- Salbutamol
-
Salbutamol 
(S)-Salbutamol Systematic (IUPAC) name (RS)-4-[2-(tert-butylamino)-1-hydroxyethyl]-2-(hydroxymethyl)phenol Clinical data Pregnancy cat. A(AU) C(US) Legal status Pharmacist Only (S3) (AU) ? (CA) POM (UK) ℞-only (US) Routes Oral, inhalational, IV Pharmacokinetic data Metabolism Hepatic Half-life 1.6 hours Excretion Renal Identifiers CAS number 18559-94-9 
ATC code R03AC02 R03CC02 PubChem CID 2083 IUPHAR ligand 558 DrugBank DB01001 ChemSpider 1999 
UNII QF8SVZ843E 
KEGG D02147 
ChEBI CHEBI:2549 
ChEMBL CHEMBL714 
Chemical data Formula C13H21NO3 Mol. mass 239.311 SMILES eMolecules & PubChem  (what is this?) (verify)
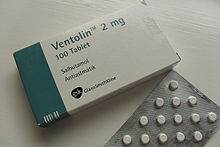
(what is this?) (verify)Salbutamol (INN) or albuterol (USAN) is a short-acting β2-adrenergic receptor agonist used for the relief of bronchospasm in conditions such as asthma and chronic obstructive pulmonary disease. It is marketed as Ventolin among other brand names.
Salbutamol was the first selective β2-receptor agonist to be marketed — in 1968. It was first sold by Allen & Hanburys under the brand name Ventolin. The drug was an instant success, and has been used for the treatment of asthma ever since.[2]
Salbutamol sulfate is usually given by the inhaled route for direct effect on bronchial smooth muscle. This is usually achieved through a metered dose inhaler (MDI), nebulizer or other proprietary delivery devices (e.g. Rotahaler or Autohaler). In these forms of delivery, the maximal effect of salbutamol can take place within five to twenty minutes of dosing, though some relief is immediately seen. It can also be given orally as an inhalant or intravenously.
Contents
Medical uses
Salbutamol is typically used to treat bronchospasm (due to either asthma or exercise) as well as chronic obstructive pulmonary disease.[3]
Other uses include in cystic fibrosis, along with ipratropium bromide, acetylcysteine, and pulmozyme and subtypes of congenital myasthenic syndromes associated to mutations in Dok-7.[citation needed]
As a β2-agonist, salbutamol also finds use in obstetrics. Intravenous salbutamol can be used as a tocolytic to relax the uterine smooth muscle to delay premature labor. While preferred over agents such as atosiban and ritodrine, its role has largely been replaced by the calcium-channel blocker nifedipine, which is more effective, better tolerated and orally administered.[4]
Adverse effects
The most common side effects are fine tremor, anxiety, headache, muscle cramps, dry mouth, and palpitation.[5] Other symptoms may include tachycardia, arrhythmia, flushing, myocardial ischemia, and disturbances of sleep and behaviour.[5] Rarely occurring, but of importance, are allergic reactions of paradoxical bronchospasm, urticaria, angioedema, hypotension, and collapse. High doses may cause hypokalaemia which are of concern in patients with renal failure and those on certain diuretics and xanthine derivatives.[5]
Society and culture
Diet and bodybuilding use
Salbutamol is taken by some as an alternative to clenbuterol for purposes of fat burning,[6] and/or as a performance enhancer. Abuse of the drug may be confirmed by detection of its presence in plasma or urine, typically in the 10-500 µg/L range.[7]
Doping
Clinical studies show no compelling evidence that salbutamol and other β2-agonists can increase performance in healthy athletes.[8] In spite of this, salbutamol requires "a declaration of Use in accordance with the International Standard for Therapeutic Use Exemptions" under the current WADA prohibited list.[9]
