Schedule 5(SA)
Injectable:Class A, Oral: A(UK)Routes Medical: Ingestion
Recreational: Ingestion, Intravenous, Insufflation, Inhalation, SuppositoryPharmacokinetic data Bioavailability 62.7% oral; 79% nasal; 90.3% smoked; 99% rectally; 100% IV Metabolism Hepatic Half-life 9–12 hours[1] Excretion Renal Identifiers CAS number 537-46-2 
ATC code N06BA03 PubChem CID 1206 DrugBank DB01577 ChemSpider 1169 
UNII 44RAL3456C 
KEGG D08187 
ChEMBL CHEMBL1201201 
Synonyms Desoxyephedrine
Methamfetamine
Pervitin
Anadrex
Methedrine
Methylamphetamine
Syndrox
DesoxynChemical data Formula C10H15N Mol. mass 149.233 g/mol SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Methamphetamine (USAN) (pronounced /ˌmɛθæmˈfɛtəmiːn/ listen) (also known as methamfetamine (INN),[2] N-methylamphetamine, methylamphetamine, and desoxyephedrine) is a psychostimulant of the phenethylamine and amphetamine class of psychoactive drugs. When used illicitly, methamphetamine is commonly referred to as "crystal meth", "meth", "crystal", "ice", "p", "shabu" or "glass".
Methamphetamine increases alertness, concentration, energy, and in high doses, may induce euphoria, enhance self-esteem and increase libido.[3][4] Methamphetamine has high potential for abuse and addiction, activating the psychological reward system by triggering a cascading release of dopamine in the brain. Methamphetamine is FDA approved for the treatment of ADHD and exogenous obesity. It is dispensed in the USA under the trademark name Desoxyn.[5]
As a result of methamphetamine-induced neurotoxicity to dopaminergic neurons, chronic abuse may also lead to post acute withdrawals which persist beyond the withdrawal period for months, and even up to a year.[6] A study performed on female Japanese prison inmates suffering from methamphetamine addiction showed 20% experienced a psychosis resembling schizophrenia which persisted for longer than six months post-methamphetamine use; this amphetamine psychosis could be resistant to traditional treatment.[7] In addition to psychological harm, physical harm, primarily consisting of cardiovascular damage, may occur with chronic use or acute overdose.[8]
Contents
Medical use
 Desoxyn 10 mg tablets (US)
Desoxyn 10 mg tablets (US)
Methamphetamine has been FDA approved for the treatment of ADHD and exogenous obesity in children and adults under the trademark name Desoxyn.[5]
Desoxyn may also be prescribed off-label for the treatment of narcolepsy and treatment-resistant depression.[9]
Effects
Physical
Physical effects can include anorexia, hyperactivity, dilated pupils, flushing, restlessness, dry mouth, headache, tachycardia, bradycardia, tachypnea, hypertension, hypotension, hyperthermia, diaphoresis, diarrhea, constipation, blurred vision, dizziness, twitching, insomnia, numbness, palpitations, arrhythmias,[10] tremors, dry and/or itchy skin, acne, pallor, and with chronic and/or high doses, convulsions,[11] heart attack,[12] stroke,[13] and death can occur.[13][14][15][16][17][18]
Psychological
Psychological effects can include euphoria, anxiety, increased libido, alertness, concentration, increased energy, increased self-esteem, self-confidence, sociability, irritability, aggressiveness, psychosomatic disorders, psychomotor agitation, dermatillomania, delusions of grandiosity, hallucinations, excessive feelings of power and invincibility, repetitive and obsessive behaviors, paranoia, and with chronic and/or high doses, amphetamine psychosis can occur.[13][19]
Withdrawal
Withdrawal symptoms of methamphetamine primarily consist of fatigue, depression and an increased appetite. Symptoms may last for days with occasional use and weeks or months with chronic use, with severity dependent on the length of time and the amount of methamphetamine used. Withdrawal symptoms may also include anxiety, irritability, headaches, agitation, akathisia, hypersomnia (excessive sleeping), vivid or lucid dreams, deep REM sleep and suicidal ideation.[20]
Long-term
Methamphetamine use has a high association with depression and suicide as well as serious heart disease, amphetamine psychosis, anxiety and violent behaviours. Methamphetamine also has a very high addiction risk.[8] Methamphetamine is neurotoxic and is associated with an increased risk of Parkinson's disease.[6][21] Methamphetamine abuse can cause neurotoxicity which is believed to be responsible for causing persisting cognitive deficits, such as memory, impaired attention and executive function. Over 20 percent of people addicted to methamphetamine develop a long-lasting psychosis resembling schizophrenia after stopping methamphetamine which persists for longer than 6 months and is often treatment resistant.[7] Methamphetamine use is frequently comorbid with other mental health issues, especially clinical depression, likely due to its dopaminergic qualities. (Dopamine imbalances are often implicated in psychological health problems.)
Tolerance
As with other amphetamines, tolerance to methamphetamine is not completely understood but known to be sufficiently complex that it cannot be explained by any single mechanism. The extent of tolerance and the rate at which it develops vary widely between individuals, and, even within one person, it is highly dependent on dosage, duration of use, and frequency of administration. Tolerance to the awakening effect of amphetamines does not readily develop, making them suitable for the treatment of narcolepsy.[22]
Short-term tolerance can be caused by depleted levels of neurotransmitters within the synaptic vesicles available for release into the synaptic cleft following subsequent reuse (tachyphylaxis). Short-term tolerance typically lasts until neurotransmitter levels are fully replenished; because of the toxic effects on dopaminergic neurons, this can be greater than 2–3 days. Prolonged overstimulation of dopamine receptors caused by methamphetamine may eventually cause the receptors to upregulate in order to compensate for increased levels of dopamine within the synaptic cleft.[23] To compensate, larger quantities of the drug are needed in order to achieve the same level of effects.
Reverse tolerance or sensitization can also occur.[22] The effect is well established, but the mechanism is not well understood.
Adverse effects
Addiction
Methamphetamine is highly addictive.[24] While the withdrawal itself may not be dangerous, withdrawal symptoms are common with heavy use and relapse is common.
Methamphetamine-induced hyperstimulation of pleasure pathways leads to anhedonia. It is possible that daily administration of the amino acids L-tyrosine and L-5HTP/tryptophan can aid in the recovery process by making it easier for the body to reverse the depletion of dopamine, norepinephrine, and serotonin. Although studies involving the use of these amino acids have shown some success, this method of recovery has not been shown to be consistently effective.
It is shown that taking ascorbic acid prior to using methamphetamine may help reduce acute toxicity to the brain, as rats given the human equivalent of 5–10 grams of ascorbic acid 30 minutes prior to methamphetamine dosage had toxicity mediated,[25][26] yet this will likely be of little avail in solving the other serious behavioral problems associated with methamphetamine use and addiction that many users experience. Large doses of ascorbic acid also lower urinary pH, reducing methamphetamine's elimination half-life and thus decreasing the duration of its actions.[27]
To combat addiction, doctors are beginning to use other forms of stimulants such as dextroamphetamine, the dextrorotatory (right-handed) isomer of the amphetamine molecule, to break the addiction cycle in a method similar to the use of methadone in the treatment of heroin addicts. There are no publicly available drugs comparable to naloxone, which blocks opiate receptors and is therefore used in treating opiate dependence, for use with methamphetamine problems.[28] However, experiments with some monoamine reuptake inhibitors such as indatraline have been successful in blocking the action of methamphetamine.[29] There are studies indicating that fluoxetine, bupropion and imipramine may reduce craving and improve adherence to treatment.[30] Research has also suggested that modafinil can help addicts quit methamphetamine use.[31][32]
Methamphetamine addiction is one of the most difficult forms of addictions to treat. Bupropion, aripiprazole, and baclofen have been employed to treat post-withdrawal cravings, although the success rate is low. Modafinil is somewhat more successful, but this is a Class IV scheduled drug. Ibogaine has been used with success in Europe, where it is a Class I drug and available only for scientific research. Mirtazapine has been reported useful in some small-population studies.[33]
As the phenethylamine phentermine is a constitutional isomer of methamphetamine, it has been suggested that it may be effective in treating methamphetamine addiction. Phentermine is a central nervous system stimulant that acts on dopamine and norepinephrine. When comparing (+)-Amphetamine, (+/-)-ephedrine, and phentermine, one key difference among the three drugs is their selectivity for norepinephrine (NE) release vs. dopamine (DA) release. The NE/DA selectivity ratios for these drugs as determined in vitro [(EC(50) NE(-1))/(EC(50) DA(-1))] are (+/-)-ephedrine (18.6) > phentermine (6.7) > (+)-amphetamine (3.5).[34]
Abrupt interruption of chronic methamphetamine use results in the withdrawal syndrome in almost 90% of the cases.
The mental depression associated with methamphetamine withdrawal lasts longer and is more severe than that of cocaine withdrawal.[30]
Meth mouth
Main article: Meth mouthMethamphetamine users and addicts may lose their teeth abnormally quickly, a condition informally known as meth mouth. According to the American Dental Association, meth mouth "is probably caused by a combination of drug-induced psychological and physiological changes resulting in xerostomia (dry mouth), extended periods of poor oral hygiene, frequent consumption of high-calorie, carbonated beverages and bruxism (teeth grinding and clenching). Some reports have also speculated that the caustic nature of the drug is a contributing factor.[35][36] Similar, though far less severe, symptoms have been reported in clinical use of regular amphetamine, where effects are not exacerbated by extended periods of poor oral hygiene.[37][38]
Public health issues
Short-term exposure to high concentrations of chemical vapors that may exist in methamphetamine laboratories can cause severe health problems or even result in death. Exposure to these substances can occur from volatile air emissions, spills, fires, and explosions.[39] Methamphetamine labs are often discovered when fire fighters respond to a blaze. Methamphetamine cooks, their families, and first responders are at highest risk of acute health effects from chemical exposure, including lung damage and chemical burns to the body. Following a seizure of a methamphetamine lab, there is often a low exposure risk to chemical residues, however this contamination should be sanitized. Chemical residues and lab wastes that are left behind at a former methamphetamine lab can result in severe health problems for people who use the property, therefore local health departments should thoroughly assess the property for hazards prior to allowing it to be reinhabited, especially by children. Those seeking home ownership in heavy meth use areas should be especially careful while house hunting and be sure to have properties inspected before purchasing.[40][41]
Pregnancy and breastfeeding
Methamphetamine present in a mother's bloodstream passes through the placenta to a fetus, and is also secreted into breast milk. Infants born to methamphetamine-abusing mothers were found to have a significantly smaller gestational age-adjusted head circumference and birth weight measurements. Methamphetamine exposure was also associated with neonatal withdrawal symptoms of agitation, vomiting and tachypnea.[42] This withdrawal syndrome is relatively mild and only requires medical intervention in approximately 4% of cases.[30]
Risk of sexually transmitted disease
See also: Sex and drugs and Party and playMen who use methamphetamine, cocaine, MDMA, and ketamine are twice as likely to have unprotected sex, according to British research.[43] American psychologist Perry N. Halkitis performed an analysis using data collected from community-based participants among gay and bisexual men to examine the associations between their methamphetamine use and sexual risk taking behaviors. Methamphetamine use was found to be related to higher frequencies of unprotected sexual intercourse in both HIV-positive and unknown casual partners in the study population. The association between methamphetamine use and unprotected acts were also more pronounced in HIV-positive participants. These findings suggested that methamphetamine use and engagement in unprotected anal intercourse are co-occurring risk behaviors that potentially heighten the risk of HIV transmission among gay and bisexual men.[44] Methamphetamine allows users of both sexes to engage in prolonged sexual activity, which may cause genital sores and abrasions. Methamphetamine can also cause sores and abrasions in the mouth via bruxism (teeth clenching and grinding), which can turn typically low-risk sex acts, such as oral sex, into high-risk sexual activity.[45] As with the injection of any drug, if a group of users share a common needle , blood-borne diseases, such as HIV or hepatitis, can be transmitted. The level of needle sharing among methamphetamine users is similar to that among other drug injection users.[46]
Pharmacokinetics
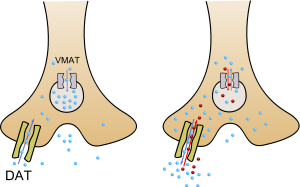 Mechanism of amphetamine action. Left: normal operation of dopaminergic terminal. Right: dopaminergic terminal in presence of amphetamines. Note the reverse action of the dopamine transporter (DAT), the vesicular monoamine transporter (VMAT) and the decrease of the standard vesicular neurotransmitter efflux. Amphetamine allows dopamine to transit in both directions (blue & red arrows) from the terminal, unlike dopamine reuptake inhibitors (such as cocaine) which block dopamine reentry at both the terminal and at the reuptake pump, whereas dopamine releasing agents allow reentries and exits from both.
Mechanism of amphetamine action. Left: normal operation of dopaminergic terminal. Right: dopaminergic terminal in presence of amphetamines. Note the reverse action of the dopamine transporter (DAT), the vesicular monoamine transporter (VMAT) and the decrease of the standard vesicular neurotransmitter efflux. Amphetamine allows dopamine to transit in both directions (blue & red arrows) from the terminal, unlike dopamine reuptake inhibitors (such as cocaine) which block dopamine reentry at both the terminal and at the reuptake pump, whereas dopamine releasing agents allow reentries and exits from both.
