- Melatonin
-
Melatonin 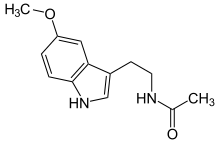
Systematic (IUPAC) name N-[2-(5-methoxy-1H-indol-3-yl)ethyl]
ethanamideClinical data AHFS/Drugs.com Consumer Drug Information Pregnancy cat. ? Legal status Prescription Only (S4) (AU) ? (UK) OTC (US) Routes In humans: orally, as capsules, tablets or liquid, sublingually, or as transdermal patches. In lab animals: also injection. Pharmacokinetic data Bioavailability 30 – 50% Metabolism Hepatic via CYP1A2 mediated 6-hydroxylation Half-life 35 to 50 minutes Excretion Urine Identifiers CAS number 73-31-4 
ATC code N05CH01 PubChem CID 896 IUPHAR ligand 224 DrugBank DB01065 ChemSpider 872 
UNII JL5DK93RCL 
KEGG D08170 
ChEBI CHEBI:16796 
ChEMBL CHEMBL45 
Chemical data Formula C13H16N2O2 Mol. mass 232.278 g/mol SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Melatonin
 i/ˌmɛləˈtoʊnɪn/, also known chemically as N-acetyl-5-methoxytryptamine,[1] is a naturally occurring compound found in animals, plants, and microbes.[2][3] In animals, circulating levels of the hormone melatonin vary in a daily cycle, thereby allowing the entrainment of the circadian rhythms of several biological functions.[4]
i/ˌmɛləˈtoʊnɪn/, also known chemically as N-acetyl-5-methoxytryptamine,[1] is a naturally occurring compound found in animals, plants, and microbes.[2][3] In animals, circulating levels of the hormone melatonin vary in a daily cycle, thereby allowing the entrainment of the circadian rhythms of several biological functions.[4]Many biological effects of melatonin are produced through activation of melatonin receptors,[5] while others are due to its role as a pervasive and powerful antioxidant,[6] with a particular role in the protection of nuclear and mitochondrial DNA.[7]
In mammals, melatonin is secreted into the blood by the pineal gland in the brain. Known as the "hormone of darkness," it is secreted in darkness in both day-active (diurnal) and night-active (nocturnal) animals.[8]
It may also be produced by a variety of peripheral cells such as bone marrow cells,[9][10] lymphocytes, and epithelial cells. Usually, the melatonin concentration in these cells is much higher than that found in the blood, but it does not seem to be regulated by the photoperiod.
Research has shown that when bird chicks ingest melatonin-rich plant feed, such as rice, the melatonin binds to melatonin receptors in their brains. [11] No food has been found to elevate plasma melatonin levels in humans.[12]
Products containing melatonin have been available over-the-counter as dietary supplements in the United States since the mid-1990s.[13] In many other countries, the over-the-counter sale of this neurohormone is not permitted or requires a prescription, and the U.S. Postal Service lists unapproved melatonin preparations among items prohibited by Germany.[14]
Contents
Medical uses
Melatonin has been studied for the treatment of cancer, immune disorders, cardiovascular diseases, depression, seasonal affective disorder (SAD), circadian rhythm sleep disorders and sexual dysfunction. It may ameliorate circadian misalignment and SAD.[15] Basic research indicates that melatonin may play a significant role in modulating the effects of drugs of abuse such as cocaine.[16] Melatonin is also a geroprotector.
Circadian rhythm disorders
Exogenous melatonin taken in the evening is, together with light therapy upon awakening, the standard treatment for delayed sleep phase syndrome (DSPS) and non-24-hour sleep-wake syndrome. It appears to have some use against other circadian rhythm sleep disorders as well, such as jet lag and the problems of people who work rotating or night shifts. Melatonin reduces sleep onset latency to a greater extent in people with DSPS than in people with insomnia.[17]
Taken 30 to 90 minutes before bedtime, melatonin supplementation acts as a mild hypnotic. It causes melatonin levels in the blood to rise earlier than the brain's own production accomplishes. This usage is now common in sleep and relaxation drinks.[18]
A very small dose taken several hours before bedtime in accordance with the phase response curve for melatonin in humans (PRC) doesn't cause sleepiness but, acting as a chronobiotic (affecting aspects of biological time structure),[19] advances the phase slightly and is additive to the effect of using light therapy upon awakening. Light therapy may advance the phase about one to two-and-a-half hours and a small oral dose of melatonin, timed correctly some hours before bedtime, can add about 30 minutes to the advance achieved with light therapy.[20]
The World Health Organization in 2007 named late night shift work as a probable cancer-causing agent.[21] Melatonin is an anti-oxidant and suppressant of tumor development that is produced at night; when someone works in artificial light, they generally have lower melatonin and may be more likely to develop cancer. Melatonin supplements may simulate the melatonin production at different times that does not occur during regular sleeping hours for people who work night shifts.[22]
Learning, memory and Alzheimer's
Melatonin receptors appear to be important in mechanisms of learning and memory in mice,[23] and melatonin can alter electrophysiological processes associated with memory, such as long-term potentiation (LTP). The first published evidence that melatonin may be useful in Alzheimer's disease was the demonstration that this neurohormone prevents neuronal death caused by exposure to the amyloid beta protein, a neurotoxic substance that accumulates in the brains of patients with the disorder.[24] Melatonin also inhibits the aggregation of the amyloid beta protein into neurotoxic microaggregates that, it seems, underlie the neurotoxicity of this protein, causing death of neurons and formation of neurofibrillary tangles, the other neuropathological landmark of Alzheimer's disease.[25]
Melatonin has been shown to prevent the hyperphosphorylation of the tau protein in rats. Hyperphosphorylation of tau protein can also result in the formation of neurofibrillary tangles. Studies in rats suggest that melatonin may be effective for treating Alzheimer's disease.[26] These same neurofibrillary tangles can be found in the hypothalamus in patients with Alzheimer's, adversely affecting their bodies' production of melatonin. Those Alzheimer's patients with this specific affliction often show heightened afternoon agitation, called sundowning, which has been shown in many studies to be effectively treated with melatonin supplements in the evening.[27]
Delirium
A randomized placebo-controlled trial showed that low-dose (0.5 mg) melatonin supplementation to elderly patients admitted to acute Medicine services significantly reduced delirium.[28]
ADHD
Research shows that after melatonin is administered to ADHD patients on methylphenidate, the time needed to fall asleep is significantly reduced. Furthermore, the effects of the melatonin after three months showed no change from its effects after one week of use.[29]
Fertility