According to two small and limited studies, performed on 8 and 16 subjects, respectively, salbutamol increases the performance even for a person without asthma.[10][11][12]
Detection of use
Salbutamol may be quantified in blood or plasma to confirm a diagnosis of poisoning in hospitalized patients or to aid in a forensic investigation. Urinary salbutamol concentrations are frequently measured in competitive sports programs, for which a level in excess of 1000 μg/L is considered to represent abuse. The window of detection for urine testing is on the order of just 24 hours, given the relatively short elimination half-life of the drug.[7][13][14]
Ban of CFC-containing inhalers
The U.S. Food and Drug Administration (FDA) in April 2005 mandated all (including salbutamol) inhalers containing chlorofluorocarbons (CFCs) were to be prohibited in the United States as of December 31, 2008.[15] CFC inhalers had previously been given "essential use" status, exempting it from a CFC-production ban; however, in accordance with the Montreal Protocol, they will be phased out; in many other countries, patients have been transitioned to non-CFC based inhalers using hydrofluoroalkane (HFA) propellant. Pharmaceutical manufacturers were expected to produce adequate supplies of alternative (HFA) inhalers by 2009.[citation needed]
Due to patent restrictions, HFA salbutamol inhalers cost significantly more per inhaler than existing generic CFC salbutamol inhalers.[16] Generic HFA salbutamol inhalers are not expected to reach the United States market until after 2012 due to existing patents.[16]
Salbutamol is widely used, and accounts for anywhere from 78% of all bronchodilator prescriptions in 2005 to 85% in 2008.[17] However, patients in the United States who cannot tolerate the HFA salbutamol inhalers will not have a single salbutamol alternative available to them domestically after December 31, 2008.[18] The FDA did not approve any alternatives to HFA and there are few standard inhaled lung medications in the United States that come in dry powder inhaler (DPI) versions. Noticeably missing is salbutamol in DPI form in the United States, although it is available in most of the rest of the world in salbutamol DPIs.
Chemistry
Structure-activity relationships
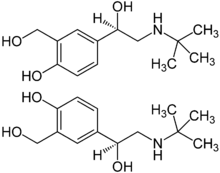
The tertiary butyl group in salbutamol (or albuterol) makes it more selective for β2-receptors. The drug is sold as a racemic mixture mainly because the (S)-enantiomer blocks metabolism pathways while the (R)-enantiomer shows activity.[19]
Synthesis
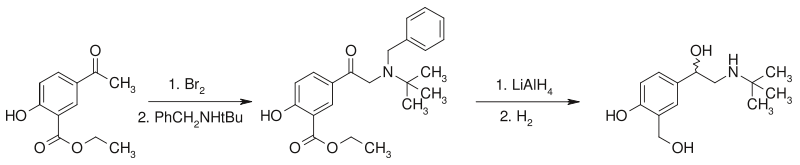
Salbutamol can be prepared from an acetophenone derivative.[20]
Brand names
It is marketed by GlaxoSmithKline as Ventolin, Ventoline, Ventilan, Aerolin or Ventorlin, depending on the market; by Cipla as Asthalin and Asthavent; by Schering-Plough as Proventil, by Teva as ProAir and by Ad-din Pharma as Ventosol
See also
References
- ^ Health Canada
- ^ "Ventolin remains a breath of fresh air for asthma sufferers, after 40 years". The Pharmaceutical Journal 279 (7473): 404-–405. http://www.pharmj.com/pdf/articles/pj_20071013_landmarkdrugs01.pdf.
- ^ "Albuterol". The American Society of Health-System Pharmacists. http://www.drugs.com/monograph/albuterol.html. Retrieved 3 April 2011.
- ^ Rossi, S (2004). Australian Medicines Handbook. AMH. ISBN 0957852142.
- ^ a b c "3.1.1.1 Selective beta2 agonists -- side effects". British National Formulary (57 ed.). London: BMJ Publishing Group Ltd and Royal Pharmaceutical Society Publishing. March 2008. ISBN 0-85369-778-7.