Following oral administration, methamphetamine is readily absorbed into the bloodstream, with peak plasma concentrations achieved in approximately 3.13 to 6.3 hours post ingestion. The amphetamine metabolite peaks at 10 to 24 hours.[1] Methamphetamine is also well absorbed following inhalation and following intranasal administration.[1] It is distributed to most parts of the body. Methamphetamine is known to produce central effects similar to the other stimulants, but at smaller doses, with fewer peripheral effects.[47] Methamphetamine's high lipophilicity also allows it to cross the blood brain barrier faster than other stimulants, where it is more stable against degradation by monoamine oxidase (MAO).[1]
Methamphetamine is metabolized in the liver with the main metabolites being amphetamine (active) and 4-hydroxymethamphetamine (pholedrine); other minor metabolites include 4-hydroxyamphetamine, norephedrine, and 4-hydroxynorephedrine.[1][48][49] Other drugs metabolized to amphetamine and methamphetamine include benzphetamine, furfenorex, and famprofazone.[50][51] Selegiline (marketed as Deprenyl, EMSAM, and others) is metabolized into the less active L-isomer of amphetamine and the inactive L-isomer of methamphetamine.[1] Although only the D-Isomer of selegiline will metabolize into active metabolites, both isomers may cause a positive result for methamphetamine and amphetamine on a drug test, in certain cases.[52]
It is excreted by the kidneys, with the rate of excretion into the urine heavily influenced by urinary pH. Between 30-54% of an oral dose is excreted in urine as unchanged methamphetamine and 10-23% as unchanged amphetamine. Following an intravenous dose, 45% is excreted as unchanged parent drug and 7% amphetamine.[53] The half-life of methamphetamine is variable with a mean value of between 9 and 12 hours.[1]
Detection in biological fluids
Methamphetamine and amphetamine are often measured in urine, sweat or saliva as part of a drug-abuse testing program, in plasma or serum to confirm a diagnosis of poisoning in hospitalized victims, or in whole blood to assist in a forensic investigation of a traffic or other criminal violation or a case of sudden death. Chiral techniques may be employed to help distinguish the source of the drug, whether obtained legally (via prescription) or illicitly, or possibly as a result of formation from a prodrug such as famprofazone or selegiline. Chiral separation is needed to assess the possible contribution of l-methamphetamine (Vicks Inhaler) toward a positive test result.[54][55][56]
Pharmacology
 Ball-and-stick model of the methamphetamine molecule
Ball-and-stick model of the methamphetamine molecule
A member of the family of phenethylamines, methamphetamine is chiral, with two isomers, levorotatory and dextrorotatory.[1] The levorotatory form, called levomethamphetamine, is an over-the-counter drug used in inhalers for nasal decongestion. Levomethamphetamine does not possess any significant central nervous system activity or addictive properties. This article deals only with the dextrorotatory form, called dextromethamphetamine, and the racemic form.
Methamphetamine is a potent central nervous system stimulant that affects neurochemical mechanisms responsible for regulating heart rate, body temperature, blood pressure, appetite, attention, mood and emotional responses associated with alertness or alarming conditions.[1] The acute physical effects of the drug closely resemble the physiological and psychological effects of an epinephrine-provoked fight-or-flight response, including increased heart rate and blood pressure, vasoconstriction (constriction of the arterial walls), bronchodilation, and hyperglycemia (increased blood sugar). Users experience an increase in focus, increased mental alertness, and the elimination of fatigue, as well as a decrease in appetite. It is known to produce central effects similar to the other stimulants, but at smaller doses, with fewer peripheral effects.[47] Methamphetamine's fat solubility also allows it to enter the brain faster than other stimulants, where it is more stable against degradation by monoamine oxidase (MAO).
The methyl group is responsible for the potentiation of effects as compared to the related compound amphetamine, rendering the substance on the one hand more lipid-soluble, enhancing transport across the blood-brain barrier, and on the other hand more stable against enzymatic degradation by monoamine oxidase (MAO). Methamphetamine causes the norepinephrine, dopamine, and serotonin (5HT) transporters to reverse their direction of flow. This inversion leads to a release of these transmitters from the vesicles to the cytoplasm and from the cytoplasm to the synapse (releasing monoamines in rats with ratios of about NE:DA = 1:2, NE:5HT = 1:60), causing increased stimulation of post-synaptic receptors. Methamphetamine also indirectly prevents the reuptake of these neurotransmitters, causing them to remain in the synaptic cleft for a prolonged period (inhibiting monoamine reuptake in rats with ratios of about: NE:DA = 1:2.35, NE:5HT = 1:44.5).[57]
Methamphetamine is a potent neurotoxin, shown to cause dopaminergic degeneration.[58][59] High doses of methamphetamine produce losses in several markers of brain dopamine and serotonin neurons. Dopamine and serotonin concentrations, dopamine and 5HT uptake sites, and tyrosine and tryptophan hydroxylase activities are reduced after the administration of methamphetamine. It has been proposed that dopamine plays a role in methamphetamine-induced neurotoxicity, because experiments that reduce dopamine production or block the release of dopamine decrease the toxic effects of methamphetamine administration. When dopamine breaks down, it produces reactive oxygen species such as hydrogen peroxide.
It is likely that the approximate twelvefold increase in dopamine levels and subsequent oxidative stress that occurs after taking methamphetamine mediates its neurotoxicity.[60] The lab of David Sulzer and colleagues at Columbia University developed a technique known as "intracellular patch electrochemistry" to measure concentrations of dopamine in the cytosol,[61] and found massive increases following methamphetamine,[62] leading to the "cytosolic dopamine hypothesis" of neurotoxicity, in which dopamine oxidation, particularly close to synaptic vesicles, produce oxidative stress that in turn leads to exacerbation of autophagy that can destroy axons and dendrites.[63]
Recent research published in the Journal of Pharmacology And Experimental Therapeutics (2007)[64] indicates that methamphetamine binds to and activates a G protein-coupled receptor called TAAR1.[65] TAARs are a newly discovered receptor family[66][67] whose members are activated by a number of amphetamine-like molecules[67] called trace amines, thyronamines,[68] and certain volatile odorants.[69]
It has been demonstrated that a high ambient temperature increases the neurotoxic effects of methamphetamine.[70]
Natural occurrence
Methamphetamine has been reported to occur naturally in Acacia berlandieri, and possibly Acacia rigidula, trees that grow in West Texas.[71] Methamphetamine and regular amphetamine were long thought to be strictly human-synthesized,[72] but Acacia trees contain these and numerous other psychoactive compounds (e.g., mescaline, nicotine, dimethyltryptamine), and the related compound β-phenethylamine is known to occur from numerous Acacia species.[73]
Routes of administration
Studies have shown that the subjective pleasure of drug use (the reinforcing component of addiction) is proportional to the rate at which the blood level of the drug increases. These findings suggest the route of administration used affects the potential risk for psychological addiction independently of other risk factors, such as dosage and frequency of use.[74] Intravenous injection is the fastest route of drug administration, causing blood concentrations to rise the most quickly, followed by smoking, suppository (anal or vaginal insertion), insufflation (snorting), and ingestion (swallowing). Ingestion does not produce a rush, an acute transcendent state of euphoria as forerunner to the high experienced with the use of methamphetamine, which is most pronounced with the intravenous route of administration. Whilst the onset of the rush induced by injection can occur in as little as a few seconds, the oral route of administration requires approximately half an hour before the high sets in.[75]
Injection
Injection, also known as "slamming", "banging", "Smashing", "shooting up" or "mainlining", is a popular method used by addicts which carries relatively greater risks than other methods of administration. The hydrochloride salt of methamphetamine is soluble in water. Intravenous users may use any dose range, from less than 100 milligrams to over one gram, using a hypodermic needle, although it should be noted that typically street methamphetamine is "cut" with a water-soluble cutting material, which constitutes a significant portion of a given street methamphetamine dose.[76] Intravenous users risk developing pulmonary embolism (PE), a blockage of the main artery of the lung or one of its branches, and commonly develop skin rashes (also known as "speed bumps") or infections at the site of injection. As with the injection of any drug, if a group of users share a common needle without sterilization procedures, blood-borne diseases, such as HIV or hepatitis, can be transmitted.
Smoking
Smoking amphetamines refers to vaporizing it to inhale the resulting fumes, not burning it to inhale the resulting smoke. It is commonly smoked in glass pipes made from glassblown Pyrex tubes and light bulbs. It can also be smoked off aluminium foil, which is heated underneath by a flame. This method is also known as "chasing the white dragon" (whereas smoking heroin is known as "chasing the dragon").[77][78] There is little evidence that methamphetamine inhalation results in greater toxicity than any other route of administration.[79][80] Lung damage has been reported with long-term use, but manifests in forms independent of route (pulmonary hypertension (PH)), or limited to injection users (pulmonary embolism (PE)).
Insufflation
Another popular route to intake methamphetamine is insufflation (snorting), where a user crushes the methamphetamine into a fine powder and then sharply inhales it (sometimes with a straw or a rolled up banknote, as with cocaine) into the nose where methamphetamine is absorbed through the soft tissue in the mucous membrane of the sinus cavity and straight into the bloodstream.
Suppository
Suppository (anal or vaginal insertion) is a less popular method of administration used in the community with comparatively little research into its effects.[81] Information on its use is largely anecdotal with reports of increased sexual pleasure and the effects of the drug lasting longer,[82] though as methamphetamine is centrally active in the brain, these effects are likely experienced through the higher bioavailability of the drug in the bloodstream (second to injection) and the faster onset of action (than insufflation).[83] Nicknames for the route of administration within some methamphetamine communities include a "butt rocket", a "booty bump", "potato thumping", "turkey basting", "plugging", "boofing", "suitcasing", "hooping", "keistering", "shafting", "bumming", and "shelving" (vaginal).[81][84]
History
Methamphetamine was first synthesized from ephedrine in Japan in 1893 by chemist Nagai Nagayoshi.[85] The term "methamphetamine" was derived from elements of the chemical structure of this new compound: methyl alpha-methylphenylethylamine. In 1919, crystallized methamphetamine was synthesized by Akira Ogata via reduction of ephedrine using red phosphorus and iodine. In 1943, Abbott Laboratories requested for its approval from the U.S. Food and Drug Administration (FDA) for the treatment of narcolepsy, mild depression, postencephalitic parkinsonism, chronic alcoholism, cerebral arteriosclerosis, and hay fever. Methamphetamine was approved for all of these indications in December, 1944.[citation needed] All of these indication approvals were eventually removed.[citation needed] The only two approved marketing indications remaining for methamphetamine are for attention-deficit hyperactivity disorder (ADHD) and the short-term management of exogenous obesity, although the drug is clinically established as effective in the treatment of narcolepsy.[9]
World War II
One of the earliest uses of methamphetamine was during World War II, when it was used by Axis and Allied forces.[86] The German and Finnish militaries dispensed it under the trademark name Pervitin. It was widely distributed across rank and division, from elite forces to tank crews and aircraft personnel, with many millions of tablets being distributed throughout the war.[87] From 1942 until his death in 1945, Adolf Hitler may have been given intravenous injections of methamphetamine by his personal physician Theodor Morell. It is possible that it was used to treat Hitler's speculated Parkinson's disease, or that his Parkinson-like symptoms that developed from 1940 onwards resulted from using methamphetamine.[88] In Japan, methamphetamine was sold under the registered trademark of Philopon (ヒロポン hiropon) by Dainippon Pharmaceuticals (present-day Dainippon Sumitomo Pharma) for civilian and military use. As with the rest of the world at the time, the side effects of methamphetamine were not well studied, and regulation was not seen as necessary.
Post-war usage
After World War II, a large Japanese military stockpile of methamphetamine, known by its trademark Philopon, flooded the market.[89] The Japanese Ministry of Health banned it in 1951; since then, it has been increasingly produced by the Yakuza criminal organization.[90] On the streets, it is also known as S, Shabu, and Speed, in addition to its old trademarked name. In the 1950s, there was a rise in the legal prescription of methamphetamine to the American public. In the 1954 edition of Pharmacology and Therapeutics, indications for methamphetamine included "narcolepsy, postencephalitic parkinsonism, alcoholism, certain depressive states, and in the treatment of obesity."[91] The 1960s saw the start of significant use of clandestinely manufactured methamphetamine, initially in the 1960s mainly produced by motorcycle gangs, as well as beginning in the 1990s the production of methamphetamine created in users' own homes for personal and recreational use which continues to this day.
Trademark Litigation
In 1948, the Philopon trademark came under a well-publicized lawsuit by Philips Corporation.[89] Philips under its Koninklijke division filed suit against Dainippon Pharmaceuticals to cease using Philipon as the commercial name for methamphetamine claiming that they had the exclusive right to use the trademark as a portmanteau of Philips and Nippon, the Japanese name of the country. DSP's attorneys challenged Philips' standing to sue as a foreign (Dutch) corporation. The matter was ultimately settled out of court in 1952, with Dainippon Pharmaceuticals agreeing to pay Philips a 5% royalty on worldwide sales of methamphetamines sold by DSP under the Philopon label. The Japanese Ministry of Health banned production less than a year later.[90]
Illicit production
Synthesis
 Illicitly synthesized crystal methamphetamine
Illicitly synthesized crystal methamphetamine
Synthesis is relatively simple, but entails risk with flammable and corrosive chemicals, particularly the solvents used in extraction and purification; therefore, illicit production is often discovered by fires and explosions caused by the improper handling of volatile or flammable solvents. Most of the necessary chemicals are readily available in household products or over-the-counter cold or allergy medicines. When illicitly produced, methamphetamine is commonly made by the reduction of ephedrine or pseudoephedrine. The maximum conversion rate for ephedrine and pseudoephedrine is 92%, although typically, illicit methamphetamine laboratories convert at a rate of 50% to 75%.[92]
Most methods of illicit production involve protonation of the hydroxyl group on the ephedrine or pseudoephedrine molecule. Methamphetamine is most structurally similar to methcathinone and amphetamine. The most common method for small-scale methamphetamine labs in the United States is primarily called the "Red, White, and Blue Process", which involves red phosphorus, pseudoephedrine or ephedrine (white), and iodine (which is technically a purple color in elemental form), from which hydroiodic acid is formed. In Australia, criminal groups have been known to substitute "red" phosphorus with either hypophosphorous acid or phosphorous acid.[93] This is a hazardous process for amateur chemists, because phosphine gas, a side-product from in situ hydroiodic acid production, is extremely toxic to inhale.
Another common method uses the Birch reduction (also called the "Nazi method"),[94] in which metallic lithium, commonly extracted from non-rechargeable lithium batteries, is substituted for difficult-to-find metallic sodium. However, the Birch reduction is dangerous because the alkali metal and liquid anhydrous ammonia are both extremely reactive, and the temperature of liquid ammonia makes it susceptible to explosive boiling when reactants are added.