A research team in Italy has found that melatonin supplementation in the evening in perimenopausal women produces an improvement in thyroid function and gonadotropin levels, as well as restoring fertility and menstruation and preventing the depression associated with the menopause.[30] However, at the same time, some resources warn women trying to conceive not to take a melatonin supplement.[31] One study reported that three mg of melatonin taken in the evening raised prolactin levels in six out of seven women.[32] Melatonin also lowers FSH levels. It is believed that these hormonal changes could in some women impair fertility.[33]
Toxicology
Melatonin has a very low toxicity in rats. Rat maternal toxicity: The no observable adverse effect level (NOAEL) and lowest observed adverse effect level (LOAEL) were 100 and 200 mg/kg/day, respectively, and the developmental toxicity NOAEL was ≥ 200 mg/kg/day.[34]
Headaches
Several clinical studies indicate that supplementation with melatonin is an effective preventive treatment for migraines and cluster headaches.[35][36]
Mood disorders
Melatonin has been shown to be effective in treating one form of depression and seasonal affective disorder,[37] and is being considered for bipolar and other disorders in which circadian disturbances are involved.[38] It has been observed that bipolar disorder might have, as a "trait marker" (something that is characteristic of being bipolar, that does not change with state), supersensitivity to light, i.e., a greater decrease in melatonin secretion in response to light exposure at night.[39] This could be contrasted with drug-free recovered bipolar patients not showing light hypersensitivity.[40]
Cancer
A systematic review of unblinded clinical trials involving a total of 643 cancer patients using melatonin found a reduced incidence of death.[41] Another clinical trial is due to be completed in 2012.[42] Melatonin levels at night are reduced to 50% by exposure to a low-level incandescent bulb for only 39 minutes, and it has been suspected that women with the brightest bedrooms have an increased risk for breast cancer.[43] Reduced melatonin production has been proposed as a likely factor in the significantly higher cancer rates in night workers.[44]
Gallbladder stones
Melatonin presence in the gallbladder has many protective properties, such as converting cholesterol to bile, preventing oxidative stress, and increasing the mobility of gallstones from the gallbladder.[45] It also decreases the amount of cholesterol produced in the gallbladder by regulating the cholesterol that passes through the intestinal wall. In guinea pigs, melatonin administration restored normal function by reducing inflammation after induced Cholecystitis, whether administered before or after onset of inflammation.[45] Concentration of melatonin in the bile is 2–3 times higher than the otherwise very low daytime melatonin levels in the blood across many diurnal mammals, including humans.[46]
Amyotrophic lateral sclerosis
In animal models, melatonin has been shown to ameliorate glutamate-induced neuronal death, it is presumed due to its antioxidant effects. In a clinical safety study involving 31 ALS patients, high-dose rectal melatonin (300 mg/day for 2 years) was shown to be tolerated well.[47]
Obesity
Melatonin is involved in energy metabolism and body weight control in small animals. Many studies show that chronic melatonin supplementation in drinking water reduces body weight and abdominal fat in experimental animals, especially in the middle-aged rats.[48] It is interesting to note that the weight loss effect of melatonin does not require the animals to eat less and to be physically more active. A potential mechanism is that melatonin promotes the recruitment of brown adipose tissue (BAT) as well as enhances its activity.[49] This effect would raise the basal metabolic rate by stimulating thermogenesis, heat generation through uncoupling oxidative phosphorylation in mitochondria. Whether the results of animal studies can be extrapolated to human obesity is a matter of future clinical trials, since substantially active BAT has been identified in adult humans.[citation needed]
Protection from radiation
Both animal [50] and human[51][52] studies have shown melatonin to be potentially radioprotective. Moreover, it is a more efficient protector than amifostine,[53] a commonly used agent for this purpose. The mechanism of melatonin in protection against ionizing radiation is thought to involve scavenging of free radicals.[54] It is estimated that nearly 70% of biological damage caused by ionizing radiation is attributable to the free radical, especially the hydroxyl radical that attacks DNA, proteins, and cellular membranes. Melatonin has been suggested as a radioprotective agent, with the proposed advantages of being broadly protective, readily available, orally self-administered, and without major known side effects.[55]
Other
Melatonin increases proliferation of cultured neural stem cells obtained from mice nervous tissue.[56]
Melatonin was used to treat Periodic limb movement disorder, a common neurological condition, which, when severe, adversely affects sleep and causes excessive daytime fatigue, in a small trial conducted by Kunz D and Bes F. In this condition, the sufferer is affected by mini arousals during sleep and limb movements that occur in a frequent rhythmic fashion. This often involves leg kicking, but sometimes also involves arm movement. Those affected are often not aware of the condition, and partners are often the first to notice the condition. 7 out of the 9 participants in the trial showed significant improvement.[57]
In recent trial for use in IBS treatment, melatonin relieved some symptoms, as published in 2010[58]
Adverse effects
Melatonin appears to cause very few side-effects in the short term, up to three months, when healthy people take it at low doses. A systematic review[59] in 2006 looked specifically at efficacy and safety in two categories of melatonin usage: first, for sleep disturbances that are secondary to other diagnoses and, second, for sleep disorders such as jet lag and shift work that accompany sleep restriction.[59]
The study concluded that There is no evidence that melatonin is effective in treating secondary sleep disorders or sleep disorders accompanying sleep restriction, such as jet lag and shiftwork disorder. There is evidence that melatonin is safe with short term use.[59]
A similar analysis[60] by the same team a year earlier on the efficacy and safety of exogenous melatonin in the management of primary sleep disorders found that: There is evidence to suggest that melatonin is safe with short-term use (3 months or less).
Some unwanted effects in some people, especially at high doses (~3 mg/day or more) may include: headaches, nausea, next-day grogginess or irritability, hormone fluctuations, vivid dreams or nightmares,[61] reduced blood flow, and hypothermia.[62]
While no large, long-term studies that might reveal side-effects have been conducted, there do exist case reports about patients having taken melatonin for months.[63]
Melatonin can cause somnolence (drowsiness), and, therefore, caution should be shown when driving, operating machinery, etc.