- ^ Carter WJ, Lynch ME (September 1994). "Comparison of the effects of salbutamol and clenbuterol on skeletal muscle mass and carcass composition in senescent rats". Metab. Clin. Exp. 43 (9): 1119–25. doi:10.1016/0026-0495(94)90054-X. PMID 7916118.
- ^ a b Baselt, R. (2008). Disposition of Toxic Drugs and Chemicals in Man (8th ed.). Biomedical Publications,. pp. 33–35. ISBN 0962652369.
- ^ Davis, E; Loiacono, R; Summers, R J (2008). "The rush to adrenaline: drugs in sport acting on the β-adrenergic system". British Journal of Pharmacology 154 (3): 584. doi:10.1038/bjp.2008.164. PMC 2439523. PMID 18500380. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2439523.
- ^ "THE 2010 PROHIBITED LIST INTERNATIONAL STANDARD". WADA. http://www.wada-ama.org/Documents/World_Anti-Doping_Program/WADP-Prohibited-list/WADA_Prohibited_List_2010_EN.pdf. Retrieved 2010-10-20.
- ^ Collomp, K; Candau, R; Lasne, F; Labsy, Z; Préfaut, C; De Ceaurriz, J (2000). "Effects of short-term oral salbutamol administration on exercise endurance and metabolism.". Journal of applied physiology (Bethesda, Md. : 1985) 89 (2): 430–6. PMID 10926623. http://jap.physiology.org/cgi/content/full/89/2/430.
- ^ Salbutamol: Ergogenic effects of salbutamol. http://www.pponline.co.uk/encyc/salbutamol.html. Retrieved 2010-10-20.
- ^ Van Baak, MA; De Hon, OM; Hartgens, F; Kuipers, H (2004). "Inhaled salbutamol and endurance cycling performance in non-asthmatic athletes.". International journal of sports medicine 25 (7): 533–8. doi:10.1055/s-2004-815716. PMID 15459835.
- ^ Berges, Rosa; S; V; F; M; F; M; D (2000). "Discrimination of Prohibited Oral Use of Salbutamol from Authorized Inhaled Asthma Treatment". Clinical Chemistry 46 (9): 1365. PMID 10973867. http://www.clinchem.org/cgi/content/abstract/46/9/1365.
- ^ Schweizer, C; Saugy, M; Kamber, M (2004). "Doping test reveals high concentrations of salbutamol in a Swiss track and field athlete". Clin. J. Sport Med. 14 (5): 312–315. doi:10.1097/00042752-200409000-00018. PMID 15377972. http://journals.lww.com/cjsportsmed/Abstract/2004/09000/Doping_Test_Reveals_High_Concentrations_of.18.aspx.
- ^ Emily Harrison (August 2008). "Unlikely Victims of Banning CFCs—Asthma Sufferers". Scientific American. http://www.sciam.com/article.cfm?id=unlikely-victims-of-banning-cfcs.
- ^ a b "HFA inhalers replacing generic albuterol inhalers, driving up costs". pharmacist.com. http://www.pharmacist.com/AM/Template.cfm?Section=Search1&template=/CM/HTMLDisplay.cfm&ContentID=12192.
- ^ IMS Health Sales & Prescription data for all inhalers sales and prescriptions (July 2008)
- ^ "FDA Advises Patients to Switch to HFA-Propelled Albuterol Inhalers Now CFC-propelled inhalers no longer available as of Dec. 31, 2008". http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2008/ucm116903.htm. Retrieved 2010-10-20.
- ^ Mehta, Akul. "Medicinal Chemistry of the Peripheral Nervous System - Adrenergics and Cholinergic their Biosynthesis, Metabolism and Structure Activity Relationships". http://pharmaxchange.info/notes/medicinal_chemistry/adrenergics_cholinergics.html. Retrieved 2010-10-20.
- ^ Collin, David T.; Hartley, David.; Jack, David.; Lunts, Lawrence H. C.; Press, J. C.; Ritchie, Alexander C.; Toon, Paul. (1970). "Saligenin analogs of sympathomimetic catechol amines". Journal of Medicinal Chemistry 13 (4): 674. doi:10.1021/jm00298a022. PMID 5452431.