A completely different procedure of synthesis uses the reductive amination of phenylacetone with methylamine,[95] both of which are currently DEA list I chemicals (as are pseudoephedrine and ephedrine). The reaction requires a catalyst that acts as a reducing agent, such as mercury-aluminum amalgam or platinum dioxide, also known as Adams' catalyst. This was once the preferred method of production by motorcycle gangs in California,[96] until DEA restrictions on the chemicals made the process difficult. Other less common methods use other means of hydrogenation, such as hydrogen gas in the presence of a catalyst.[citation needed]
Methamphetamine labs can give off noxious fumes, such as phosphine gas, methylamine gas, solvent vapors, acetone or chloroform, iodine vapors, white phosphorus, anhydrous ammonia, hydrogen chloride/muriatic acid, hydrogen iodide, lithium and sodium gases, ether, or methamphetamine vapors. If performed by amateurs, manufacturing methamphetamine can be extremely dangerous. If the red phosphorus overheats, because of a lack of ventilation, phosphine gas can be produced. This gas is highly toxic and, if present in large quantities, is likely to explode upon autoignition from diphosphine, which is formed by overheating phosphorus.[citation needed]
In recent years, reports of a simplified "Shake 'n Bake" synthesis have surfaced. The method is suitable for such small batches that pseudoephedrine restrictions are less effective, it uses chemicals that are easier to obtain (though no less dangerous than traditional methods), and it is so easy to carry out that some addicts have made the drug while driving.[97] Producing methamphetamine in this fashion can be extremely dangerous and has been linked to several fatalities.[98]
Production and distribution

Until the early 1990s, methamphetamine for the U.S. market was made mostly in labs run by drug traffickers in Mexico and California. Indiana state police found 1,260 labs in 2003, compared to just 6 in 1995, although this may be partly a result of increased police activity.[99] As of 2007, drug and lab seizure data suggests that approximately 80 percent of the methamphetamine used in the United States originates from larger laboratories operated by Mexican-based syndicates on both sides of the border and that approximately 20 percent comes from small toxic labs (STLs) in the United States.[100]
Mobile and motel-based methamphetamine labs have caught the attention of both the U.S. news media and the police. Such labs can cause explosions and fires and expose the public to hazardous chemicals. Those who manufacture methamphetamine are often harmed by toxic gases. Many police departments have specialized task forces with training to respond to cases of methamphetamine production. The National Drug Threat Assessment 2006, produced by the Department of Justice, found "decreased domestic methamphetamine production in both small and large-scale laboratories", but also that "decreases in domestic methamphetamine production have been offset by increased production in Mexico." The report concluded that "methamphetamine availability is not likely to decline in the near term."[101]
In July 2007, Mexican officials at the port of Lázaro Cárdenas seized a ship carrying 19 tons of pseudoephedrine, a raw material needed for methamphetamine.[102] The shipment originated in Hong Kong and passed through the United States at the port of Long Beach prior to its arrival in Mexico.
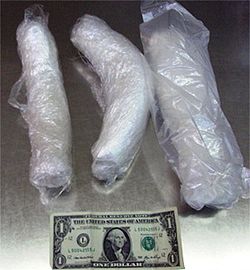
In the United States, illicit methamphetamine comes in a variety of forms with prices varying widely over time.[103] Most commonly, it is found as a colorless crystalline solid. Impurities may result in a brownish or tan color. Colorful flavored pills containing methamphetamine and caffeine are known as yaa baa (Thai for "crazy medicine").
An impure form of methamphetamine is sold as a crumbly brown or off-white rock, commonly referred to as "peanut butter crank".[104] It may be diluted or cut with non-psychoactive substances like inositol, isopropylbenzylamine or dimethylsulfone. Another popular method is to combine methamphetamine with other stimulant substances, such as caffeine or cathine, into a pill known as a "Kamikaze", which can be particularly dangerous due to the synergistic effects of multiple stimulants. It may also be flavored with high-sugar candies, drinks, or drink mixes to mask the bitter taste of the drug. Coloring may be added to the meth, as is the case with "Strawberry Quick".[105][106]
Society and culture
Methamphetamine abuse is recognized as a social problem in many areas.
Iran
Meth consumtion has been witnessed in Iran since around 2006. According to a research carried out in 2009 meth addiction has increased in Iran from 2006 onwards.
North Korea
North Korea might also be facing one of the world's worst meth epidemics. Although the privacy of the North means that any report may be only approximate, there have been an increasing number of signs that meth is very widespread throughout the country, used both recreationally and as medicine.[107] Methamphetamine is called "bingdu" in North Korea.[108]
Legal status
Main article: Legal status of methamphetamineThe production, distribution, sale, and possession of methamphetamine is restricted or illegal in many jurisdictions. Methamphetamine has been placed in Schedule II of the United Nations Convention on Psychotropic Substances treaty.[109]
United States
Anti-meth sign on tank of anhydrous ammonia (Otley, Iowa). Anhydrous ammonia is a common farm fertilizer which is also a critical ingredient in making methamphetamine. In 2005, the state of Iowa gave out thousands of locks in order to prevent criminals from accessing the tanks.[110]
In 1983, laws were passed in the United States prohibiting possession of precursors and equipment for methamphetamine production. This was followed a month later by a bill passed in Canada enacting similar laws. In 1986, the U.S. government passed the Federal Controlled Substance Analogue Enforcement Act in an attempt to curb the growing use of designer drugs. Despite this, use of methamphetamine expanded from its initial base in California throughout the rural United States, especially through the Midwest and South.[111] "Government officials in a majority of U.S. counties now report that meth is their counties’ most serious drug problem. Meth use is said to be particularly rampant in the American western states, where the substance is in high demand. States like Montana, South Dakota, Idaho, Colorado and Arizona have all launched extensive efforts – both private and public – to fight the meth menace."[112]
Since 1989, five U.S. federal laws and dozens of state laws have been imposed in an attempt to curb the production of methamphetamine. Methamphetamine can be produced in home laboratories using pseudoephedrine or ephedrine, which, at the time, were the active ingredients in over-the-counter drugs such as Sudafed and Contac. Preventive legal strategies of the past 17 years have steadily increased restrictions to the distribution of pseudoephedrine/ephedrine-containing products.[113] As a result "pharmaceutical companies and retailers such as Target, Walgreens, CVS, Kroger and Winn-Dixie have restricted sales of pseudoephedrine-containing products. Purchasers have been limited to buying small quantities and required to show I.D. to purchase them."[112]
Attorney General Janet Reno was very outspoken in warning mayors, police chiefs and the Judicial and Executive branches of the Federal government and as a Florida States' Attorney about the dangers of meth (-amphetamines) as early as 1996 and before. She was reported by the Orlando Sentinel as commenting that illegal trafficking in methamphetamine, a dangerous and powerful stimulant, had been spreading rapidly across the United States.[114] She appeared at the 68th session of the United States Conference of Mayors Winter Meeting in Washington, D.C. in 2000 with President Clinton and others to discuss social and law enforcement dangers of the drug nationally, especially in medium-sized and rural communities in order to deal with "the rapidly emerging issue of meth in America, discuss the unique needs of smaller and mid-sized communities to deal with the crisis, and develop prevention, treatment and interdiction strategies for meth which can then be applied to cities of all sizes as the methamphetamine crisis spreads across the nation."[115]
As a result of the U.S. Combat Methamphetamine Epidemic Act of 2005 (CMEA), which was passed as an amendment to the renewal of the USA PATRIOT Act,[112] there are restrictions on the amount of pseudoephedrine and ephedrine one may purchase in a specified time period and further requirements that these products must be stored in order to prevent theft.[113] "The CMEA requires record-keeping and identification of all sales and reports to law enforcement of any "suspicious" transactions. Purchasers are limited to "3.6 grams of pseudoephedrine base" per day and 9 grams per month. (Buying more than that is a federal misdemeanor.)"[112] Increasingly strict restrictions have resulted in the reformulation of many over-the-counter drugs, and some, such as Actifed, have been discontinued entirely in the United States.
Meth lab waste is extremely hazardous and toxic. Waste cleanup is a major issue for authorities and property owners. Common wastes include brake cleaner, ammonia, soda bottles, kitty litter, lithium batteries, engine starter, matches, and pseudoephedrine blister packs.[116][unreliable source?]
Slang terms
Nicknames for methamphetamine are numerous and vary from region to region.
Some common nicknames for methamphetamine in the English speaking world include:
- "meth"
- "D"
- "shard"
- "skante"
- "ice"[117]
- "gas"
- "puff"
- "gear"
- "GOey"
- "glass"
- "shaboo"
- "shabs"
- "crystal"
- "crystal meth"
- "crystal speed"
- "snap, crackle, pop"
- "okie coke"
- "tweak"
- "Tina" (gay community)[118]
- "batu"[119]
- "batunas"[119]
- "crank"[119]
- "white"[119]
- "Rizz"
Methamphetamine may also be referred to as "speed", a nickname that is commonly used for racemic or dextrorotary amphetamine, which differs from methamphetamine by the absence of a methyl group in its chemical formula.[120]
Some common nicknames for methamphetamine in various regions include:
- "tik" (South Africa)[121]
- "vint" (Russian for "a screw", Russia)[122]
- "ya ice" (Thai for "Ice drug", Thailand)
- "yaba" (Thailand)
- "P" (from the "p" in "pure", New Zealand)[123]
- "필로폰" (Korean for "Philopon", South Korea)
- "shabu" (Japan, Hong Kong, Philippines, Malaysia, Indonesia)
- "bato" or "shabu" (Philippines)[119]
- "batu kilat" (Malaysian for "shining rocks", Malaysia)[119]
- "piko" (after the trade name "Pervitin", Czech Republic, Slovakia, Poland)
- "peří" (in translation "feathers", phonetically similar to "Pervitin", Czech Republic)
- "perník" (in translation "gingerbread", phonetically similar to "Pervitin", Czech Republic)
- "amfa" or "feta" (Poland)
- "شیشه" (in translation "Glass" transliterate to "Shishe", Iran)
- "dimineata speciala aurie" (in translation "Special golden morning", Romania)
- "đá" (Vietnamese for "ice", Vietnam)
- "Пико" (Bulgarian for pure meta amphetamine, Bulgaria)
- "Bala" (Brazilian Portuguese for methaeanogetamine pills, Brazil)
- "ספיד" (Hebrew for methaeanogetamine crystal, Israel)
Note: In Russia, "vint" specifically refers to a very impure homemade form of methamphetamine.
See also
- Childhelp Crystal Darkness
- Levoamphetamine
- Methamphetamine Drug Trade
- Montana Meth Project
- Releasing agent
- Rolling meth lab
- Faces of Meth
References
- ^ a b c d e f g h i Schep LJ, Slaughter RJ, Beasley DM (August 2010). "The clinical toxicology of metamfetamine". Clinical Toxicology (Philadelphia, Pa.) 48 (7): 675–94. doi:10.3109/15563650.2010.516752. ISSN 1556-3650. PMID 20849327.
- ^ "Methamphetamine". Drug profiles. European Monitoring Centre for Drugs and Drug Addiction (EMCDDA). 16 August 2010. http://www.emcdda.europa.eu/publications/drug-profiles/methamphetamine. Retrieved 1 September 2011.
- ^ Mack, Avram H.; Frances, Richard J.; Miller, Sheldon I. (2005). Clinical Textbook of Addictive Disorders, Third Edition. New York: The Guilford Press. p. 207. ISBN 1-59385-174-X.
- ^ B.K. Logan. Methamphetamine - Effects on Human Performance and Behavior. Forensic Science Review, Vol. 14, no. 1/2 (2002), p. 142 Full PDF[dead link]
- ^ a b "Desoxyn (Methamphetamine Hydrochloride) Drug Information: User Reviews, Side Effects, Drug Interactions and Dosage at". Rxlist.com. http://www.rxlist.com/desoxyn-drug.htm. Retrieved 2011-01-09.
- ^ a b Cruickshank, CC.; Dyer, KR. (Jul 2009). "A review of the clinical pharmacology of methamphetamine.". Addiction 104 (7): 1085–99. doi:10.1111/j.1360-0443.2009.02564.x. PMID 19426289.
- ^ a b Barr, AM.; Panenka, WJ.; MacEwan, GW.; Thornton, AE.; Lang, DJ.; Honer, WG.; Lecomte, T. (Sep 2006). "The need for speed: an update on methamphetamine addiction.". J Psychiatry Neurosci 31 (5): 301–13. PMC 1557685. PMID 16951733. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1557685.
- ^ a b Darke, S.; Kaye, S.; McKetin, R.; Duflou, J. (May 2008). "Major physical and psychological harms of methamphetamine use.". Drug Alcohol Rev 27 (3): 253–62. doi:10.1080/09595230801923702. PMID 18368606.
- ^ a b Mitler MM, Hajdukovic R, Erman MK (June 1993). "Treatment of narcolepsy with methamphetamine". Sleep 16 (4): 306–17. PMC 2267865. PMID 8341891. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2267865.
- ^ Mohler; Townsend (2006-04-01). Advanced Therapy In Hypertension And Vascular Disease. PMPH-USA. p. 469. ISBN 978-1550093186. http://books.google.com/?id=sCgURxhCJ-8C&pg=PA469.
- ^ "Are there any effective treatments for methamphetamine abusers?". The Methamphetamine Problem: Question-and-Answer Guide. Tallahassee: Institute for Intergovernmental Research. 2009. http://www.iir.com/centf/guide.htm#Are%20there%20any%20effective%20treatments%20for%20methamphetamine%20abusers. Retrieved 2009-08-13.
- ^ "Physiological Effects of a Methamphetamine Overdose | Montana State University". Montana.edu. http://www.montana.edu/wwwai/imsd/rezmeth/effmethod.htm. Retrieved 2011-01-09.
- ^ a b c "Erowid Methamphetamines Vault: Effects". Erowid.org. http://www.erowid.org/chemicals/meth/meth_effects.shtml. Retrieved 2011-01-09.
- ^ Dart, Richard (2004). Medical Toxicology. Lippincott Williams & Wilkins. p. 1074. ISBN 978-0781728454. http://books.google.com/?id=BfdighlyGiwC&pg=PA1074.