In individuals with auto-immune disorders, there is conflicting evidence that melatonin supplementation may either ameliorate or exacerbate symptoms due to immunomodulation.[64][65]
Individuals experiencing orthostatic intolerance, a cardiovascular condition that results in reduced blood pressure and blood flow to the brain when a person stands, may experience a worsening of symptoms when taking melatonin supplements, a study at Penn State College of Medicine's Milton S. Hershey Medical Center suggests. Melatonin can exacerbate symptoms by reducing nerve activity in those experiencing the condition, the study found.[66]
The use of melatonin derived from animal pineal tissue may carry the risk of contamination or the means of transmitting viral material. The synthetic form of this medication does not carry this risk.[4][67]
In plants
Melatonin has been identified in many plants including Feverfew (Tanacetum parthenium), and St John's wort (Hypericum perforatum).[3] It occurs in trace amounts in some foods, especially cherries to about 0.17-13.46 ng/g.[68] The physiological roles of melatonin in plants involve regulation of their response to photoperiod, defense against harsh environments, and the function of an antioxidant. The latter may be the original function of melatonin in organisms with the others being added during evolution.[69] Melatonin has been reported in foodstuffs including bananas and grapes, rice and cereals, herbs, olive oil, wine and beer. While no food has been found to elevate plasma melatonin levels in humans,[12] when other animals consume melatonin-containing food, blood levels of melatonin do increase.[11]
In animals
Many animals use the variation in duration of melatonin production each day as a seasonal clock.[70] In animals including humans[71] the profile of melatonin synthesis and secretion is affected by the variable duration of night in summer as compared to winter. The change in duration of secretion thus serves as a biological signal for the organisation of daylength-dependent (photoperiodic) seasonal functions such as reproduction, behaviour, coat growth and camouflage colouring in seasonal animals.[71] In seasonal breeders that do not have long gestation periods and that mate during longer daylight hours, the melatonin signal controls the seasonal variation in their sexual physiology, and similar physiological effects can be induced by exogenous melatonin in animals including mynah birds[72] and hamsters.[73]
In mammals
Melatonin is produced in the pineal gland, which is outside of the blood-brain barrier, acts as an endocrine hormone since it is released into the blood.[74] By contrast, melatonin produced by the retina and the gastrointestinal (GI) tract acts as a paracrine hormone.[citation needed]
Melatonin can suppress libido by inhibiting secretion of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) from the anterior pituitary gland, especially in mammals that have a breeding season when daylight hours are long. The reproduction of long-day breeders is repressed by melatonin and the reproduction of short-day breeders is stimulated by melatonin. During the night, melatonin regulates leptin, lowering the levels; see leptin.
Light/dark information reaches the suprachiasmatic nuclei (SCN) via retinal photosensitive ganglion cells, intrinsically photosensitive photoreceptor cells, distinct from those involved in image forming (that is, these light-sensitive cells are a third type in the retina, in addition to rods and cones). These cells represent approximately 2% of the retinal ganglion cells in humans and express the photopigment melanopsin.[75] The sensitivity of melanopsin is consistent with that of a vitamin A-based photopigment, with a peak sensitivity at 484 nm (blue light).[76] This photoperiod cue entrains the circadian rhythm, and the resultant production of specific "dark"- and "light"-induced neural and endocrine signals that regulate behavioral and physiological circadian rhythms. Melatonin is secreted in darkness in both day-active (diurnal) and night-active (nocturnal) animals.[8]
In humans
Circadian rhythm
In humans, melatonin is produced by the pineal gland, located in the center of the brain but outside the blood-brain barrier. The melatonin signal forms part of the system that regulates the sleep-wake cycle by chemically causing drowsiness and lowering the body temperature, but it is the central nervous system (to be more specific, the SCN) that controls the daily cycle in most components of the paracrine and endocrine systems[77][78] rather than the melatonin signal (as was once postulated).
Infants' melatonin levels become regular in about the third month after birth, with the highest levels measured between midnight and 08:00 (8 AM).[79]
In humans, 90% of melatonin is cleared in a single passage through the liver, a small amount is excreted in urine,[17] and a small amount is found in saliva.
Human melatonin production decreases as a person ages.[80] It is believed that as children become teenagers, the nightly schedule of melatonin release is delayed, leading to later sleeping and waking times.[81]
Light dependence
Production of melatonin by the pineal gland is inhibited by light and permitted by darkness. For this reason melatonin has been called "the hormone of darkness." Its onset each evening is called the Dim-Light Melatonin Onset (DLMO). Secretion of melatonin as well as its level in the blood, peaks in the middle of the night, and gradually falls during the second half of the night, with normal variations in timing according to an individual's chronotype.[citation needed]
Terman et al. devised a formulation that mimics that gradual washout (vs. the spikes in blood concentration and rapid washout associated with most over-the-counter melatonin tablets). When used several hours before sleep, the compound shifts the circadian clock earlier, thus promoting earlier sleep onset and morning awakening.[82]
It is principally blue light, around 460 to 480nm, that suppresses melatonin,[83] increasingly with increased light intensity and length of exposure. Until recent history, humans in temperate climates were exposed to few hours of (blue) daylight in the winter; their fires gave predominantly yellow light. Wearing glasses that block blue light in the hours before bedtime may avoid melatonin loss. Kayumov et al. showed that light containing only wavelengths greater than 530 nm does not suppress melatonin in bright-light conditions.[84] Use of blue-blocking goggles the last hours before bedtime has also been advised for people who need to adjust to an earlier bedtime, as melatonin promotes sleepiness.