Additional notes
- Moore, NG; Pegg, GG; Sillence, MN (September 1994). "Anabolic effects of the beta 2-adrenoceptor agonist salmeterol are dependent on route of administration". Am. J. Physiol. 267 (3 Pt 1): E475–84. PMID 7943228. http://ajpendo.physiology.org/cgi/pmidlookup?view=reprint&pmid=7943228.
- Schiffelers, SL; Saris, WH; Boomsma, F; Van Baak, MA (May 2001). "beta(1)- and beta(2)-Adrenoceptor-mediated thermogenesis and lipid utilization in obese and lean men". J. Clin. Endocrinol. Metab. 86 (5): 2191–9. doi:10.1210/jc.86.5.2191. PMID 11344226. http://jcem.endojournals.org/cgi/pmidlookup?view=long&pmid=11344226.
- Van Baak, MA; Mayer, LH; Kempinski, RE; Hartgens, F (July 2000). "Effect of salbutamol on muscle strength and endurance performance in nonasthmatic men". Med Sci Sports Exerc 32 (7): 1300–6. doi:10.1097/00005768-200007000-00018. PMID 10912897. http://meta.wkhealth.com/pt/pt-core/template-journal/lwwgateway/media/landingpage.htm?issn=0195-9131&volume=32&issue=7&spage=1300.
- Caruso, JF; Hamill, JL; De Garmo, N (February 2005). "Oral albuterol dosing during the latter stages of a resistance exercise program". J Strength Cond Res 19 (1): 102–7. doi:10.1519/R-14793.1. PMID 15705021.
- Caruso JF, Signorile JF, Perry AC, et al. (November 1995). "The effects of albuterol and isokinetic exercise on the quadriceps muscle group". Med Sci Sports Exerc 27 (11): 1471–6. PMID 8587482.
- Martineau, L; Horan, MA; Rothwell, NJ; Little, RA (November 1992). "Salbutamol, a beta 2-adrenoceptor agonist, increases skeletal muscle strength in young men". Clin. Sci. 83 (5): 615–21. PMID 1335400.S
- Desaphy, JF; Pierno, S; De Luca, A; Didonna, P; Camerino, DC (March 2003). "Different ability of clenbuterol and salbutamol to block sodium channels predicts their therapeutic use in muscle excitability disorders". Mol. Pharmacol. 63 (3): 659–70. doi:10.1124/mol.63.3.659. PMID 12606775. http://molpharm.aspetjournals.org/cgi/pmidlookup?view=long&pmid=12606775.
- Maki, KC; Skorodin, MS; Jessen, JH; Laghi, F (June 1996). "Effects of oral albuterol on serum lipids and carbohydrate metabolism in healthy men". Metab. Clin. Exp. 45 (6): 712–7. doi:10.1016/S0026-0495(96)90136-5. PMID 8637445.