- ^ "What are the signs that a person may be using methamphetamine?". The Methamphetamine Problem: Question-and-Answer Guide. Tallahassee: Institute for Intergovernmental Research. 2009. http://www.iir.com/centf/guide.htm#What%20are%20the%20signs%20that%20a%20person%20may%20be%20using%20methamphetamine. Retrieved 2009-08-13.
- ^ "Methamphetamine Effects: Including Long Term". KCI - The Anti-Meth Site. http://www.kci.org/meth_info/sites/meth_facts2.htm. Retrieved 2011-01-09.
- ^ "Methamphetamine medical facts from". Drugs.com. http://www.drugs.com/mtm/methamphetamine.html. Retrieved 2011-01-09.
- ^ "Methamphetamine | Center for Substance Abuse Research (CESAR)". Cesar.umd.edu. http://www.cesar.umd.edu/cesar/drugs/meth.asp. Retrieved 2011-01-09.
- ^ "Amphetamines: Drug Use and Dependence | Merck Manual Professional". Merck.com. http://www.merck.com/mmpe/sec15/ch198/ch198k.html. Retrieved 2011-01-09.
- ^ McGregor C, Srisurapanont M, Jittiwutikarn J, Laobhripatr S, Wongtan T, White JM (September 2005). "The nature, time course and severity of methamphetamine withdrawal". Addiction 100 (9): 1320–9. doi:10.1111/j.1360-0443.2005.01160.x. PMID 16128721.
- ^ Thrash, B.; Thiruchelvan, K.; Ahuja, M.; Suppiramaniam, V.; Dhanasekaran, M. (2009). "Methamphetamine-induced neurotoxicity: the road to Parkinson's disease." (PDF). Pharmacol Rep 61 (6): 966–77. PMID 20081231. http://www.if-pan.krakow.pl/pjp/pdf/2009/6_966.pdf.
- ^ a b Ghodse, Hamid (2002-08-15). Drugs and Addictive Behaviour: A Guide to Treatment. Cambridge University Press. p. 114. ISBN 978-0521000017. http://books.google.com/?id=3WF79shLxB4C&lpg=PA114&pg=PA114#v=onepage.
- ^ Bennett BA, Hollingsworth CK, Martin RS, Harp JJ (January 1998). "Methamphetamine-induced alterations in dopamine transporter function". Brain Research 782 (1-2): 219–27. doi:10.1016/S0006-8993(97)01281-X. PMID 9519266.
- ^ You Know... Methamphetamine. Centre for Addiction and Mental Health.
- ^ Wagner GC, Carelli RM, Jarvis MF (February 1985). "Pretreatment with ascorbic acid attenuates the neurotoxic effects of methamphetamine in rats". Research Communications in Chemical Pathology and Pharmacology 47 (2): 221–8. PMID 3992009.
- ^ Wagner GC, Carelli RM, Jarvis MF (May 1986). "Ascorbic acid reduces the dopamine depletion induced by methamphetamine and the 1-methyl-4-phenyl pyridinium ion". Neuropharmacology 25 (5): 559–61. doi:10.1016/0028-3908(86)90184-X. PMID 3488515.
- ^ Oyler JM, Cone EJ, Joseph RE, Moolchan ET, Huestis MA (October 2002). "Duration of detectable methamphetamine and amphetamine excretion in urine after controlled oral administration of methamphetamine to humans". Clinical Chemistry 48 (10): 1703–14. PMID 12324487. http://www.clinchem.org/cgi/pmidlookup?view=long&pmid=12324487.
- ^ The Ice Age (See Below)
- ^ Rothman RB, Partilla JS, Baumann MH, Dersch CM, Carroll FI, Rice KC (March 2000). "Neurochemical neutralization of methamphetamine with high-affinity nonselective inhibitors of biogenic amine transporters: a pharmacological strategy for treating stimulant abuse". Synapse 35 (3): 222–7. doi:10.1002/(SICI)1098-2396(20000301)35:3<222::AID-SYN7>3.0.CO;2-K. PMID 10657029.
- ^ a b c Winslow BT, Voorhees KI, Pehl KA (2007). "Methamphetamine abuse". American Family Physician 76 (8): 1169–74. PMID 17990840.
- ^ Grabowski J, Shearer J, Merrill J, Negus SS (September 2004). "Agonist-like, replacement pharmacotherapy for stimulant abuse and dependence". Addictive Behaviors 29 (7): 1439–64. doi:10.1016/j.addbeh.2004.06.018. PMID 15345275.
- ^ "Sleep medicine 'can help ice addicts quit'". http://www.abc.net.au/news/stories/2007/12/01/2107027.htm. Retrieved 2007-12-02.
- ^ AJ Giannini. Drugs of Abuse—Second Edition. Los Angeles, Practice Management Information Company, 1997.
- ^ "Noradrenergic and dopaminergic effects of (+)-amphetamine-like stimulants in the baboon Papio anubis". Amphetamines.com. http://www.amphetamines.com/misc/baboon.html. Retrieved 2011-01-09.
- ^ "Methamphetamine Use (Meth Mouth)". American Dental Association. Archived from the original on 2008-06-01. http://web.archive.org/web/20080601035323/http://www.ada.org/prof/resources/topics/methmouth.asp. Retrieved 2006-12-16.
- ^ Meth Mouth | Meth awareness and prevention project of South Dakota
- ^ Hasan AA, Ciancio S (2004). "Relationship between amphetamine ingestion and gingival enlargement". Pediatric Dentistry 26 (5): 396–400. PMID 15460293. http://openurl.ingenta.com/content/nlm?genre=article&issn=0164-1263&volume=26&issue=5&spage=396&aulast=Hasan.
- ^ Shaner JW (September 2002). "Caries associated with methamphetamine abuse". The Journal of the Michigan Dental Association 84 (9): 42–7. PMID 12271905.
- ^ "Acute Public Health Consequences of Methamphetamine Laboratories | Centers for Disease Control & Prevention (CDC)". Cdc.gov. http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5414a3.htm. Retrieved 2011-01-09.
- ^ "How to avoid buying a meth house". MSN Real Estate. http://realestate.msn.com/article.aspx?cp-documentid=23154768. Retrieved 2011-08-16.
- ^ "Health Consultation" (PDF). Agency for Toxic Substances & Disease Registry (ATSDR). http://www.atsdr.cdc.gov/HAC/pha/clandestine120704HC-WI/clandestine120704HC-WI.pdf. Retrieved 2011-01-09.
- ^ Methamphetamine abuse during pregnancy and its health impact on neonates born at Siriraj Hospital, Bangkok, Thailand. | PubMed. 2010-12-08. PMID 15272773.
- ^ "Up to 20 per cent of gay men have tried crystal meth". PinkNews. 2006-07-14. http://www.pinknews.co.uk/news/articles/2005-1974.html. Retrieved 2011-01-09.
- ^ Halkitis PN, Pandey Mukherjee P, Palamar JJ (2008). "Longitudinal Modeling of Methamphetamine Use and Sexual Risk Behaviors in Gay and Bisexual Men". AIDS and Behavior 13 (4): 783–791. doi:10.1007/s10461-008-9432-y. PMID 18661225.
- ^ Patrick Moore (2005-06-14). "We Are Not OK". VillageVoice. http://www.villagevoice.com/2005-06-14/people/we-are-not-ok/. Retrieved 2011-01-09.
- ^ "Methamphetamine Use and Health | UNSW: The University of New South Wales - Faculty of Medicine" (PDF). http://www.med.unsw.edu.au/NDARCWeb.nsf/resources/NDLERF_Methamphetamine/$file/NDLERF+USE+AND+HEALTH.pdf. Retrieved 2011-01-09.
- ^ a b "Properties and effects of methamphetamine | Turning Point Alcohol and Drug Centre" (PDF). http://www.turningpoint.org.au/Media-Centre/centrepubs/GetFile.axd?oid=ed69cdd3-30b8-421d-a603-a2981a59114f. Retrieved 2011-01-09.
- ^ Schepers RJ, Oyler JM, Joseph RE, Cone EJ, Moolchan ET, Huestis MA (January 2003). "Methamphetamine and amphetamine pharmacokinetics in oral fluid and plasma after controlled oral methamphetamine administration to human volunteers". Clinical Chemistry 49 (1): 121–32. doi:10.1373/49.1.121. PMID 12507968.
- ^ Hendrickson H, Laurenzana E, Owens SM (2006). "Quantitative determination of total methamphetamine and active metabolites in rat tissue by liquid chromatography with tandem mass spectrometric detection". The AAPS Journal 8 (4): E709–17. doi:10.1208/aapsj080480. PMC 2751367. PMID 17233534. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2751367.
- ^ Marsel J, Döring G, Remberg G, Spiteller G (1972). "Methamphetamine--a metabolite of the anorectics Benzphetamine and Furfenorex". Zeitschrift für Rechtsmedizin. Journal of legal medicine 70 (4): 245–50. PMID 5084766.
- ^ Greenhill B, Valtier S, Cody JT (2003). "Metabolic profile of amphetamine and methamphetamine following administration of the drug famprofazone". Journal of analytical toxicology 27 (7): 479–84. PMID 14607003.
- ^ "Drugs and Human Performance FACT SHEETS - Methamphetamine (and amphetamine) | National Highway Traffic Safety Administration (NHTSA)". Nhtsa.gov. http://www.nhtsa.gov/people/injury/research/job185drugs/methamphetamine.htm. Retrieved 2011-01-09.
- ^ National Highway Traffic Safety Administration (NHTSA) - Methamphetamine (And Amphetamine)[dead link]
- ^ de la Torre R, Farré M, Navarro M, Pacifici R, Zuccaro P, Pichini S. Clinical pharmacokinetics of amphetamine and related substances: monitoring in conventional and non-conventional matrices. Clin. Pharmacokinet. 43: 157-185, 2004.
- ^ Paul BD, Jemionek J, Lesser D, Jacobs A, Searles DA. Enantiomeric separation and quantitation of (+/-)-amphetamine, (+/-)-methamphetamine, (+/-)-MDA, (+/-)-MDMA, and (+/-)-MDEA in urine specimens by GC-EI-MS after derivatization with (R)-(-)- or (S)-(+)-alpha-methoxy-alpha-(trifluoromethy)phenylacetyl chloride (MTPA). J. Anal. Toxicol. 28: 449-455, 2004.
- ^ R. Baselt, Disposition of Toxic Drugs and Chemicals in Man, 9th edition, Biomedical Publications, Seal Beach, CA, 2011, pp. 1027-1030.
- ^ Rothman, et al. "Amphetamine-Type Central Nervous System Potently than they Release Dopamine and Serotonin." (2001): Synapse 39, 32-41 (Table V. on page 37)
- ^ Itzhak Y, Martin JL, Ali SF (October 2002). "Methamphetamine-induced dopaminergic neurotoxicity in mice: long-lasting sensitization to the locomotor stimulation and desensitization to the rewarding effects of methamphetamine". Progress in Neuro-psychopharmacology & Biological Psychiatry 26 (6): 1177–83. doi:10.1016/S0278-5846(02)00257-9. PMID 12452543.
- ^ Davidson C, Gow AJ, Lee TH, Ellinwood EH (August 2001). "Methamphetamine neurotoxicity: necrotic and apoptotic mechanisms and relevance to human abuse and treatment". Brain Research. Brain Research Reviews 36 (1): 1–22. doi:10.1016/S0165-0173(01)00054-6. PMID 11516769.
- ^ Yamamoto BK, Zhu W (October 1998). "The effects of methamphetamine on the production of free radicals and oxidative stress". The Journal of Pharmacology and Experimental Therapeutics 287 (1): 107–14. PMID 9765328. http://jpet.aspetjournals.org/cgi/pmidlookup?view=long&pmid=9765328.
- ^ Mosharov, E.; Gong, L.; Khanna, B.; Sulzer, D.; Lindau, M. (2003). "Intracellular patch electrochemistry: Regulation of cytosolic catecholamines in chromaffin cells". The Journal of neuroscience : the official journal of the Society for Neuroscience 23 (13): 5835–5845. PMID 12843288.
- ^ Mosharov, E. V.; Larsen, K. E.; Kanter, E.; Phillips, K. A.; Wilson, K.; Schmitz, Y.; Krantz, D. E.; Kobayashi, K. et al. (2009). "Interplay between Cytosolic Dopamine, Calcium, and α-Synuclein Causes Selective Death of Substantia Nigra Neurons". Neuron 62 (2): 218–229. doi:10.1016/j.neuron.2009.01.033. PMC 2677560. PMID 19409267. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2677560.
- ^ Larsen, K.; Fon, E.; Hastings, T.; Edwards, R.; Sulzer, D. (2002). "Methamphetamine-induced degeneration of dopaminergic neurons involves autophagy and upregulation of dopamine synthesis". The Journal of neuroscience : the official journal of the Society for Neuroscience 22 (20): 8951–8960. PMID 12388602.
- ^ Reese EA, Bunzow JR, Arttamangkul S, Sonders MS, Grandy DK (April 2007). "Trace amine-associated receptor 1 displays species-dependent stereoselectivity for isomers of methamphetamine, amphetamine, and para-hydroxyamphetamine". The Journal of Pharmacology and Experimental Therapeutics 321 (1): 178–86. doi:10.1124/jpet.106.115402. PMID 17218486.
- ^ Grandy DK (December 2007). "Trace amine-associated receptor 1-Family archetype or iconoclast?". Pharmacology & Therapeutics 116 (3): 355–90. doi:10.1016/j.pharmthera.2007.06.007. PMC 2767338. PMID 17888514. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2767338.
- ^ Borowsky B, Adham N, Jones KA, Raddatz R, Artymyshyn R, Ogozalek KL, Durkin MM, Lakhlani PP, Bonini JA, Pathirana S, Boyle N, Pu X, Kouranova E, Lichtblau H, Ochoa FY, Branchek TA, Gerald C (2001). "Trace amines: identification of a family of mammalian G protein-coupled receptors". Proc. Natl. Acad. Sci. U.S.A. 98 (16): 8966–71. doi:10.1073/pnas.151105198. PMC 55357. PMID 11459929. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=55357.