Antioxidant
Besides its function as synchronizer of the biological clock, melatonin also exerts a powerful antioxidant activity. The discovery of melatonin as an antioxidant was made in 1993.[85] In many less complex life forms, this is its only known function.[86] Melatonin is an antioxidant that can easily cross cell membranes and the blood-brain barrier.[6][87] Melatonin is a direct scavenger of OH, O2−, and NO.[88] Unlike other antioxidants, melatonin does not undergo redox cycling, the ability of a molecule to undergo reduction and oxidation repeatedly. Redox cycling may allow other antioxidants (such as vitamin C) to act as pro-oxidants, counterintuitively promoting free radical formation. Melatonin, on the other hand, once oxidized, cannot be reduced to its former state because it forms several stable end-products upon reacting with free radicals. Therefore, it has been referred to as a terminal (or suicidal) antioxidant.[89]
Recent research indicates that the first metabolite of melatonin in the melatonin antioxidant pathway may be N(1)-acetyl-N(2)-formyl-5-methoxykynuramine (or AFMK) rather than the common, excreted 6-hydroxymelatonin sulfate. AFMK alone is detectable in unicellular organisms and metazoans. A single AFMK molecule can neutralize up to 10 ROS/RNS (reactive oxygen species/reactive nitrogen species) since many of the products of the reaction/derivatives (including melatonin) are themselves antioxidants. This capacity to absorb free radicals extends at least to the quaternary metabolites of melatonin, a process referred to as "the free radical scavenging cascade." This is not true of other, conventional antioxidants.[86]
In animal models, melatonin has been demonstrated to prevent the damage to DNA by some carcinogens, stopping the mechanism by which they cause cancer.[90]
It also has been found to be effective in protecting against brain injury caused by ROS release in experimental hypoxic brain damage in newborn rats.[91] Melatonin's antioxidant activity may reduce damage caused by some types of Parkinson's disease, play a role in preventing cardiac arrhythmia and possibly increase longevity; it has been shown to increase the average life span of mice by 20% in some studies as well.[92][93][94]
Immune system
While it is known that melatonin interacts with the immune system,[95][96] the details of those interactions are unclear. There have been few trials designed to judge the effectiveness of melatonin in disease treatment. Most existing data are based on small, incomplete clinical trials. Any positive immunological effect is thought to result from melatonin acting on high affinity receptors (MT1 and MT2) expressed in immunocompetent cells. In preclinical studies, melatonin may enhance cytokine production,[97] and by doing this counteract acquired immunodeficiences. Some studies also suggest that melatonin might be useful fighting infectious disease[9] including viral, such as HIV, and bacterial infections, and potentially in the treatment of cancer.[98]
Endogenous melatonin in human lymphocytes has been related to interleukin-2 (IL-2) production and to the expression of IL-2 receptor.[99] This suggests that melatonin is involved in the clonal expansion of antigen-stimulated human T lymphocytes. When taken in conjunction with calcium, it is an immunostimulator[citation needed] and is used as an adjuvant in some clinical protocols[citation needed]; conversely, the increased immune system activity may aggravate autoimmune disorders. In rheumatoid arthritis patients, melatonin production has been found increased when compared to age-matched healthy controls.[100]
Although it has not yet been clearly demonstrated whether melatonin increases non-specific immunity with resulting contraindication in autoimmune diseases, an increase in the production of IL-2 and IL-1 was noted in cultured splenocytes.[101]
Dreaming
Some supplemental melatonin users report an increase in vivid dreaming. Extremely high doses of melatonin (50 mg) dramatically increased REM sleep time and dream activity in both people with and without narcolepsy.[102] Many psychoactive drugs, such as cannabis and lysergic acid diethylamide (LSD), increase melatonin synthesis.[102] It has been suggested that nonpolar (lipid-soluble) indolic hallucinogenic drugs emulate melatonin activity in the awakened state and that both act on the same areas of the brain.[102]
Autism
Individuals with autism spectrum disorders (ASD) may have lower than normal levels of melatonin. A 2008 study found that unaffected parents of individuals with ASD also have lower melatonin levels, and that the deficits were associated with low activity of the ASMT gene, which encodes the last enzyme of melatonin synthesis.[103]
Multiple small studies have demonstrated that 2 to 10 mg of melatonin may benefit children with ASD who have trouble falling asleep and/or maintaining sleep. A small 2011 randomized crossover trial found that the administration of melatonin, when compared to placebo, decreased sleep latency and increased total sleep time, but had no effect on the number of night time awakenings.[104] At this time, no guidelines exist for the use of melatonin in children with ASD.
Aging
Research has supported the anti-aging properties of melatonin. Younger children hit their peak melatonin production at night, and some researchers believe that the level of melatonin peaks earlier as we get older. This may explain why older adults go to bed earlier, wake up earlier, and have more sleep problems than children do.[105]
Some studies have shown that melatonin plays a crucial part in the aging process and that it may act as an anti-aging agent when taken by older adults. It has been reported in one study that while elderly people have different gene expression levels in 100 of 10,000 genes, administration of melatonin may reverse this change in gene expression thus making the genes of elderly people similar to those of younger people.[106]
One study conducted by researchers of the University of Granada’s Institute of Biotechnology found that consuming melatonin may neutralize oxidative damage and delay the neurodegenerative process of aging. When small amounts of melatonin were administered to lab mice, it reduced the oxidative damage caused by aging and delayed the inflammatory process, which in turn increased the longevity of the mice. The researchers hope these results can also be applied to humans.[107]
Melanin production
Along with melanocyte-stimulating hormone, melatonin controls the dispersion of melanin throughout melanocyte cells.[108] Melatonin controls pigmentation changes by aggregation of melanin into the melanocytes within the skin, causing the skin to change color. This is responsible for the change in skin color due to amount of sleep or the appearance of those who are sleep deprived, since melatonin also controls the circadian cycle. This interaction is also responsible for the skin color of elderly people, since melatonin production reduces with age.[citation needed]
Use as medication
The hormone melatonin is used to treat circadian rhythm sleep disorders and some types of insomnia.
Studies have found that the use of melatonin can help entrain the circadian clock to environmental cycles and have beneficial effects for the treatment of certain forms of insomnia (2004).[109] Prolonged release melatonin has shown good results in treating insomnia in older adults (2007).[110]
A 2004 review found that melatonin significantly increased total sleep time in people suffering from sleep restriction.[17]
Other studies have found that for certain types of sleep disorders, melatonin is not effective. A 2006 review found that although it is safe for short term use (of three months or less), there is "no evidence that melatonin is effective in treating secondary sleep disorders or sleep disorders accompanying sleep restriction, such as jet lag and shiftwork disorder."[59]
In a 2005 study, researchers concluded that while "there is some evidence to suggest that melatonin is effective in treating delayed sleep phase syndrome, ...there is evidence to suggest that melatonin is not effective in treating most primary sleep disorders with short-term use (4 weeks or less)."[60]
Dosage
Melatonin tablets/capsules often contain three to ten times the amount needed to produce physiologic nocturnal blood melatonin levels for a more rapid sleep onset. Studies suggest that smaller doses (for example 0.3 mg as opposed to 3 mg) are just as effective.[62]
Large doses of melatonin can even be counterproductive: Lewy et al.[111] provide support to the "idea that too much melatonin may spill over onto the wrong zone of the melatonin phase-response curve" (PRC). In one of their blind subjects, 0.5 mg of melatonin was effective while 20 mg was not. Solomon Labs tested initial doses of 30 and 60 milligrams and found very little efficacy even at those levels.[111][112]
History
Melatonin is related to the mechanism by which some amphibians and reptiles change the color of their skin and, indeed, it was in this connection the substance first was discovered.[113][114] As early as 1917, McCord and Allen discovered (J Exptl Zool, 1917) that extract of the pineal glands of cows lightened frog skin.[12] Dermatology professor Aaron B. Lerner and colleagues at Yale University, in the hope that a substance from the pineal might be useful in treating skin diseases, isolated and named the hormone melatonin in 1958.[115] In the mid-70s Lynch et al. demonstrated[116] that the production of melatonin exhibits a circadian rhythm in human pineal glands. The discovery that melatonin is an antioxidant was made in 1993.[85] Around the same time, the hormone got a lot of press as a possible treatment for many illnesses.[117] The New England Journal of Medicine editorialized in 2000: "The hype and the claims of the so-called miraculous powers of melatonin several years ago did a great disservice to a scientific field of real importance to human health. (...) Our 24-hour society, with its chaotic time cues and lack of natural light, may yet reap substantial benefits."[118]
Availability
Legal availability of melatonin varies in different countries, ranging from being available without prescription (e.g., in most of North America) to being available only on prescription or not at all (although its possession and use may not be illegal). The hormone may be administered orally, as capsules, tablets or liquid, sublingually, or as transdermal patches.
