- Tyramine
-
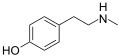
Tyramine 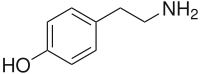
 4-(2-aminoethyl)phenol
4-(2-aminoethyl)phenolIdentifiers CAS number 51-67-2 
PubChem 5610 ChemSpider 5408 
UNII X8ZC7V0OX3 
KEGG C00483 
MeSH Tyramine ChEBI CHEBI:15760 
ChEMBL CHEMBL11608 
Jmol-3D images Image 1 - Oc1ccc(cc1)CCN
Properties Molecular formula C8H11NO Molar mass 137.179 g/mol[1]  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Tyramine (4-Hydroxyphenethylamine; para-tyramine, Mydrial or Uteramin) is a naturally occurring monoamine compound and trace amine derived from the amino acid tyrosine.[1] Tyramine acts as a catecholamine (dopamine, norepinephrine (noradrenaline), epinephrine (adrenaline)) releasing agent. Notably, however, it is unable to cross the blood-brain-barrier (BBB), resulting in only non-psychoactive peripheral sympathomimetic effects. When tyramine-rich foods are ingested in conjunction with a monoamine oxidase inhibitor (MAOI), tyramine is responsible for the so-called "cheese effect" sometimes seen with their use.
Contents
Occurrence
Tyramine occurs widely in plants and animals, and is metabolized by the enzyme monoamine oxidase. In foods, it is often produced by the decarboxylation of tyrosine during fermentation or decay. Foods containing considerable amounts of tyramine include meats that are potentially spoiled or pickled, aged, smoked, fermented, or marinated (some fish, poultry, and beef); most pork (except cured ham); chocolate; alcoholic beverages; and fermented foods, such as most cheeses (except ricotta, cottage cheese, cream cheese and neufchatel cheese), sour cream, yogurt, shrimp paste, soy sauce, soy bean condiments, teriyaki sauce, tofu, tempeh, miso soup, sauerkraut, broad (fava) beans, green bean pods, Italian flat (Romano) beans, snow peas, avocados, bananas, pineapple, eggplants, figs, red plums, raspberries, peanuts, Brazil nuts, coconuts, processed meat, yeast, and an array of cacti.
Physical effects and pharmacology
Tyramine is physiologically metabolized by MAOA. In humans, if monoamine metabolism is compromised by the use of monoamine oxidase inhibitors (MAOIs) and foods high in tyramine are ingested, a hypertensive crisis can result, as tyramine can cause the release of stored monoamines, such as dopamine, norepinephrine and epinephrine. The first signs of this were discovered by a neurologist who noticed his wife, who at the time was on MAOI medication, had severe headaches when eating cheese. For this reason, the crisis is still called the "cheese effect", though other foods can cause the same problem.[citation needed]. Most processed cheeses do not contain high enough tyramine to cause hypertensive effects, although some aged cheeses (such as Stilton cheese) do. [2]
A large dietary intake of tyramine (or a dietary intake of tyramine while taking MAO inhibitors) can cause the tyramine pressor response, which is defined as an increase in systolic blood pressure of 30 mmHg or more. The displacement of norepinephrine (noradrenaline) from neuronal storage vesicles by acute tyramine ingestion is thought to cause the vasoconstriction and increased heart rate and blood pressure of the pressor response. In severe cases, adrenergic crisis can occur.
However, if one has had repeated exposure to tyramine, there is a decreased pressor response; tyramine is degraded to octopamine, which is subsequently packaged in synaptic vesicles with norepinephrine (noradrenaline). Therefore, after repeated tyramine exposure, these vesicles contain an increased amount of octopamine and a relatively reduced amount of norepinephrine. When these vesicles are secreted upon tyramine ingestion, there is a decreased pressor response, as less norepinephrine is secreted into the synapse, and octopamine does not activate alpha or beta adrenergic receptors.
When using a MAO inhibitor (MAOI), the intake of approximately 10 to 25 mg of tyramine is required for a severe reaction compared to 6 to 10 mg for a mild reaction.
The possibility that tyramine acts directly as a neurotransmitter was revealed by the discovery of a G protein-coupled receptor with high affinity for tyramine, called TA1. The TA1 receptor is found in the brain as well as peripheral tissues, including the kidney. The existence of a receptor with high affinity for tyramine supports the hypothesis that tyramine may also act directly to affect blood pressure regulation.
Dietary tyramine intake has also been associated with migraine in select populations, leading many sufferers to restrict foods high in tyramine.[3] Reports on the tyramine-migraine link have been both affirmed and denied. A 2007 review published in Neurological Sciences[4] presented data showing migraine and cluster headaches are characterised by an increase of circulating neurotransmitters and neuromodulators (including tyramine, octopamine and synephrine) in the hypothalamus, amygdala and dopaminergic system.
Biosynthesis
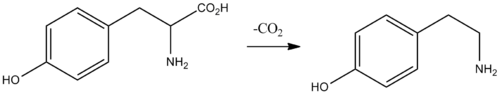
Biochemically, tyramine is produced by the decarboxylation of tyrosine via the action of the enzyme tyrosine decarboxylase.[5] Tyramine can in turn be converted to methylated alkaloid derivatives N-methyltyramine, N,N-dimethyltyramine (hordenine), and
Tyramine