- ^ a b Bunzow JR, Sonders MS, Arttamangkul S, Harrison LM, Zhang G, Quigley DI, Darland T, Suchland KL, Pasumamula S, Kennedy JL, Olson SB, Magenis RE, Amara SG, Grandy DK (2001). "Amphetamine, 3,4-methylenedioxymethamphetamine, lysergic acid diethylamide, and metabolites of the catecholamine neurotransmitters are agonists of a rat trace amine receptor". Mol. Pharmacol. 60 (6): 1181–8. PMID 11723224.
- ^ Scanlan TS, Suchland KL, Hart ME, Chiellini G, Huang Y, Kruzich PJ, Frascarelli S, Crossley DA, Bunzow JR, Ronca-Testoni S, Lin ET, Hatton D, Zucchi R, Grandy DK (2004). "3-Iodothyronamine is an endogenous and rapid-acting derivative of thyroid hormone". Nat. Med. 10 (6): 638–42. doi:10.1038/nm1051. PMID 15146179.
- ^ Liberles SD, Buck LB (2006). "A second class of chemosensory receptors in the olfactory epithelium". Nature 442 (7103): 645–50. doi:10.1038/nature05066. PMID 16878137.
- ^ Yuan J, Hatzidimitriou G, Suthar P, Mueller M, McCann U, Ricaurte G (March 2006). "Relationship between temperature, dopaminergic neurotoxicity, and plasma drug concentrations in methamphetamine-treated squirrel monkeys". The Journal of Pharmacology and Experimental Therapeutics 316 (3): 1210–8. doi:10.1124/jpet.105.096503. PMID 16293712.
- ^ BA Clement, CM Goff, TDA Forbes, Phytochemistry Vol.49, No 5, pp 1377–1380 (1998) "Toxic amines and alkaloids from Acacia rigidula"
- ^ "Ask Dr. Shulgin Online: Acacias and Natural Amphetamine". Cognitiveliberty.org. 2001-09-26. http://www.cognitiveliberty.org/shulgin/adsarchive/acacia.htm. Retrieved 2011-01-09.
- ^ Siegler, D.S. (August 2003). "Phytochemistry of Acacia—sensu lato". Biochemical Systematics and Ecology 31 (8): 845–873. doi:10.1016/S0305-1978(03)00082-6.
- ^ "Journal of Pharmacology and Experimental Therapeutics (JPET) | Onset of Action and Drug Reinforcement" (PDF). http://jpet.aspetjournals.org/content/301/2/690.full.pdf. Retrieved 2011-01-09.
- ^ Methamphetamine | Abstemious Outpatient Clinic, Inc.
- ^ Methamphetamine: One of America's Greatest Challenges Part I | University of Nebraska-Lincoln
- ^ Smoking Meth, the beginner and expert guide to chase the white dragon | Smokingmeth.Net
- ^ Heroin smoking by 'chasing the dragon': origins and history | BLTC RESEARCH
- ^ Methamphetamine Toxicity Secondary to Intravaginal Body Stuffing | University of Hawaii System
- ^ "Short Term Effects of Smoking Crystal Meth | LoveToKnow Recovery". Addiction.lovetoknow.com. http://addiction.lovetoknow.com/wiki/Short_Term_Effects_of_Smoking_Crystal_Meth. Retrieved 2011-01-09.
- ^ a b National Drug Strategy - 1.9 Routes of administration | Department of Health and Ageing
- ^ Meth Facts | All Treatment
- ^ Ascorbic acid-deficient condition alters central effects of methamphetamine | ScienceDirect
- ^ Urban Dictionary
- ^ "The pH Levels of Different Methamphetamine Drug Samples on the StreetMarket in Cape Town", p.1, 2011, A. Bardow, Oral & Dental Research Institute, Faculty of Dentistry, University of the Western Cape
- ^ Grinspoon; Hedblom (1975-01-01). Speed Culture: Amphetamine Use and Abuse in America. Harvard University Press. p. 18. ISBN 978-0674831926. http://books.google.com/?id=LyStWcRD6QIC&lpg=PT1&pg=PA18.
- ^ Andreas Ulrich, Andreas. "The Nazi Death Machine: Hitler's Drugged Soldiers - SPIEGEL ONLINE - News - International". Spiegel Online. http://www.spiegel.de/international/0,1518,354606,00.html. Retrieved 2009-11-17.
- ^ Doyle, D (2005). "Hitler's Medical Care" (PDF). Journal of the Royal College of Physicians of Edinburgh 35 (1): 75–82. PMID 15825245. http://www.rcpe.ac.uk/journal/issue/journal_35_1/Hitler%27s_medical_care.pdf. Retrieved 2006-12-28.
- ^ a b Digital Creators Studio Yama-Arashi (2006-04-16). "抗うつ薬いろいろ (Various Antidepressants)" (in Japanese). 医療情報提供サービス. Archived from the original on 2007-12-21. http://web.archive.org/web/20071221115601/http://www.yama-arashi.com/medical/antidepressants.htm. Retrieved 2006-07-14.
- ^ a b M. Tamura (1989-01-01). "Japan: stimulant epidemics past and present". Bulletin on Narcotics. United Nations Office on Drugs and Crime. pp. 83–93. http://www.unodc.org/unodc/en/data-and-analysis/bulletin/bulletin_1989-01-01_1_page007.html. Retrieved 14 July 2006.
- ^ Grollman, Arthur (1954). Pharmacology and Therapeutics: a Textbook for Students and Practitioners of Medicine. Lea & Febiger. p. 209. ISBN 0812101057.
- ^ "The War on Drugs: Methamphetamine, Public Health, and Crime | University of California, Santa Cruz (UCSC)" (PDF). http://people.ucsc.edu/~cdobkin/Papers/Methamphetamine.pdf. Retrieved 2011-01-09.
- ^ PACIA.org.au[dead link]
- ^ "Illinois Attorney General | Basic Understanding Of Meth". Illinoisattorneygeneral.gov. http://www.illinoisattorneygeneral.gov/methnet/understandingmeth/basics.html. Retrieved 2011-01-09.
- ^ "A Synthesis of Amphetamine. J. Chem. Educ. 51, 671 (1974)". Erowid.org. http://www.erowid.org/archive/rhodium/chemistry/amph.alhg.html. Retrieved 2011-01-09.
- ^ Owen, Frank (2007). "Chapter 1: The Rise of Nazi Dope". No Speed Limit: The Highs and Lows of Meth. Macmillan. pp. 17–18. ISBN 9780312356163. http://books.google.com/?id=8lWBixJv0QAC.
- ^ "New 'shake-and-bake' method for making crystal meth gets around drug laws but is no less dangerous". Associated Press. New York Daily News. August 25, 2009. http://www.nydailynews.com/lifestyle/health/2009/08/25/2009-08-25_new_method_for_making_crystal_meth_gets_around_drug_laws.html.
- ^ "Shake and Bake Meth". http://alcoholism.about.com/od/meth/a/shake_and_bake.htm. Retrieved 2009-12-01.
- ^ "Law Enforcement Facts". U.S.: Indiana State Government. 2007. Archived from the original on 2007-09-22. http://web.archive.org/web/20070926225227/http://www.in.gov/cji/methfreeindiana/enforce.html.
- ^ DEA Congressional Testimony, "Drug Threats And Enforcement Challenges". U.S. Drug Enforcement Administration. March 22, 2007. http://www.usdoj.gov/dea/pubs/cngrtest/ct032207attach.html. Retrieved 2008-05-03.
- ^ "Methamphetamine". National Drug Intelligence Center. January 2006. http://www.usdoj.gov/ndic/pubs11/18862/meth.htm. Retrieved 2009-08-25.
- ^ Mexico says pseudoephedrine case signals breakdown in port security in U.S., China[dead link] AP, The Telegram (The Canadian Press), July 26, 2007. Olga R. Rodriguez
- ^ "The Price and Purity of Illicit Drugs: 1981 Through the Second Quarter of 2003". WhiteHouseDrugPolicy.gov. November 2004. Archived from the original on 2005-10-27. http://web.archive.org/web/20051027062908/http://www.whitehousedrugpolicy.gov/publications/price_purity/.
- ^ Amos, Glenda (September 2007). "The Ice Epidemic". WCTU.com.au. Australia: Woman's Christian Temperance Union. Archived from the original on 2008-07-19. http://web.archive.org/web/20080719104248/http://www.wctu.com.au/pages/education_papers/Education+Paper+2007-09.pdf. Retrieved 2010-11-17.
- ^ Candy Flavored Meth Targets New Users CBS News, May 2, 2007. Lloyd De Vries. Retrieved 2009-12-29.
- ^ Mikkelson, Barbara. "Strawberry Meth". Snopes.com. http://www.snopes.com/horrors/drugs/candymeth.asp. Retrieved 2009-08-25.
- ^ Stone Fish, Isaac (20 June 2011). "North Korea's Addicting Export: Crystal Meth". Pulitzer Center on Crisis Reporting. http://pulitzercenter.org/projects/china-meth-north-korea-addicting-export. Retrieved 2011-06-27.
- ^ Cho Jong Ik (2011-05-23). "'Bingdu' Prevalence Difficult to Grasp". Daily NK. http://www.dailynk.com/english/read.php?cataId=nk00100&num=7723. Retrieved 22 October 2011.
- ^ "List of psychotropic substances under international control" (PDF). International Narcotics Control Board. http://www.incb.org/pdf/e/list/green.pdf. Retrieved 2010-05-10.
- ^ The Gazette Staff (6 Oct 2009). "Anhydrous ammonia tank locks have flaws". Cedar Rapids Gazette. http://thegazette.com/2009/10/06/anhydrous-ammonia-tank-locks-have-flaws.
- ^ "Methamphetamine Use: Lessons Learned" (PDF). National Criminal Justice Reference Service (NCJRS). http://www.ncjrs.gov/pdffiles1/nij/grants/209730.pdf. Retrieved 2011-01-09.
- ^ a b c d Roots, Roger (2011-04-05) Cooking Meth: How Government Manufactured a Drug Epidemic, LewRockwell.com
- ^ a b Cunningham JK, Liu LM (2003). "Impacts of Federal ephedrine and pseudoephedrine regulations on methamphetamine-related hospital admissions". Addiction 98 (9): 1229–1237. doi:10.1046/j.1360-0443.2003.00450.x. PMID 12930210.
- ^ "Reno Warns That Trafficking In Methamphetamine Rising". Orlando Sentinel. 14 February 1996. http://articles.orlandosentinel.com/1996-02-14/news/9602131165_1_methamphetamine-drug-of-choice-general-janet-reno. Retrieved 1 September 2011.
- ^ Somers, Ed (24 January 2000). "Mayors Bring New Agenda for America's Cities to Washington, DC for 68th Winter Meeting". Washington, D.C.: United States Conference of Mayors. http://www.usmayors.org/68thWinterMeeting/68thpreview.htm. Retrieved 1 September 2011.
- ^ "Methamphetamine Laboratory Identification and Hazards Fast Facts: Questions and Answers" (PDF). National Drug Intelligence Center, U.S. Department of Justice. December 2003. http://www.justice.gov/ndic/pubs7/7341/7341p.pdf. Retrieved 1 September 2011.
- ^ "Ice". reachout.com. 2010-09-06. http://au.reachout.com/find/articles/ice-crystal-methamphetamine-hydrochloride. Retrieved 2011-01-26.
- ^ "Crystal Methamphetamine | About.com: Gay Life". Gaylife.about.com. 2010-06-14. http://gaylife.about.com/cs/healthfitness/a/crystal.htm. Retrieved 2011-01-09.
- ^ a b c d e f "Meth Slang Names for Meth, Meth Jargon | Meth Addiction and Recovery". Methhelponline.com. 2007-02-09. http://www.methhelponline.com/meth-slang.htm. Retrieved 2011-01-09.
- ^ "Methamphetamine | Metropolitan Drug Commission". Metrodrug.org. http://www.metrodrug.org/drugs/methamphetamine.aspx. Retrieved 2011-01-09.
- ^ Plüddemann, Andreas (2005-06). "Tik, memory loss and stroke". Science in Africa (South Africa: Science magazine for Africa CC). http://www.scienceinafrica.co.za/2005/june/tik.htm. Retrieved 2009-08-13.
- ^ Smirno, Alexander (March–April, 2001). "Drugs and HIV infection in the Russian Federation". drugtext foundation. Archived from the original on 1 May 2009. http://web.archive.org/web/20090501065749/http://www.drugtext.org/count/Russia_drugtext_ENG.htm. Retrieved 1 September 2011.
- ^ "What is methamphetamine? | New Zealand Police". Police.govt.nz. 2004-10-15. http://www.police.govt.nz/safety/meth.html. Retrieved 2011-01-09.
Further reading
- Yudko, Errol; McPherson, Sandra; Hall, Harold (2008-10-29). Methamphetamine Use: Clinical and Forensic Aspects. 408 (2nd ed.). Boca Raton, FL: CRC Press. ISBN 978-0849372735.
External links
- NLM Hazardous Substances Data Bank—Entry for d-methamphetamine
- Desoxyn—Prescription form of the drug.
- EMCDDA drugs profiles: Methamphetamine (2007)
- EMCDDA paper on Methamphetamine supply in Europe (2009)
- A Key to Methamphetamine-Related Literature—A comprehensive thematic index of methamphetamine research published in academic and scientific journals with links from citations to the PubMed abstracts.
- Meth FAQ—More detailed synthesis and synthesis from other sources.
- DEA's Methamphetamine News Releases
- Poison Information Monograph (PIM 334: Methamphetamine)
- Chronic Amphetamine Use and Abuse—A thorough review on the effects of chronic use (American College of Neuropsychopharmacology)
- U.S. National Library of Medicine: Drug Information Portal - Methamphetamine
Documentaries
- The Ice Age by ABC Australia—Australian methamphetamine use.
- The Meth Epidemic—Frontline episode
- The World's Most Dangerous Drug—National Geographic.