External links
- Volmax Drug Information
- Side Effects
- U.S. National Library of Medicine: Drug Information Portal - Albuterol
Adrenergics Receptor ligands Agonists: 5-FNE • 6-FNE • Amidephrine • Anisodamine • Anisodine • Cirazoline • Dipivefrine • Dopamine • Ephedrine • Epinephrine (Adrenaline) • Etilefrine • Ethylnorepinephrine • Indanidine • Levonordefrin • Metaraminol • Methoxamine • Methyldopa • Midodrine • Naphazoline • Norepinephrine (Noradrenaline) • Octopamine • Oxymetazoline • Phenylephrine • Phenylpropanolamine • Pseudoephedrine • Synephrine • Tetrahydrozoline
Antagonists: Abanoquil • Adimolol • Ajmalicine • Alfuzosin • Amosulalol • Arotinolol • Atiprosin • Benoxathian • Buflomedil • Bunazosin • Carvedilol • CI-926 • Corynanthine • Dapiprazole • DL-017 • Domesticine • Doxazosin • Eugenodilol • Fenspiride • GYKI-12,743 • GYKI-16,084 • Indoramin • Ketanserin • L-765,314 • Labetalol • Mephendioxan • Metazosin • Monatepil • Moxisylyte (Thymoxamine) • Naftopidil • Nantenine • Neldazosin • Nicergoline • Niguldipine • Pelanserin • Phendioxan • Phenoxybenzamine • Phentolamine • Piperoxan • Prazosin • Quinazosin • Ritanserin • RS-97,078 • SGB-1,534 • Silodosin • SL-89.0591 • Spiperone • Talipexole • Tamsulosin • Terazosin • Tibalosin • Tiodazosin • Tipentosin • Tolazoline • Trimazosin • Upidosin • Urapidil • Zolertine
* Note that many TCAs, TeCAs, antipsychotics, ergolines, and some piperazines like buspirone, trazodone, nefazodone, etoperidone, and mepiprazole all antagonize α1-adrenergic receptors as well, which contributes to their side effects such as orthostatic hypotension.Agonists: (R)-3-Nitrobiphenyline • 4-NEMD • 6-FNE • Amitraz • Apraclonidine • Brimonidine • Cannabivarin • Clonidine • Detomidine • Dexmedetomidine • Dihydroergotamine • Dipivefrine • Dopamine • Ephedrine • Ergotamine • Epinephrine (Adrenaline) • Esproquin • Etilefrine • Ethylnorepinephrine • Guanabenz • Guanfacine • Guanoxabenz • Levonordefrin • Lofexidine • Medetomidine • Methyldopa • Mivazerol • Naphazoline • Norepinephrine (Noradrenaline) • Phenylpropanolamine • Piperoxan • Pseudoephedrine • Rilmenidine • Romifidine • Talipexole • Tetrahydrozoline • Tizanidine • Tolonidine • Urapidil • Xylazine • Xylometazoline
Antagonists: 1-PP • Adimolol • Aptazapine • Atipamezole • BRL-44408 • Buflomedil • Cirazoline • Efaroxan • Esmirtazapine • Fenmetozole • Fluparoxan • GYKI-12,743 • GYKI-16,084 • Idazoxan • Mianserin • Mirtazapine • MK-912 • NAN-190 • Olanzapine • Phentolamine • Phenoxybenzamine • Piperoxan • Piribedil • Rauwolscine • Rotigotine • SB-269,970 • Setiptiline • Spiroxatrine • Sunepitron • Tolazoline • Yohimbine