Dietary supplement
In the USA, because it is sold as a dietary supplement, sometimes combined with other ingredients, such as vitamins and herbal extracts, and not as a drug, the Food and Drug Administration (FDA) regulations that apply to medications are not applicable to melatonin.[4] However, new FDA rules required that by June 2010 all production of dietary supplements must comply with "current good manufacturing practices" (cGMP), and be manufactured with "controls that result in a consistent product free of contamination, with accurate labeling."[119] In addition, the industry has been required to report to the FDA "all serious dietary supplement related adverse events" and the FDA has, within the cGMP guidelines, recently begun enforcement of that requirement.
Food Products
As reported in the New York Times in May 2011,[120] melatonin is sold in grocery stores, convenience stores, and clubs in both beverage and snack forms. The FDA is considering whether these food products can continue to be sold with the label "dietary supplements".
Pediatrics
While the packaging of melatonin often warns against use in children, at least one long-term study[121] does assess effectiveness and safety in children. No serious safety concerns were noted in any of the 94 cases studied by means of a structured questionnaire for the parents. With a mean follow-up time of 3.7 years, long-term medication was effective against sleep onset problems in 88% of the cases.
Prolonged release
Melatonin is available as a prolonged-release prescription drug, trade-name Circadin, manufactured by Neurim Pharmaceuticals. The European Medicines Agency (EMA) has approved Circadin 2 mg (prolonged-release melatonin) for patients aged 55 or over, as monotherapy for the short-term treatment (up to 13 weeks) of primary insomnia characterized by poor quality of sleep.[122]
See also
- Ramelteon
- 5-Methoxytryptamine
- Agomelatine
- Tasimelteon
- Risks and benefits of sun exposure
- Discovery and development of melatonin receptor agonists
References
- ^ "Melatonin". Sleepdex. http://www.sleepdex.org/melatonin.htm. Retrieved 2011-08-17.
- ^ Caniato, R; Filippini, R; Piovan, A; Puricelli, L; Borsarini, A; Cappelletti, EM (2003). "Melatonin in plants". Advances in experimental medicine and biology 527: 593–7. PMID 15206778.
- ^ a b Paredes, S. D.; Korkmaz, A.; Manchester, L. C.; Tan, D.-X.; Reiter, R. J. (2008). "Phytomelatonin: a review". Journal of Experimental Botany 60 (1): 57–69. doi:10.1093/jxb/ern284. PMID 19033551.
- ^ a b c Altun, A.; Ugur-Altun, B. (2007). "Melatonin: therapeutic and clinical utilization". International Journal of Clinical Practice 61 (5): 835–45. doi:10.1111/j.1742-1241.2006.01191.x. PMID 17298593.
- ^ Boutin, Jean A.; Audinot, Valérie; Ferry, Gilles; Delagrange, Philippe (2005). "Molecular tools to study melatonin pathways and actions". Trends in Pharmacological Sciences 26 (8): 412–9. doi:10.1016/j.tips.2005.06.006. PMID 15992934.
- ^ a b Hardeland, RüDiger (2005). "Antioxidative Protection by Melatonin: Multiplicity of Mechanisms from Radical Detoxification to Radical Avoidance". Endocrine 27 (2): 119–30. doi:10.1385/ENDO:27:2:119. PMID 16217125.
- ^ Reiter, Russel J.; Acuña-Castroviejo, Dario; Tan, DUN-Xian; Burkhardt, Susanne (2006). "Free Radical-Mediated Molecular Damage". Annals of the New York Academy of Sciences 939: 200–15. doi:10.1111/j.1749-6632.2001.tb03627.x. PMID 11462772.
- ^ a b Challet, E. (2007). "Minireview: Entrainment of the Suprachiasmatic Clockwork in Diurnal and Nocturnal Mammals". Endocrinology 148 (12): 5648–55. doi:10.1210/en.2007-0804. PMID 17901231.
- ^ a b Maestroni, Georges JM (2001). "The immunotherapeutic potential of melatonin". Expert Opinion on Investigational Drugs 10 (3): 467–76. doi:10.1517/13543784.10.3.467. PMID 11227046.
- ^ Conti, Ario; Conconi, Stefano; Hertens, Elisabeth; Skwarlo-Sonta, Krystyna; Markowska, Magda; Maestroni, Georges J.M. (2000). "Evidence for melatonin synthesis in mouse and human bone marrow cells". Journal of Pineal Research 28 (4): 193–202. doi:10.1034/j.1600-079X.2000.280401.x. PMID 10831154.
- ^ a b Hattori, A; Migitaka, H; Iigo, M; Itoh, M; Yamamoto, K; Ohtani-Kaneko, R; Hara, M; Suzuki, T et al. (1995). "Identification of melatonin in plants and its effects on plasma melatonin levels and binding to melatonin receptors in vertebrates". Biochemistry and molecular biology international 35 (3): 627–34. PMID 7773197.
- ^ a b c Coates, Paul M. (2005). Encyclopedia of Dietary Supplements. Marc R. Blackman, Gordon M. Cragg, Mark Levine, Joel Moss, Jeffrey D. White. CRC Press. pp. 457–466. ISBN 0824755049. http://books.google.com/?id=Sfmc-fRCj10C&pg=PA457&lpg=PA457&dq=Lerner+melatonin+history. Retrieved 2009-03-31.
- ^ Ratzburg, Courtney (Undated). "Melatonin: The Myths and Facts". Vanderbilt University. http://www.vanderbilt.edu/AnS/psychology/health_psychology/melatonin.htm. Retrieved 2007-12-02.