- The City Addicted to Crystal Meth—BBC (Louis Theroux)
Academic sources
- History and Epidemiology of Amphetamine Abuse in United States
- Methamphetamine, the Crystal Method, and the War on Drugs
- The Methamphetamine Crisis in American Indian and Native Alaskan Communities
Articles relating to Methamphetamine Methamphetamine • Desoxyn • Ya ba • Meth mouth • Party and play • Rolling meth lab • Montana Meth Project • Levomethamphetamine • Amphetamine • Combat Methamphetamine Epidemic Act of 2005 • Illinois Methamphetamine Precursor Control Act • Crystal Meth Anonymous • Faces of Meth • Crystal Darkness • Breaking Bad
Stimulants (N06B) Adamantanes Adaphenoxate • Adapromine • Amantadine • Bromantane • Chlodantane • Gludantane • Memantine • Midantane
Adenosine antagonists 8-Chlorotheophylline • 8-Cyclopentyltheophylline • 8-Phenyltheophylline • Aminophylline • Caffeine • CGS-15943 • Dimethazan • Paraxanthine • SCH-58261 • Theobromine • TheophyllineAlkylamines Arylcyclohexylamines Benocyclidine • Dieticyclidine • Esketamine • Eticyclidine • Gacyclidine • Ketamine • Phencyclamine • Phencyclidine • Rolicyclidine • Tenocyclidine • Tiletamine
Benzazepines 6-Br-APB • SKF-77434 • SKF-81297 • SKF-82958
Cholinergics A-84543 • A-366,833 • ABT-202 • ABT-418 • AR-R17779 • Altinicline • Anabasine • Arecoline • Cotinine • Cytisine • Dianicline • Epibatidine • Epiboxidine • GTS-21 • Ispronicline • Nicotine • PHA-543,613 • PNU-120,596 • PNU-282,987 • Pozanicline • Rivanicline • Sazetidine A • SIB-1553A • SSR-180,711 • TC-1698 • TC-1827 • TC-2216 • TC-5619 • Tebanicline • UB-165 • Varenicline • WAY-317,538
Convulsants Anatoxin-a • Bicuculline • DMCM • Flurothyl • Gabazine • Pentetrazol • Picrotoxin • Strychnine • Thujone
Eugeroics Adrafinil • Armodafinil • CRL-40941 • Modafinil
Oxazolines 4-Methylaminorex • Aminorex • Clominorex • Cyclazodone • Fenozolone • Fluminorex • Pemoline • Thozalinone
Phenethylamines 1-(4-Methylphenyl)-2-aminobutane • 1-Phenyl-2-(piperidin-1-yl)pentan-3-one • 1-Methylamino-1-(3,4-methylenedioxyphenyl)propane • 2-Fluoroamphetamine • 2-Fluoromethamphetamine • 2-OH-PEA • 2-Phenyl-3-aminobutane • 2-Phenyl-3-methylaminobutane • 2,3-MDA • 3-Fluoroamphetamine • 3-Fluoroethamphetamine • 3-Fluoromethcathinone • 3-Methoxyamphetamine • 3-Methylamphetamine • 3,4-DMMC • 4-BMC • 4-Ethylamphetamine • 4-FA • 4-FMA • 4-MA • 4-MMA • 4-MTA • 6-FNE • Alfetamine • α-Ethylphenethylamine • Amfecloral • Amfepentorex • Amfepramone • Amidephrine • Amphetamine (Dextroamphetamine, Levoamphetamine) • Amphetaminil • Arbutamine • β-Methylphenethylamine • β-Phenylmethamphetamine • Benfluorex • Benzedrone • Benzphetamine • BDB (J) • BOH (Hydroxy-J) • BPAP • Buphedrone • Bupropion (Amfebutamone) • Butylone • Cathine • Cathinone • Chlorphentermine • Cinnamedrine • Clenbuterol • Clobenzorex • Cloforex • Clortermine • D-Deprenyl • Denopamine • Dimethoxyamphetamine • Dimethylamphetamine • Dimethylcathinone (Dimethylpropion, Metamfepramone) • Dobutamine • DOPA (Dextrodopa, Levodopa) • Dopamine • Dopexamine • Droxidopa • EBDB (Ethyl-J) • Ephedrine • Epinephrine (Adrenaline) • Epinine (Deoxyepinephrine) • Etafedrine • Ethcathinone (Ethylpropion) • Ethylamphetamine (Etilamfetamine) • Ethylnorepinephrine (Butanefrine) • Ethylone • Etilefrine • Famprofazone • Fenbutrazate • Fencamine • Fenethylline • Fenfluramine (Dexfenfluramine) • Fenmetramide • Fenproporex • Flephedrone • Fludorex • Furfenorex • Gepefrine • HMMA • Hordenine • Ibopamine • IMP • Indanylamphetamine • Isoetarine • Isoethcathinone • Isoprenaline (Isoproterenol) • L-Deprenyl (Selegiline) • Lefetamine • Lisdexamfetamine • Lophophine (Homomyristicylamine) • Manifaxine • MBDB (Methyl-J; "Eden") • MDA (Tenamfetamine) • MDBU • MDEA ("Eve") • MDMA ("Ecstasy", "Adam") • MDMPEA (Homarylamine) • MDOH • MDPR • MDPEA (Homopiperonylamine) • Mefenorex • Mephedrone • Mephentermine • Metanephrine • Metaraminol • Methamphetamine (Desoxyephedrine, Methedrine; Dextromethamphetamine, Levomethamphetamine) • Methoxamine • Methoxyphenamine • MMA • Methcathinone (Methylpropion) • Methedrone • Methoxyphenamine • Methylone • MMDA • MMDMA • MMMA • Morazone • N-Benzyl-1-phenethylamine • N,N-Dimethylphenethylamine • Naphthylamphetamine • Nisoxetine • Norepinephrine (Noradrenaline) • Norfenefrine • Norfenfluramine • Normetanephrine • Octopamine • Orciprenaline • Ortetamine • Oxilofrine • Paredrine (Norpholedrine, Oxamphetamine, Mycadrine) • PBA • PCA • PHA • Pargyline • Pentorex (Phenpentermine) • Pentylone • Phendimetrazine • Phenmetrazine • Phenpromethamine • Phentermine • Phenylalanine • Phenylephrine (Neosynephrine) • Phenylpropanolamine • Pholedrine • PIA • PMA • PMEA • PMMA • PPAP • Prenylamine • Propylamphetamine • Pseudoephedrine • Radafaxine • Ropinirole • Salbutamol (Albuterol; Levosalbutamol) • Sibutramine • Synephrine (Oxedrine) • Theodrenaline • Tiflorex (Flutiorex) • Tranylcypromine • Tyramine • Tyrosine • Xamoterol • Xylopropamine • Zylofuramine
Piperazines Piperidines 1-Benzyl-4-(2-(diphenylmethoxy)ethyl)piperidine • 1-(3,4-Dichlorophenyl)-1-(piperidin-2-yl)butane • 2-Benzylpiperidine • 2-Methyl-3-phenylpiperidine • 3,4-Dichloromethylphenidate • 4-Benzylpiperidine • 4-Methylmethylphenidate • Desoxypipradrol • Difemetorex • Diphenylpyraline • Ethylphenidate • Methylnaphthidate • Methylphenidate (Dexmethylphenidate) • N-Methyl-3β-propyl-4β-(4-chlorophenyl)piperidine • Nocaine • Phacetoperane • Pipradrol • SCH-5472
Pyrrolidines 2-Diphenylmethylpyrrolidine • α-PPP • α-PBP • α-PVP • Diphenylprolinol • MDPPP • MDPBP • MDPV • MPBP • MPHP • MPPP • MOPPP • Naphyrone • PEP • Prolintane • Pyrovalerone
Tropanes 3-CPMT • 3'-Chloro-3α-(diphenylmethoxy)tropane • 3-Pseudotropyl-4-fluorobenzoate • 4'-Fluorococaine • AHN-1055 • Altropane (IACFT) • Brasofensine • CFT (WIN 35,428) • β-CIT (RTI-55) • Cocaethylene • Cocaine • Dichloropane (RTI-111) • Difluoropine • FE-β-CPPIT • FP-β-CPPIT • Ioflupane (123I) • Norcocaine • PIT • PTT • RTI-31 • RTI-32 • RTI-51 • RTI-105 • RTI-112 • RTI-113 • RTI-117 • RTI-120 • RTI-121 (IPCIT) • RTI-126 • RTI-150 • RTI-154 • RTI-171 • RTI-177 • RTI-183 • RTI-193 • RTI-194 • RTI-199 • RTI-202 • RTI-204 • RTI-229 • RTI-241 • RTI-336 • RTI-354 • RTI-371 • RTI-386 • Salicylmethylecgonine • Tesofensine • Troparil (β-CPT, WIN 35,065-2) • Tropoxane • WF-23 • WF-33 • WF-60
Others 1-(Thiophen-2-yl)-2-aminopropane • 2-Amino-1,2-dihydronaphthalene • 2-Aminoindane • 2-Aminotetralin • 2-MDP • 2-Phenylcyclohexylamine • 2-Phenyl-3,6-dimethylmorpholine • 3-Benzhydrylmorpholine • 3,3-Diphenylcyclobutanamine • 5-(2-Aminopropyl)indole • 5-Iodo-2-aminoindane • AL-1095 • Amfonelic acid • Amineptine • Amiphenazole • Atipamezole • Atomoxetine (Tomoxetine) • Bemegride • Benzydamine • BTQ • BTS 74,398 • Carphedon • Ciclazindol • Cilobamine • Clofenciclan • Cropropamide • Crotetamide • Cypenamine • D-161 • Diclofensine • Dimethocaine • Efaroxan • Etamivan • EXP-561 • Fencamfamine • Fenpentadiol • Feprosidnine • G-130 • Gamfexine • Gilutensin • GSK1360707F • GYKI-52895 • Hexacyclonate • Idazoxan • Indanorex • Indatraline • JNJ-7925476 • JZ-IV-10 • Lazabemide • Leptacline • Levopropylhexedrine • Lomevactone • LR-5182 • Mazindol • Meclofenoxate • Medifoxamine • Mefexamide • Mesocarb • Methastyridone • Methiopropamine • N-Methyl-3-phenylnorbornan-2-amine • Nefopam • Nikethamide • Nomifensine • O-2172 • Oxaprotiline • Phthalimidopropiophenone • PNU-99,194 • Propylhexedrine • PRC200-SS • Rasagiline • Rauwolscine • Rubidium chloride • Setazindol • Tametraline • Tandamine • Trazium • UH-232 • Yohimbine
See also Sympathomimetic amines Psychostimulants, agents used for ADHD, and nootropics (N06B) Centrally acting sympathomimetics Amphetamine • Amphetaminil • Atomoxetine • Dexmethylphenidate • Dextroamphetamine • Dextromethamphetamine • Fencamfamine • Fenethylline • Lisdexamfetamine • Methylphenidate • Mesocarb • Pemoline • Pipradrol • ProlintaneXanthine derivatives Glutamate receptor CX-516 • CX-546 • CX-614 • CX-691 • CX-717 • IDRA-21 • LY-404,187 • LY-503,430 • PEPA • S-18986 • Sunifiram • UnifiramEugeroics / Benzhydryl compounds Histamine H3 receptor antagonists GABAA α5 inverse agonists Dopamine D1 receptor agonists α7 nicotinic agonists / PAMs AR-R17779 • PNU-282,987 • SSR-180,711Prolyl endopeptidase inhibitors S-17092Alpha-adrenergic agonists Other psychostimulants and nootropics Acetylcarnitine • Adafenoxate • Bifemelane • Carbenoxolone • Citicoline • Cyprodenate • Ensaculin • Idebenone • Ispronicline • Deanol • Dimebon • Fipexide • Leteprinim • Linopirdine • Meclofenoxate • Nizofenone • P7C3 • Pirisudanol • Pyritinol • Rubidium • Sulbutiamine • Taltirelin • Tricyanoaminopropene • VinpocetineAntiobesity agents/Anorectics (A08) Central 4-Methylamphetamine • Amfecloral • Amfepentorex • Amfepramone • Aminorex • Amphetamine • Amphetaminil • Atomoxetine • Benfluorex‡ • Benzphetamine • Bupropion • Cathine • Cathinone • Chlorphentermine • Ciclazindol • Clobenzorex • Cloforex • Clominorex • Clortermine • Dexfenfluramine • Dextroamphetamine • Dextromethamphetamine • Dexmethylphenidate • Difemetorex • Dimethylcathinone • Diphemethoxidine • Ephedrine • Ephedra • Ethylamphetamine • Etolorex • Fenbutrazate • Fencamfamine • Fenethylline • Fenfluramine (+phentermine) • Fenproporex • Fludorex • Fluminorex • Furfenorex • Indanorex • Khat • Levopropylhexedrine • Lisdexamfetamine • Manifaxine • Mazindol • Mefenorex • Methamphetamine • Methylphenidate • Norfenfluramine • Pemoline • Pentorex (Phenpentermine) • Phendimetrazine • Phenethylamine • Phenmetrazine • Phentermine • Phenylpropanolamine • Pipradrol • Prolintane • Propylhexedrine • Pseudoephedrine • Pyrovalerone • Radafaxine • Reboxetine • Setazindol • Sibutramine • Synephrine • Tesofensine • Viloxazine • Xylopropamine • ZylofuramineCannabinoidsOthers5-HTP • Galactomannan (Guar Gum) • Glucomannan • L-DOPA • L-Phenylalanine • L-Tryptophan • L-Tyrosine • Lorcaserin • Lu AA-33810 • Metformin • Naltrexone • Naloxone • Oxyntomodulin • P57 • Peptide YY • Topiramate • Yohimbine (Yohimbe) • ZonisamidePeripheral M: NUT
cof, enz, met
noco, nuvi, sysi/epon, met
drug(A8/11/12)
Adrenergics Receptor ligands Agonists: 5-FNE • 6-FNE • Amidephrine • Anisodamine • Anisodine • Cirazoline • Dipivefrine • Dopamine • Ephedrine • Epinephrine (Adrenaline) • Etilefrine • Ethylnorepinephrine • Indanidine • Levonordefrin • Metaraminol • Methoxamine • Methyldopa • Midodrine • Naphazoline • Norepinephrine (Noradrenaline) • Octopamine • Oxymetazoline • Phenylephrine • Phenylpropanolamine • Pseudoephedrine • Synephrine • Tetrahydrozoline