* Note that many atypical antipsychotics and azapirones like buspirone and gepirone (via metabolite 1-PP) antagonize α2-adrenergic receptors as well.βAgonists: 2-FNE • 5-FNE • Amibegron • Arbutamine • Arformoterol • Arotinolol • BAAM • Bambuterol • Befunolol • Bitolterol • Broxaterol • Buphenine • Carbuterol • Cimaterol • Clenbuterol • Denopamine • Deterenol • Dipivefrine • Dobutamine • Dopamine • Dopexamine • Ephedrine • Epinephrine (Adrenaline) • Etafedrine • Etilefrine • Ethylnorepinephrine • Fenoterol • Formoterol • Hexoprenaline • Higenamine • Indacaterol • Isoetarine • Isoprenaline (Isoproterenol) • Isoxsuprine • Labetalol • Levonordefrin • Levosalbutamol • Mabuterol • Methoxyphenamine • Methyldopa • Norepinephrine (Noradrenaline) • Orciprenaline • Oxyfedrine • Phenylpropanolamine • Pirbuterol • Prenalterol • Ractopamine • Procaterol • Pseudoephedrine • Reproterol • Rimiterol • Ritodrine • Salbutamol (Albuterol) • Salmeterol • Solabegron • Terbutaline • Tretoquinol • Tulobuterol • Xamoterol • Zilpaterol • Zinterol
Antagonists: Acebutolol • Adaprolol • Adimolol • Afurolol • Alprenolol • Alprenoxime • Amosulalol • Ancarolol • Arnolol • Arotinolol • Atenolol • Befunolol • Betaxolol • Bevantolol • Bisoprolol • Bopindolol • Bormetolol • Bornaprolol • Brefonalol • Bucindolol • Bucumolol • Bufetolol • Buftiralol • Bufuralol • Bunitrolol • Bunolol • Bupranolol • Burocrolol • Butaxamine • Butidrine • Butofilolol • Capsinolol • Carazolol • Carpindolol • Carteolol • Carvedilol • Celiprolol • Cetamolol • Cicloprolol • Cinamolol • Cloranolol • Cyanopindolol • Dalbraminol • Dexpropranolol • Diacetolol • Dichloroisoprenaline • Dihydroalprenolol • Dilevalol • Diprafenone • Draquinolol • Dropranolol • Ecastolol • Epanolol • Ericolol • Ersentilide • Esatenolol • Esmolol • Esprolol • Eugenodilol • Exaprolol • Falintolol • Flestolol • Flusoxolol • Hydroxycarteolol • Hydroxytertatolol • ICI-118,551 • Idropranolol • Indenolol • Indopanolol • Iodocyanopindolol • Iprocrolol • Isoxaprolol • Isamoltane • Labetalol • Landiolol • Levobetaxolol • Levobunolol • Levocicloprolol • Levomoprolol • Medroxalol • Mepindolol • Metalol • Metipranolol • Metoprolol • Moprolol • Nadolol • Nadoxolol • Nafetolol • Nebivolol • Neraminol • Nifenalol • Nipradilol • Oberadilol • Oxprenolol • Pacrinolol • Pafenolol • Pamatolol • Pargolol • Parodilol • Penbutolol • Penirolol • PhQA-33 • Pindolol • Pirepolol • Practolol • Primidolol • Procinolol • Pronethalol • Propafenone • Propranolol • Ridazolol • Ronactolol • Soquinolol • Sotalol • Spirendolol • SR 59230A • Sulfinalol • TA-2005 • Talinolol • Tazolol • Teoprolol • Tertatolol • Terthianolol • Tienoxolol • Tilisolol • Timolol • Tiprenolol • Tolamolol • Toliprolol • Tribendilol • Trigevolol • Xibenolol • XipranololReuptake inhibitors Selective norepinephrine reuptake inhibitors: Amedalin • Atomoxetine (Tomoxetine) • Ciclazindol • Daledalin • Esreboxetine • Lortalamine • Mazindol • Nisoxetine • Reboxetine • Talopram • Talsupram • Tandamine • Viloxazine; Norepinephrine-dopamine reuptake inhibitors: Amineptine • Bupropion (Amfebutamone) • Fencamine • Fencamfamine • Lefetamine • Levophacetoperane • LR-5182 • Manifaxine • Methylphenidate • Nomifensine • O-2172 • Radafaxine; Serotonin-norepinephrine reuptake inhibitors: Bicifadine • Desvenlafaxine • Duloxetine • Eclanamine • Levomilnacipran • Milnacipran • Sibutramine • Venlafaxine; Serotonin-norepinephrine-dopamine reuptake inhibitors: Brasofensine • Diclofensine • DOV-102,677 • DOV-21,947 • DOV-216,303 • JNJ-7925476 • JZ-IV-10 • Methylnaphthidate • Naphyrone • NS-2359 • PRC200-SS • SEP-225,289 • SEP-227,162 • Tesofensine; Tricyclic antidepressants: Amitriptyline • Butriptyline • Cianopramine • Clomipramine • Desipramine • Dosulepin • Doxepin • Imipramine • Lofepramine • melitracen • Nortriptyline • Protriptyline • Trimipramine; Tetracyclic antidepressants: Amoxapine • Maprotiline • Mianserin • Oxaprotiline • Setiptiline; Others: Cocaine • CP-39,332 • EXP-561 • Fezolamine • Ginkgo biloba • Indeloxazine • Nefazodone • Nefopam • Pridefrine • Tapentadol • Teniloxazine • Tramadol • ZiprasidoneEnzyme inhibitors 3,4-DihydroxystyreneDBHCGS-19281A • SKF-64139 • SKF-7698Nonselective: Benmoxin • Caroxazone • Echinopsidine • Furazolidone • Hydralazine • Indantadol • Iproclozide • Iproniazid • Isocarboxazid • Isoniazid • Linezolid • Mebanazine • Metfendrazine • Nialamide • Octamoxin • Paraxazone • Phenelzine • Pheniprazine • Phenoxypropazine • Pivalylbenzhydrazine • Procarbazine • Safrazine • Tranylcypromine; MAO-A selective: Amiflamine • Bazinaprine • Befloxatone • Befol • Brofaromine • Cimoxatone • Clorgiline • Esuprone • Harmala alkaloids (Harmine, Harmaline, Tetrahydroharmine, Harman, Norharman, etc) • Methylene Blue • Metralindole • Minaprine • Moclobemide • Pirlindole • Sercloremine • Tetrindole • Toloxatone • Tyrima; MAO-B selective: D-Deprenyl • Selegiline (L-Deprenyl) • Ladostigil • Lazabemide • Milacemide • Mofegiline • Pargyline • Rasagiline • Safinamide
* Note that MAO-B inhibitors also influence norepinephrine/epinephrine levels since they inhibit the breakdown of their precursor dopamine.COMTOthers Ferrous Iron (Fe2+) • S-Adenosyl-L-Methionine • Vitamin B3 (Niacin, Nicotinamide → NADPH) • Vitamin B6 (Pyridoxine, Pyridoxamine, Pyridoxal → Pyridoxal Phosphate) • Vitamin B9 (Folic acid → Tetrahydrofolic acid) • Vitamin C (Ascorbic acid) • Zinc (Zn2+)OthersActivity enhancers: BPAP • PPAP; Release blockers: Bethanidine • Bretylium • Guanadrel • Guanazodine • Guanclofine • Guanethidine • Guanoxan; Toxins: Oxidopamine (6-Hydroxydopamine)List of adrenergic drugsDrugs for obstructive airway diseases: asthma/COPD (R03) Adrenergics, inhalants Salbutamol#/Levosalbutamol • Fenoterol • Terbutaline • Pirbuterol • Procaterol • Bitolterol • Rimiterol • Carbuterol • Tulobuterol • ReproterolLong acting β2-agonists (LABA)otherGlucocorticoids Anticholinergics/
muscarinic antagonistMast cell stabilizers Cromoglicate • NedocromilXanthines Eicosanoid inhibition Thromboxane receptor antagonistsCombination products Phenethylamines Phenethylamines Psychedelics: 2C-B • 2C-B-FLY • 2C-C • 2C-D • 2C-E • 2C-F • 2C-G • 2C-I • 2C-N • 2C-P • 2C-SE • 2C-T • 2C-T-2 • 2C-T-4 • 2C-T-7 • 2C-T-8 • 2C-T-9 • 2C-T-13 • 2C-T-15 • 2C-T-17 • 2C-T-21 • 2C-TFM • 2C-YN • Allylescaline • DESOXY • Escaline • Isoproscaline • Jimscaline • Macromerine • MEPEA • Mescaline • Metaescaline • Methallylescaline • Proscaline • Psi-2C-T-4 • TCB-2