- ^ USPS. "Country Conditions for Mailing — Germany". http://pe.usps.gov/text/imm/fh_011.htm. Retrieved 2008-01-15.
- ^ Lewy, A.; Sack, R.; Miller, L.; Hoban, T. (1987). "Antidepressant and circadian phase-shifting effects of light". Science 235 (4786): 352–4. doi:10.1126/science.3798117. PMID 3798117.
- ^ Uz, Tolga; Akhisaroglu, Mustafa; Ahmed, Rehan; Manev, Hari (2003). "The Pineal Gland is Critical for Circadian Period1 Expression in the Striatum and for Circadian Cocaine Sensitization in Mice". Neuropsychopharmacology 28 (12): 2117–23. doi:10.1038/sj.npp.1300254. PMID 12865893.
- ^ a b c Buscemi, N. et al. (2004). "Melatonin for Treatment of Sleep Disorders. Summary, Evidence Report/Technology Assessment: Number 108" (Review). U.S. Department of Health & Human Services, Agency for Healthcare Research and Quality. http://www.ahrq.gov/clinic/epcsums/melatsum.htm. Retrieved 2010-05-25.
- ^ Weichselbaum, Simone (2010-02-07). "New melatonin-based drinks help restless sleepers hit the hay with just a gulp - but pack hormones". Daily News (New York). http://www.nydailynews.com/lifestyle/2010/02/07/2010-02-07_these_drinksll_knock_you_out_dozeinducing_concoctions_include_dose_of_hormones.html.
- ^ Chronobiotics: Selected Agents of Potential Value in Jet Lag and other Dyschronisms, H. W. Simpson, of Glasgow University, in Chronobiology: Principles and Application to Shifts in Schedules, ed: L.E. Sheving and F. Hagberg, Springer, Berlin, 1979. Retrieved September 23, 2009.[page needed]
- ^ Mundey, K; Benloucif, S; Harsanyi, K; Dubocovich, ML; Zee, PC (2005). "Phase-dependent treatment of delayed sleep phase syndrome with melatonin". Sleep 28 (10): 1271–8. PMID 16295212.
- ^ Straif et al. (2007). "Carcinogenicity of shift-work, painting, and fire-fighting". The Lancet Oncology 8 (12): 1065–1066. doi:10.1016/S1470-2045(07)70373-X. PMID 19271347. http://www.thelancet.com/journals/lanonc/article/PIIS147020450770373X/fulltext. Retrieved 2011-08-17.
- ^ Straif, Kurt; Baan, Robert; Grosse, Yann; Secretan, BéAtrice; Ghissassi, Fatiha El; Bouvard, VéRonique; Altieri, Andrea; Benbrahim-Tallaa, Lamia et al. (2007). "Carcinogenicity of shift-work, painting, and fire-fighting". The Lancet Oncology 8 (12): 1065–6. doi:10.1016/S1470-2045(07)70373-X. PMID 19271347.
- ^ Larson, John; Jessen, Ruth E.; Uz, Tolga; Arslan, Ahmet D.; Kurtuncu, Murat; Imbesi, Marta; Manev, Hari (2006). "Impaired hippocampal long-term potentiation in melatonin MT2 receptor-deficient mice". Neuroscience Letters 393 (1): 23–6. doi:10.1016/j.neulet.2005.09.040. PMID 16203090.
- ^ Pappolla, MA; Sos, M; Omar, RA; Bick, RJ; Hickson-Bick, DL; Reiter, RJ; Efthimiopoulos, S; Robakis, NK (1997). "Melatonin prevents death of neuroblastoma cells exposed to the Alzheimer amyloid peptide". The Journal of neuroscience : the official journal of the Society for Neuroscience 17 (5): 1683–90. PMID 9030627.
- ^ Pappolla, M.; Bozner, P; Soto, C; Shao, H; Robakis, NK; Zagorski, M; Frangione, B; Ghiso, J (1998). "Inhibition of Alzheimer beta -Fibrillogenesis by Melatonin". Journal of Biological Chemistry 273 (13): 7185–8. doi:10.1074/jbc.273.13.7185. PMID 9516407.
- ^ Wang, XC; Zhang, J; Yu, X; Han, L; Zhou, ZT; Zhang, Y; Wang, JZ (2005). "Prevention of isoproterenol-induced tau hyperphosphorylation by melatonin in the rat". Sheng li xue bao : [Acta physiologica Sinica] 57 (1): 7–12. PMID 15719129.
- ^ Volicer, L.; Harper, DG; Manning, BC; Goldstein, R; Satlin, A (2001). "Sundowning and Circadian Rhythms in Alzheimer's Disease". American Journal of Psychiatry 158 (5): 704–11. doi:10.1176/appi.ajp.158.5.704. PMID 11329390.
- ^ Al-Aama, Tareef; Brymer, Christopher; Gutmanis, Iris; Woolmore-Goodwin, Sarah M.; Esbaugh, Jacquelin; Dasgupta, Monidipa (2011). "Melatonin decreases delirium in elderly patients: A randomized, placebo-controlled trial". International Journal of Geriatric Psychiatry 26 (7): 687–694. doi:10.1002/gps.2582. PMID 20845391.
- ^ Tjon Pian Gi, Cecil V.; Broeren, Joris P. A.; Starreveld, J. Sander; A. Versteegh, Florens G (2003). "Melatonin for treatment of sleeping disorders in children with attention deficit/hyperactivity disorder: a preliminary open label study". European Journal of Pediatrics 162 (7–8): 554–5. doi:10.1007/s00431-003-1207-x. PMID 12783318.
- ^ Bellipanni, G; Di Marzo, F; Blasi, F; Di Marzo, A (2005). "Effects of Melatonin in Perimenopausal and Menopausal Women: Our Personal Experience". Annals of the New York Academy of Sciences 1057: 393–402. doi:10.1196/annals.1356.030. PMID 16399909.
- ^ "Melatonin". About.com: Sleep Disorders. pp. 4. http://sleepdisorders.about.com/cs/melatonin/a/melatonin_4.htm.
- ^ Terzolo, M; Revelli, A; Guidetti, D; Piovesan, A; Cassoni, P; Paccotti, P; Angeli, A; Massobrio, M (1993). "Evening administration of melatonin enhances the pulsatile secretion of prolactin but not of LH and TSH in normally cycling women". Clinical endocrinology 39 (2): 185–91. doi:10.1111/j.1365-2265.1993.tb01772.x. PMID 8370131.