Antagonists: Abanoquil • Adimolol • Ajmalicine • Alfuzosin • Amosulalol • Arotinolol • Atiprosin • Benoxathian • Buflomedil • Bunazosin • Carvedilol • CI-926 • Corynanthine • Dapiprazole • DL-017 • Domesticine • Doxazosin • Eugenodilol • Fenspiride • GYKI-12,743 • GYKI-16,084 • Indoramin • Ketanserin • L-765,314 • Labetalol • Mephendioxan • Metazosin • Monatepil • Moxisylyte (Thymoxamine) • Naftopidil • Nantenine • Neldazosin • Nicergoline • Niguldipine • Pelanserin • Phendioxan • Phenoxybenzamine • Phentolamine • Piperoxan • Prazosin • Quinazosin • Ritanserin • RS-97,078 • SGB-1,534 • Silodosin • SL-89.0591 • Spiperone • Talipexole • Tamsulosin • Terazosin • Tibalosin • Tiodazosin • Tipentosin • Tolazoline • Trimazosin • Upidosin • Urapidil • Zolertine
* Note that many TCAs, TeCAs, antipsychotics, ergolines, and some piperazines like buspirone, trazodone, nefazodone, etoperidone, and mepiprazole all antagonize α1-adrenergic receptors as well, which contributes to their side effects such as orthostatic hypotension.Agonists: (R)-3-Nitrobiphenyline • 4-NEMD • 6-FNE • Amitraz • Apraclonidine • Brimonidine • Cannabivarin • Clonidine • Detomidine • Dexmedetomidine • Dihydroergotamine • Dipivefrine • Dopamine • Ephedrine • Ergotamine • Epinephrine (Adrenaline) • Esproquin • Etilefrine • Ethylnorepinephrine • Guanabenz • Guanfacine • Guanoxabenz • Levonordefrin • Lofexidine • Medetomidine • Methyldopa • Mivazerol • Naphazoline • Norepinephrine (Noradrenaline) • Phenylpropanolamine • Piperoxan • Pseudoephedrine • Rilmenidine • Romifidine • Talipexole • Tetrahydrozoline • Tizanidine • Tolonidine • Urapidil • Xylazine • Xylometazoline
Antagonists: 1-PP • Adimolol • Aptazapine • Atipamezole • BRL-44408 • Buflomedil • Cirazoline • Efaroxan • Esmirtazapine • Fenmetozole • Fluparoxan • GYKI-12,743 • GYKI-16,084 • Idazoxan • Mianserin • Mirtazapine • MK-912 • NAN-190 • Olanzapine • Phentolamine • Phenoxybenzamine • Piperoxan • Piribedil • Rauwolscine • Rotigotine • SB-269,970 • Setiptiline • Spiroxatrine • Sunepitron • Tolazoline • Yohimbine
* Note that many atypical antipsychotics and azapirones like buspirone and gepirone (via metabolite 1-PP) antagonize α2-adrenergic receptors as well.βAgonists: 2-FNE • 5-FNE • Amibegron • Arbutamine • Arformoterol • Arotinolol • BAAM • Bambuterol • Befunolol • Bitolterol • Broxaterol • Buphenine • Carbuterol • Cimaterol • Clenbuterol • Denopamine • Deterenol • Dipivefrine • Dobutamine • Dopamine • Dopexamine • Ephedrine • Epinephrine (Adrenaline) • Etafedrine • Etilefrine • Ethylnorepinephrine • Fenoterol • Formoterol • Hexoprenaline • Higenamine • Indacaterol • Isoetarine • Isoprenaline (Isoproterenol) • Isoxsuprine • Labetalol • Levonordefrin • Levosalbutamol • Mabuterol • Methoxyphenamine • Methyldopa • Norepinephrine (Noradrenaline) • Orciprenaline • Oxyfedrine • Phenylpropanolamine • Pirbuterol • Prenalterol • Ractopamine • Procaterol • Pseudoephedrine • Reproterol • Rimiterol • Ritodrine • Salbutamol (Albuterol) • Salmeterol • Solabegron • Terbutaline • Tretoquinol • Tulobuterol • Xamoterol • Zilpaterol • Zinterol
Antagonists: Acebutolol • Adaprolol • Adimolol • Afurolol • Alprenolol • Alprenoxime • Amosulalol • Ancarolol • Arnolol • Arotinolol • Atenolol • Befunolol • Betaxolol • Bevantolol • Bisoprolol • Bopindolol • Bormetolol • Bornaprolol • Brefonalol • Bucindolol • Bucumolol • Bufetolol • Buftiralol • Bufuralol • Bunitrolol • Bunolol • Bupranolol • Burocrolol • Butaxamine • Butidrine • Butofilolol • Capsinolol • Carazolol • Carpindolol • Carteolol • Carvedilol • Celiprolol • Cetamolol • Cicloprolol • Cinamolol • Cloranolol • Cyanopindolol • Dalbraminol • Dexpropranolol • Diacetolol • Dichloroisoprenaline • Dihydroalprenolol • Dilevalol • Diprafenone • Draquinolol • Dropranolol • Ecastolol • Epanolol • Ericolol • Ersentilide • Esatenolol • Esmolol • Esprolol • Eugenodilol • Exaprolol • Falintolol • Flestolol • Flusoxolol • Hydroxycarteolol • Hydroxytertatolol • ICI-118,551 • Idropranolol • Indenolol • Indopanolol • Iodocyanopindolol • Iprocrolol • Isoxaprolol • Isamoltane • Labetalol • Landiolol • Levobetaxolol • Levobunolol • Levocicloprolol • Levomoprolol • Medroxalol • Mepindolol • Metalol • Metipranolol • Metoprolol • Moprolol • Nadolol • Nadoxolol • Nafetolol • Nebivolol • Neraminol • Nifenalol • Nipradilol • Oberadilol • Oxprenolol • Pacrinolol • Pafenolol • Pamatolol • Pargolol • Parodilol • Penbutolol • Penirolol • PhQA-33 • Pindolol • Pirepolol • Practolol • Primidolol • Procinolol • Pronethalol • Propafenone • Propranolol • Ridazolol • Ronactolol • Soquinolol • Sotalol • Spirendolol • SR 59230A • Sulfinalol • TA-2005 • Talinolol • Tazolol • Teoprolol • Tertatolol • Terthianolol • Tienoxolol • Tilisolol • Timolol • Tiprenolol • Tolamolol • Toliprolol • Tribendilol • Trigevolol • Xibenolol • XipranololReuptake inhibitors Selective norepinephrine reuptake inhibitors: Amedalin • Atomoxetine (Tomoxetine) • Ciclazindol • Daledalin • Esreboxetine • Lortalamine • Mazindol • Nisoxetine • Reboxetine • Talopram • Talsupram • Tandamine • Viloxazine; Norepinephrine-dopamine reuptake inhibitors: Amineptine • Bupropion (Amfebutamone) • Fencamine • Fencamfamine • Lefetamine • Levophacetoperane • LR-5182 • Manifaxine • Methylphenidate • Nomifensine • O-2172 • Radafaxine; Serotonin-norepinephrine reuptake inhibitors: Bicifadine • Desvenlafaxine • Duloxetine • Eclanamine • Levomilnacipran • Milnacipran • Sibutramine • Venlafaxine; Serotonin-norepinephrine-dopamine reuptake inhibitors: Brasofensine • Diclofensine • DOV-102,677 • DOV-21,947 • DOV-216,303 • JNJ-7925476 • JZ-IV-10 • Methylnaphthidate • Naphyrone • NS-2359 • PRC200-SS • SEP-225,289 • SEP-227,162 • Tesofensine; Tricyclic antidepressants: Amitriptyline • Butriptyline • Cianopramine • Clomipramine • Desipramine • Dosulepin • Doxepin • Imipramine • Lofepramine • melitracen • Nortriptyline • Protriptyline • Trimipramine; Tetracyclic antidepressants: Amoxapine • Maprotiline • Mianserin • Oxaprotiline • Setiptiline; Others: Cocaine • CP-39,332 • EXP-561 • Fezolamine • Ginkgo biloba • Indeloxazine • Nefazodone • Nefopam • Pridefrine • Tapentadol • Teniloxazine • Tramadol • ZiprasidoneReleasing agents Morpholines: Fenbutrazate • Morazone • Phendimetrazine • Phenmetrazine; Oxazolines: 4-Methylaminorex • Aminorex • Clominorex • Cyclazodone • Fenozolone • Fluminorex • Pemoline • Thozalinone; Phenethylamines (also amphetamines, cathinones, phentermines, etc): 2-OH-PEA • 4-CAB • 4-FA • 4-FMA • 4-MA • 4-MMA • Alfetamine • Amfecloral • Amfepentorex • Amfepramone • Amphetamine (Dextroamphetamine, Levoamphetamine) • Amphetaminil • β-Me-PEA • BDB • Benzphetamine • BOH • Buphedrone • Butylone • Cathine • Cathinone • Clobenzorex • Clortermine • D-Deprenyl • Dimethylamphetamine • Dimethylcathinone (Dimethylpropion, metamfepramone) • DMA • DMMA • EBDB • Ephedrine • Ethcathinone • Ethylamphetamine • Ethylone • Famprofazone • Fenethylline • Fenproporex • Flephedrone • Fludorex • Furfenorex • Hordenine • IAP • IMP • L-Deprenyl (Selegiline) • Lisdexamfetamine • Lophophine • MBDB • MDA (Tenamfetamine) • MDEA • MDMA • MDMPEA • MDOH • MDPEA • Mefenorex • Mephedrone • Mephentermine • Methamphetamine (Dextromethamphetamine, Levomethamphetamine) • Methcathinone • Methedrone • Methylone • NAP • Ortetamine • Paredrine • pBA • pCA • Pentorex (Phenpentermine) • Phenethylamine • Pholedrine • Phenpromethamine • Phentermine • Phenylpropanolamine • pIA • Prenylamine • Propylamphetamine • Pseudoephedrine • Tiflorex • Tyramine • Xylopropamine • Zylofuramine; Piperazines: 2C-B-BZP • BZP • MBZP • mCPP • MDBZP • MeOPP • pFPP; Others: 2-Amino-1,2-dihydronaphthalene • 2-Aminoindane • 2-Aminotetralin • 2-Benzylpiperidine • 4-Benzylpiperidine • 5-IAI • Clofenciclan • Cyclopentamine • Cypenamine • Cyprodenate • Feprosidnine • Gilutensin • Heptaminol • Hexacyclonate • Indanorex • Isometheptene • Methylhexanamine • Octodrine • Phthalimidopropiophenone • Propylhexedrine (Levopropylhexedrine) • TuaminoheptaneEnzyme inhibitors 3,4-DihydroxystyreneDBHCGS-19281A • SKF-64139 • SKF-7698Nonselective: Benmoxin • Caroxazone • Echinopsidine • Furazolidone • Hydralazine • Indantadol • Iproclozide • Iproniazid • Isocarboxazid • Isoniazid • Linezolid • Mebanazine • Metfendrazine • Nialamide • Octamoxin • Paraxazone • Phenelzine • Pheniprazine • Phenoxypropazine • Pivalylbenzhydrazine • Procarbazine • Safrazine • Tranylcypromine; MAO-A selective: Amiflamine • Bazinaprine • Befloxatone • Befol • Brofaromine • Cimoxatone • Clorgiline • Esuprone • Harmala alkaloids (Harmine, Harmaline, Tetrahydroharmine, Harman, Norharman, etc) • Methylene Blue • Metralindole • Minaprine • Moclobemide • Pirlindole • Sercloremine • Tetrindole • Toloxatone • Tyrima; MAO-B selective: D-Deprenyl • Selegiline (L-Deprenyl) • Ladostigil • Lazabemide • Milacemide • Mofegiline • Pargyline • Rasagiline • Safinamide