Stimulants: 2-OH-PEA • β-Me-PEA • Hordenine • N-Me-PEA • Phenethylamine (PEA)
Entactogens: Lophophine • MDPEA • MDMPEA
Others: BOH • DMPEAAmphetamines
PhenylisopropylaminesPsychedelics: 3C-BZ • 3C-E • 3C-P • Aleph • Beatrice • Bromo-DragonFLY • D-Deprenyl • DMA • DMCPA • DMMDA • DOB • DOC • DOEF • DOET • DOI • DOM • DON • DOPR • DOTFM • Ganesha • MMDA • MMDA-2 • Psi-DOM • TMA • TeMA
Stimulants: 4-MA • 4-MMA • 4-MTA • 5-IT • Alfetamine • Amfecloral • Amfepentorex • Amphetamine (Dextroamphetamine, Levoamphetamine) • Amphetaminil • Benfluorex • Benzphetamine • Cathine • Clobenzorex • Dimethylamphetamine • Ephedrine (EPH) • Ethylamphetamine • Fencamfamine • Fencamine • Fenethylline • Fenfluramine (Dexfenfluramine) • Fenproporex • Fludorex • Furfenorex • Isopropylamphetamine • Lefetamine • Mefenorex • Methamphetamine (Dextromethamphetamine, Levomethamphetamine) • Methoxyphenamine • MMA • Norfenfluramine • Oxilofrine • Ortetamine • PBA • PCA • Phenpromethamine • PFA • PFMA • PIA • PMA • PMEA • PMMA • Phenylpropanolamine (PPA) • Prenylamine • Propylamphetamine • Pseudoephedrine (PSE) • Sibutramine • Tiflorex (Flutiorex) • Tranylcypromine • Xylopropamine • Zylofuramine
Entactogens: 5-APDB • 6-APB • 6-APDB • EDA • IAP • MDA • MDEA • MDHMA (FLEA) • MDMA ("Ecstasy") • MDOH • MMDMA • NAP • TAP
Others: Amiflamine • DFMDA • D-Deprenyl • L-Deprenyl (Selegiline)Phentermines Stimulants: Chlorphentermine • Cloforex • Clortermine • Etolorex • Mephentermine • Pentorex (Phenpentermine) • Phentermine
Entactogens: MDPH • MDMPHCathinones Stimulants: Amfepramone • Brephedrone • Buphedrone • Bupropion (Amfebutamone) • Cathinone (Propion) • Dimethylcathinone (Dimethylpropion, Metamfepramone) • Ethcathinone (Ethylpropion) • Flephedrone • Methcathinone (Methylpropion) • Mephedrone • Methedrone
Entactogens: Ethylone • MethylonePhenylisobutylamines Phenylalkylpyrrolidines Stimulants: α-PBP • α-PPP • α-PVP • MDPBP • MDPPP • MDPV • MOPPP • MPBP • MPHP • MPPP • Naphyrone • PEP • Prolintane • PyrovaleroneCatecholamines
(and relatives..)6-FNE • 6-OHDA • α-Me-DA • α-Me-TRA • Adrenochrome • Ciladopa • D-DOPA (Dextrodopa) • Dopamine • Epinephrine (Adrenaline) • Epinine • Fenclonine • Ibopamine • L-DOPA (Levodopa) • L-DOPS (Droxidopa) • L-Phenylalanine • L-Tyrosine • meta-Octopamine • meta-Tyramine • Metanephrine • Metirosine • Methyldopa • Nordefrin (Levonordefrin) • Norepinephrine (Noradrenaline) • Normetanephrine • para-Octopamine • para-TyramineMiscellaneous Amidephrine • Arbutamine • Cafedrine • Denopamine • Dobutamine • Dopexamine • Etafedrine • Ethylnorepinephrine • Etilefrine • Famprofazone • Gepefrine • Isoprenaline (Isoproterenol) • Isoetarine • Metaraminol • Metaterol • Methoxamine • Norfenefrine • Orciprenaline • Phenylephrine (Neosynephrine) • Phenoxybenzamine • Prenalterol • Pronethalol • Propranolol • Salbutamol (Albuterol; Levosalbutamol) • Synephrine (Oxedrine) • Theodrenaline • Xamoterol
Wikimedia Foundation. 2010.