- ^ "The infertility drug?". What Doctors Don't Tell You. http://www.wddty.com/the-infertility-drug.html. Retrieved 2011-08-17.
- ^ Jahnke, G; Marr, M; Myers, C; Wilson, R; Travlos, G; Price, C (1999). "Maternal and developmental toxicity evaluation of melatonin administered orally to pregnant Sprague-Dawley rats". Toxicological Sciences 50 (2): 271–9. doi:10.1093/toxsci/50.2.271. PMID 10478864.
- ^ Dodick, David W.; Capobianco, David J. (2001). "Treatment and management of cluster headache". Current Pain and Headache Reports 5 (1): 83–91. doi:10.1007/s11916-001-0015-0. PMID 11252143.
- ^ Gagnier, JJ (2001). "The therapeutic potential of melatonin in migraines and other headache types". Alternative medicine review : a journal of clinical therapeutic 6 (4): 383–9. PMID 11578254.
- ^ "Properly Timed Light, Melatonin Lift Winter Depression By Syncing Rhythms". National Institute of Mental Health. 2006-05-01. http://www.nimh.nih.gov/science-news/2006/properly-timed-light-melatonin-lift-winter-depression-by-syncing-rhythms.shtml. Retrieved 2011-08-17.
- ^ Bhattacharjee, Yudhijit (14 September 2007). "Is Internal Timing Key to Mental Health?" (PDF). ScienceMag (AAAS) 317: 1488–90. http://www.ohsu.edu/ohsuedu/academic/som/images/Al-Lewy-Science.pdf. Retrieved 2008-02-18.
- ^ Lewy, AJ; Nurnberger Jr, JI; Wehr, TA; Pack, D; Becker, LE; Powell, RL; Newsome, DA (1985). "Supersensitivity to light: possible trait marker for manic-depressive illness". The American journal of psychiatry 142 (6): 725–7. PMID 4003592. http://ajp.psychiatryonline.org/cgi/pmidlookup?view=long&pmid=4003592.
- ^ Whalley, L; Perini, T; Shering, A; Bennie, J (1991). "Melatonin response to bright light in recovered, drug-free, bipolar patients". Psychiatry Research 38 (1): 13–9. doi:10.1016/0165-1781(91)90048-T. PMID 1658841.
- ^ Mills, Edward; Wu, Ping; Seely, Dugald; Guyatt, Gordon (2005). "Melatonin in the treatment of cancer: a systematic review of randomized controlled trials and meta-analysis". Journal of Pineal Research 39 (4): 360–6. doi:10.1111/j.1600-079X.2005.00258.x. PMID 16207291.
- ^ "Melatonin as an anticancer agent with and without chemotherapy: systematic review and meta-analysis [item 3]." Canadian College of Naturopathic Medicine. Current Research.
- ^ Navara, Kristen J.; Nelson, Randy J. (2007). "The dark side of light at night: physiological, epidemiological, and ecological consequences". Journal of Pineal Research 43 (3): 215–224. doi:10.1111/j.1600-079X.2007.00473.x. PMID 17803517. http://www.psy.ohio-state.edu/nelson/documents/JPinealRes2007.pdf.
- ^ Schernhammer, ES; Rosner, B; Willett, WC; Laden, F; Colditz, GA; Hankinson, SE (2004). "Epidemiology of urinary melatonin in women and its relation to other hormones and night work". Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology 13 (6): 936–43. PMID 15184249.
- ^ a b Koppisetti, Sreedevi; Jenigiri, Bharat; Terron, M. Pilar; Tengattini, Sandra; Tamura, Hiroshi; Flores, Luis J.; Tan, Dun-Xian; Reiter, Russel J. (2008). "Reactive Oxygen Species and the Hypomotility of the Gall Bladder as Targets for the Treatment of Gallstones with Melatonin: A Review". Digestive Diseases and Sciences 53 (10): 2592–603. doi:10.1007/s10620-007-0195-5. PMID 18338264.
- ^ Tan, D; Manchester, LC; Reiter, RJ; Qi, W; Hanes, MA; Farley, NJ (1999). "High physiological levels of melatonin in the bile of mammals". Life Sciences 65 (23): 2523–9. doi:10.1016/S0024-3205(99)00519-6. PMID 10622237.
- ^ Weishaupt, Jochen H.; Bartels, Claudia; Pölking, Esther; Dietrich, Jeannine; Rohde, Gundula; Poeggeler, Burkhard; Mertens, Nina; Sperling, Swetlana et al. (2006). "Reduced oxidative damage in ALS by high-dose enteral melatonin treatment". Journal of Pineal Research 41 (4): 313–23. doi:10.1111/j.1600-079X.2006.00377.x. PMID 17014688.
- ^ Wolden-Hanson, T; Mitton, DR; McCants, RL; Yellon, SM; Wilkinson, CW; Matsumoto, AM; Rasmussen, DD (2000). "Daily melatonin administration to middle-aged male rats suppresses body weight, intraabdominal adiposity, and plasma leptin and insulin independent of food intake and total body fat". Endocrinology 141 (2): 487–97. doi:10.1210/en.141.2.487. PMID 10650927.
- ^ Tan, D.-X.; Manchester, L. C.; Fuentes-Broto, L.; Paredes, S. D.; Reiter, R. J. (2011). "Significance and application of melatonin in the regulation of brown adipose tissue metabolism: relation to human obesity". Obesity Reviews 12 (3): 167–88. doi:10.1111/j.1467-789X.2010.00756.x. PMID 20557470.
- ^ <Please add first missing authors to populate metadata.>; Meltz, ML; Reiter, RJ; Herman, TS; Kumar, KS (1999). "Melatonin and protection from whole-body irradiation: survival studies in mice". Mutation research 425 (1): 21–7. PMID 10082913.
- ^ Reiter, R; Herman, T; Meltz, M (1996). "Melatonin and radioprotection from genetic damage: In vivo/in vitro studies with human volunteers". Mutation Research/Genetic Toxicology 371 (3–4): 221–8. doi:10.1016/S0165-1218(96)90110-X.
- ^ <Please add first missing authors to populate metadata.>; Reiter, RJ; Herman, TS; Meltz, ML (1998). "Melatonin reduces gamma radiation-induced primary DNA damage in human blood lymphocytes". Mutation research 397 (2): 203–8. PMID 9541644.
- ^ Topkan, Erkan; Tufan, Hale; Yavuz, Ali Aydin; Bacanli, Didem; Onal, Cem; Kosdak, Sebnem; Yavuz, Melek Nur (2008). "Comparison of the protective effects of melatonin and amifostine on radiation-induced epiphyseal injury". International Journal of Radiation Biology 84 (10): 796–802. doi:10.1080/09553000802389678. PMID 18979313.