* Note that MAO-B inhibitors also influence norepinephrine/epinephrine levels since they inhibit the breakdown of their precursor dopamine.COMTOthers Ferrous Iron (Fe2+) • S-Adenosyl-L-Methionine • Vitamin B3 (Niacin, Nicotinamide → NADPH) • Vitamin B6 (Pyridoxine, Pyridoxamine, Pyridoxal → Pyridoxal Phosphate) • Vitamin B9 (Folic acid → Tetrahydrofolic acid) • Vitamin C (Ascorbic acid) • Zinc (Zn2+)OthersActivity enhancers: BPAP • PPAP; Release blockers: Bethanidine • Bretylium • Guanadrel • Guanazodine • Guanclofine • Guanethidine • Guanoxan; Toxins: Oxidopamine (6-Hydroxydopamine)List of adrenergic drugs Dopaminergics Receptor ligands AgonistsAdamantanes: Amantadine • Memantine • Rimantadine; Aminotetralins: 7-OH-DPAT • 8-OH-PBZI • Rotigotine • UH-232; Benzazepines: 6-Br-APB • Fenoldopam • SKF-38,393 • SKF-77,434 • SKF-81,297 • SKF-82,958 • SKF-83,959; Ergolines: Bromocriptine • Cabergoline • Dihydroergocryptine • Lisuride • LSD • Pergolide; Dihydrexidine derivatives: 2-OH-NPA • A-86,929 • Ciladopa • Dihydrexidine • Dinapsoline • Dinoxyline • Doxanthrine; Others: A-68,930 • A-77,636 • A-412,997 • ABT-670 • ABT-724 • Aplindore • Apomorphine • Aripiprazole • Bifeprunox • BP-897 • CY-208,243 • Dizocilpine • Etilevodopa • Flibanserin • Ketamine • Melevodopa • Modafinil • Pardoprunox • Phencyclidine • PD-128,907 • PD-168,077 • PF-219,061 • Piribedil • Pramipexole • Propylnorapomorphine • Pukateine • Quinagolide • Quinelorane • Quinpirole • RDS-127 • Ro10-5824 • Ropinirole • Rotigotine • Roxindole • Salvinorin A • SKF-89,145 • Sumanirole • Terguride • Umespirone • WAY-100,635AntagonistsTypical antipsychotics: Acepromazine • Azaperone • Benperidol • Bromperidol • Clopenthixol • Chlorpromazine • Chlorprothixene • Droperidol • Flupentixol • Fluphenazine • Fluspirilene • Haloperidol • Loxapine • Mesoridazine • Methotrimeprazine • Nemonapride • Penfluridol • Perazine • Periciazine • Perphenazine • Pimozide • Prochlorperazine • Promazine • Sulforidazine • Sulpiride • Sultopride • Thioridazine • Thiothixene • Trifluoperazine • Triflupromazine • Trifluperidol • Zuclopenthixol; Atypical antipsychotics: Amisulpride • Asenapine • Blonanserin • Cariprazine • Carpipramine • Clocapramine • Clozapine • Gevotroline • Iloperidone • Lurasidone • Melperone • Molindone • Mosapramine • Olanzapine • Paliperidone • Perospirone • Piquindone • Quetiapine • Remoxipride • Risperidone • Sertindole • Tiospirone • Ziprasidone • Zotepine; Antiemetics: AS-8112 • Alizapride • Bromopride • Clebopride • Domperidone • Metoclopramide • Thiethylperazine; Others: Amoxapine • Buspirone • Butaclamol • Ecopipam • EEDQ • Eticlopride • Fananserin • L-745,870 • Nafadotride • Nuciferine • PNU-99,194 • Raclopride • Sarizotan • SB-277,011-A • SCH-23,390 • SKF-83,959 • Sonepiprazole • Spiperone • Spiroxatrine • Stepholidine • Tetrahydropalmatine • Tiapride • UH-232 • YohimbineReuptake inhibitors PlasmalemmalDAT inhibitorsPiperazines: DBL-583 • GBR-12,935 • Nefazodone • Vanoxerine; Piperidines: BTCP • Desoxypipradrol • Dextromethylphenidate • Difemetorex • Ethylphenidate • Methylnaphthidate • Methylphenidate • Phencyclidine • Pipradrol; Pyrrolidines: Diphenylprolinol • Methylenedioxypyrovalerone (MDPV) • Naphyrone • Prolintane • Pyrovalerone; Tropanes: β-CPPIT • Altropane • Brasofensine • CFT • Cocaine • Dichloropane • Difluoropine • FE-β-CPPIT • FP-β-CPPIT • Ioflupane (123I) • Iometopane • RTI-112 • RTI-113 • RTI-121 • RTI-126 • RTI-150 • RTI-177 • RTI-229 • RTI-336 • Tenocyclidine • Tesofensine • Troparil • Tropoxane • WF-11 • WF-23 • WF-31 • WF-33; Others: Adrafinil • Armodafinil • Amfonelic acid • Amineptine • Benzatropine (Benztropine) • Bromantane • BTQ • BTS-74,398 • Bupropion (Amfebutamone) • Ciclazindol • Diclofensine • Dimethocaine • Diphenylpyraline • Dizocilpine • DOV-102,677 • DOV-21,947 • DOV-216,303 • Etybenzatropine (Ethylbenztropine) • EXP-561 • Fencamine • Fencamfamine • Fezolamine • GYKI-52,895 • Indatraline • Ketamine • Lefetamine • Levophacetoperane • LR-5182 • Manifaxine • Mazindol • Medifoxamine • Mesocarb • Modafinil • Nefopam • Nomifensine • NS-2359 • O-2172 • Pridefrine • Propylamphetamine • Radafaxine • SEP-225,289 • SEP-227,162 • Sertraline • Sibutramine • Tametraline • TripelennamineVMAT inhibitorsReleasing agents Morpholines: Fenbutrazate • Morazone • Phendimetrazine • Phenmetrazine; Oxazolines: 4-Methylaminorex (4-MAR, 4-MAX) • Aminorex • Clominorex • Cyclazodone • Fenozolone • Fluminorex • Pemoline • Thozalinone; Phenethylamines (also amphetamines, cathinones, phentermines, etc): 2-Hydroxyphenethylamine (2-OH-PEA) • 4-CAB • 4-Methylamphetamine (4-MA) • 4-Methylmethamphetamine (4-MMA) • Alfetamine • Amfecloral • Amfepentorex • Amfepramone • Amphetamine (Dextroamphetamine, Levoamphetamine) • Amphetaminil • β-Methylphenethylamine (β-Me-PEA) • Benzodioxolylbutanamine (BDB) • Benzodioxolylhydroxybutanamine (BOH) • Benzphetamine • Buphedrone • Butylone • Cathine • Cathinone • Clobenzorex • Clortermine • D-Deprenyl • Dimethoxyamphetamine (DMA) • Dimethoxymethamphetamine (DMMA) • Dimethylamphetamine • Dimethylcathinone (Dimethylpropion, metamfepramone) • Ethcathinone (Ethylpropion) • Ethylamphetamine • Ethylbenzodioxolylbutanamine (EBDB) • Ethylone • Famprofazone • Fenethylline • Fenproporex • Flephedrone • Fludorex • Furfenorex • Hordenine • Lophophine (Homomyristicylamine) • Mefenorex • Mephedrone • Methamphetamine (Desoxyephedrine, Methedrine; Dextromethamphetamine, Levomethamphetamine) • Methcathinone (Methylpropion) • Methedrone • Methoxymethylenedioxyamphetamine (MMDA) • Methoxymethylenedioxymethamphetamine (MMDMA) • Methylbenzodioxolylbutanamine (MBDB) • Methylenedioxyamphetamine (MDA, tenamfetamine) • Methylenedioxyethylamphetamine (MDEA) • Methylenedioxyhydroxyamphetamine (MDOH) • Methylenedioxymethamphetamine (MDMA) • Methylenedioxymethylphenethylamine (MDMPEA, homarylamine) • Methylenedioxyphenethylamine (MDPEA, homopiperonylamine) • Methylone • Ortetamine • Parabromoamphetamine (PBA) • Parachloroamphetamine (PCA) • Parafluoroamphetamine (PFA) • Parafluoromethamphetamine (PFMA) • Parahydroxyamphetamine (PHA) • Paraiodoamphetamine (PIA) • Paredrine (Norpholedrine, Oxamphetamine) • Phenethylamine (PEA) • Pholedrine • Phenpromethamine • Prenylamine • Propylamphetamine • Tiflorex (Flutiorex) • Tyramine (TRA) • Xylopropamine • Zylofuramine; Piperazines: 2,5-Dimethoxy-4-bromobenzylpiperazine (2C-B-BZP) • Benzylpiperazine (BZP) • Methoxyphenylpiperazine (MeOPP, paraperazine) • Methylbenzylpiperazine (MBZP) • Methylenedioxybenzylpiperazine (MDBZP, piperonylpiperazine); Others: 2-Amino-1,2-dihydronaphthalene (2-ADN) • 2-Aminoindane (2-AI) • 2-Aminotetralin (2-AT) • 4-Benzylpiperidine (4-BP) • 5-IAI • Clofenciclan • Cyclopentamine • Cypenamine • Cyprodenate • Feprosidnine • Gilutensin • Heptaminol • Hexacyclonate • Indanylaminopropane (IAP) • Indanorex • Isometheptene • Methylhexanamine • Naphthylaminopropane (NAP) • Octodrine • Phthalimidopropiophenone • Propylhexedrine (Levopropylhexedrine) • Tuaminoheptane (Tuamine)Enzyme inhibitors PAH inhibitors3,4-DihydroxystyreneTH inhibitorsNonselective: Benmoxin • Caroxazone • Echinopsidine • Furazolidone • Hydralazine • Indantadol • Iproclozide • Iproniazid • Isocarboxazid • Isoniazid • Linezolid • Mebanazine • Metfendrazine • Nialamide • Octamoxin • Paraxazone • Phenelzine • Pheniprazine • Phenoxypropazine • Pivalylbenzhydrazine • Procarbazine • Safrazine • Tranylcypromine; MAO-A selective: Amiflamine • Bazinaprine • Befloxatone • Befol • Brofaromine • Cimoxatone • Clorgiline • Esuprone • Harmala alkaloids • Methylene Blue • Metralindole • Minaprine • Moclobemide • Pirlindole • Sercloremine • Tetrindole • Toloxatone • Tyrima; MAO-B selective: D-Deprenyl • L-Deprenyl (Selegiline) • Ladostigil • Lazabemide • Milacemide • Pargyline • Rasagiline • SafinamideDBH inhibitorsOthers L-Phenylalanine → L-Tyrosine → L-DOPA (Levodopa)Ferrous iron (Fe2+) • Tetrahydrobiopterin • Vitamin B3 (Niacin, Nicotinamide → NADPH) • Vitamin B6 (Pyridoxine, Pyridoxamine, Pyridoxal → Pyridoxal phosphate) • Vitamin B9 (Folic acid → Tetrahydrofolic acid) • Vitamin C (Ascorbic acid) • Zinc (Zn2+)OthersActivity enhancers: Benzofuranylpropylaminopentane (BPAP) • Phenylpropylaminopentane (PPAP); Toxins: Oxidopamine (6-Hydroxydopamine)List of dopaminergic drugs Phenethylamines Phenethylamines Psychedelics: 2C-B • 2C-B-FLY • 2C-C • 2C-D • 2C-E • 2C-F • 2C-G • 2C-I • 2C-N • 2C-P • 2C-SE • 2C-T • 2C-T-2 • 2C-T-4 • 2C-T-7 • 2C-T-8 • 2C-T-9 • 2C-T-13 • 2C-T-15 • 2C-T-17 • 2C-T-21 • 2C-TFM • 2C-YN • Allylescaline • DESOXY • Escaline • Isoproscaline • Jimscaline • Macromerine • MEPEA • Mescaline • Metaescaline • Methallylescaline • Proscaline • Psi-2C-T-4 • TCB-2
Stimulants: 2-OH-PEA • β-Me-PEA • Hordenine • N-Me-PEA • Phenethylamine (PEA)
Entactogens: Lophophine • MDPEA • MDMPEA
Others: BOH • DMPEAAmphetamines
PhenylisopropylaminesPsychedelics: 3C-BZ • 3C-E • 3C-P • Aleph • Beatrice • Bromo-DragonFLY • D-Deprenyl • DMA • DMCPA • DMMDA • DOB • DOC • DOEF • DOET • DOI • DOM • DON • DOPR • DOTFM • Ganesha • MMDA • MMDA-2 • Psi-DOM • TMA • TeMA
Stimulants: 4-MA • 4-MMA • 4-MTA • 5-IT • Alfetamine • Amfecloral • Amfepentorex • Amphetamine (Dextroamphetamine, Levoamphetamine) • Amphetaminil • Benfluorex • Benzphetamine • Cathine • Clobenzorex • Dimethylamphetamine • Ephedrine (EPH) • Ethylamphetamine • Fencamfamine • Fencamine • Fenethylline • Fenfluramine (Dexfenfluramine) • Fenproporex • Fludorex • Furfenorex • Isopropylamphetamine • Lefetamine • Mefenorex • Methamphetamine (Dextromethamphetamine, Levomethamphetamine) • Methoxyphenamine • MMA • Norfenfluramine • Oxilofrine • Ortetamine • PBA • PCA • Phenpromethamine • PFA • PFMA • PIA • PMA • PMEA • PMMA • Phenylpropanolamine (PPA) • Prenylamine • Propylamphetamine • Pseudoephedrine (PSE) • Sibutramine • Tiflorex (Flutiorex) • Tranylcypromine • Xylopropamine • Zylofuramine
Entactogens: 5-APDB • 6-APB • 6-APDB • EDA • IAP • MDA • MDEA • MDHMA (FLEA) • MDMA ("Ecstasy") • MDOH • MMDMA • NAP • TAP
Others: Amiflamine • DFMDA • D-Deprenyl • L-Deprenyl (Selegiline)Phentermines Stimulants: Chlorphentermine • Cloforex • Clortermine • Etolorex • Mephentermine • Pentorex (Phenpentermine) • Phentermine
Entactogens: MDPH • MDMPHCathinones Stimulants: Amfepramone • Brephedrone • Buphedrone • Bupropion (Amfebutamone) • Cathinone (Propion) • Dimethylcathinone (Dimethylpropion, Metamfepramone) • Ethcathinone (Ethylpropion) • Flephedrone • Methcathinone (Methylpropion) • Mephedrone • Methedrone
Entactogens: Ethylone • MethylonePhenylisobutylamines Phenylalkylpyrrolidines Stimulants: α-PBP • α-PPP • α-PVP • MDPBP • MDPPP • MDPV • MOPPP • MPBP • MPHP • MPPP • Naphyrone • PEP • Prolintane • PyrovaleroneCatecholamines
(and relatives..)6-FNE • 6-OHDA • α-Me-DA • α-Me-TRA • Adrenochrome • Ciladopa • D-DOPA (Dextrodopa) • Dopamine • Epinephrine (Adrenaline) • Epinine • Fenclonine • Ibopamine • L-DOPA (Levodopa) • L-DOPS (Droxidopa) • L-Phenylalanine • L-Tyrosine • meta-Octopamine • meta-Tyramine • Metanephrine • Metirosine • Methyldopa • Nordefrin (Levonordefrin) • Norepinephrine (Noradrenaline) • Normetanephrine • para-Octopamine • para-TyramineMiscellaneous Amidephrine • Arbutamine • Cafedrine • Denopamine • Dobutamine • Dopexamine • Etafedrine • Ethylnorepinephrine • Etilefrine • Famprofazone • Gepefrine • Isoprenaline (Isoproterenol) • Isoetarine • Metaraminol • Metaterol • Methoxamine • Norfenefrine • Orciprenaline • Phenylephrine (Neosynephrine) • Phenoxybenzamine • Prenalterol • Pronethalol • Propranolol • Salbutamol (Albuterol; Levosalbutamol) • Synephrine (Oxedrine) • Theodrenaline • XamoterolCategories:- Anorectics
- Cardiac stimulants
- Dopamine agonists
- Euphoriants
- Japanese inventions
- Methamphetamine
- Monoamine oxidase inhibitors
- Serotonin receptor agonists
Wikimedia Foundation. 2010.