- ^ Tan, Dun-Xian; Manchester, Lucien C.; Terron, Maria P.; Flores, Luis J.; Reiter, Russel J. (2007). "One molecule, many derivatives: A never-ending interaction of melatonin with reactive oxygen and nitrogen species?". Journal of Pineal Research 42 (1): 28–42. doi:10.1111/j.1600-079X.2006.00407.x. PMID 17198536.
- ^ Shirazi, Alireza; Ghobadi, Ghazaleh; Ghazi-Khansari, Mahmoud (2007). "A Radiobiological Review on Melatonin: A Novel Radioprotector". Journal of Radiation Research 48 (4): 263–72. doi:10.1269/jrr.06070. PMID 17641465.
- ^ Sotthibundhu, Areechun; Phansuwan-Pujito, Pansiri; Govitrapong, Piyarat (2010). "Melatonin increases proliferation of cultured neural stem cells obtained from adult mouse subventricular zone". Journal of Pineal Research 49 (3): 291–300. doi:10.1111/j.1600-079X.2010.00794.x. PMID 20663047.
- ^ Kunz, D; Bes, F (2001). "Exogenous melatonin in periodic limb movement disorder: an open clinical trial and a hypothesis". Sleep 24 (2): 183–7. PMID 11247054.
- ^ Basu P.P., Pacana T., Shah N., Hampole H., Krishnaswamy N., Rayapudi K. "Role of melatonin in colonic motility in irritable bowel syndrome - Constipation MIMI-C-a double blinded randomized placebocontrol clinical trial". in <Please add first missing authors to populate metadata.> (2010). "Poster Presentations". Neurogastroenterology & Motility 22: 23–90. doi:10.1111/j.1365-2982.2010.01549.x.
- ^ a b c d Buscemi, N.; Vandermeer, B; Hooton, N; Pandya, R; Tjosvold, L; Hartling, L; Vohra, S; Klassen, TP et al. (2006). "Efficacy and safety of exogenous melatonin for secondary sleep disorders and sleep disorders accompanying sleep restriction: meta-analysis". BMJ 332 (7538): 385–93. doi:10.1136/bmj.38731.532766.F6. PMC 1370968. PMID 16473858. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1370968.
- ^ a b Buscemi, Nina; Vandermeer, Ben; Hooton, Nicola; Pandya, Rena; Tjosvold, Lisa; Hartling, Lisa; Baker, Glen; Klassen, Terry P. et al. (2005). "The Efficacy and Safety of Exogenous Melatonin for Primary Sleep Disorders. A Meta-Analysis". Journal of General Internal Medicine 20 (12): 1151–8. doi:10.1111/j.1525-1497.2005.0243.x. PMC 1490287. PMID 16423108. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1490287.
- ^ Brent Bauer, M.D.. "Melatonin side effects: What are the risks?". Mayo Clinic. http://www.mayoclinic.com/health/melatonin-side-effects/AN01717. Retrieved 2011-08-17.
- ^ a b Zhdanova, I. V. (2001). "Melatonin Treatment for Age-Related Insomnia". Journal of Clinical Endocrinology & Metabolism 86 (10): 4727–30. doi:10.1210/jc.86.10.4727.
- ^ Sack, Robert L.; Brandes, Richard W.; Kendall, Adam R.; Lewy, Alfred J. (2000). "Entrainment of Free-Running Circadian Rhythms by Melatonin in Blind People". New England Journal of Medicine 343 (15): 1070–7. doi:10.1056/NEJM200010123431503. PMID 11027741.
- ^ Morera, AL; Henry, M; De La Varga, M (2001). "Safety in melatonin use". Actas espanolas de psiquiatria 29 (5): 334–7. PMID 11602091.
- ^ Terry, Paul D.; Villinger, Francois; Bubenik, George A.; Sitaraman, Shanti V. (2009). "Melatonin and ulcerative colitis: Evidence, biological mechanisms, and future research". Inflammatory Bowel Diseases 15 (1): 134–40. doi:10.1002/ibd.20527. PMID 18626968.
- ^ "Study Shows Melatonin Supplements May Make Standing A Hazard For The Cardiovascular-Challenged" (DOC) (Press release). Penn State College of Medicine, Milton S. Hershey Medical Center. September 2003. http://www.hmc.psu.edu/news/pr/2003/sept/Ray_melatonin.doc. Retrieved 2006-07-21. (MS Word Format)
- ^ "Melatonin". Drugs.com. http://www.drugs.com/melatonin.html. Retrieved 2011-08-17.
- ^ Burkhardt, Susanne; Tan, Dun Xian; Manchester, Lucien C.; Hardeland, RüDiger; Reiter, Russel J. (2001). "Detection and Quantification of the Antioxidant Melatonin in Montmorency and Balaton Tart Cherries (Prunus cerasus)". Journal of Agricultural and Food Chemistry 49 (10): 4898–902. doi:10.1021/jf010321. PMID 11600041.
- ^ Tan, Dun-Xian; Hardeland, RüDiger; Manchester, Lucien C.; Paredes, Sergio D.; Korkmaz, Ahmet; Sainz, Rosa M.; Mayo, Juan C.; Fuentes-Broto, Lorena et al. (2009). "The changing biological roles of melatonin during evolution: from an antioxidant to signals of darkness, sexual selection and fitness". Biological Reviews: 607–23. doi:10.1111/j.1469-185X.2009.00118.x.
- ^ Lincoln, G.; Andersson, H; Loudon, A (2003). "Clock genes in calendar cells as the basis of annual timekeeping in mammals--a unifying hypothesis". Journal of Endocrinology 179 (1): 1–13. doi:10.1677/joe.0.1790001. PMID 14529560.
- ^ a b Arendt, Josephine; Skene, Debra Jean (2005). "Melatonin as a chronobiotic". Sleep Medicine Reviews 9 (1): 25–39. doi:10.1016/j.smrv.2004.05.002. PMID 15649736. "Exogenous melatonin has acute sleepiness-inducing and temperature-lowering effects during 'biological daytime', and when suitably timed (it is most effective around dusk and dawn) it will shift the phase of the human circadian clock (sleep, endogenous melatonin, core body temperature, cortisol) to earlier (advance phase shift) or later (delay phase shift) times."
- ^ Chaturvedi, CM (1984). "Effect of Melatonin on the Adrenl and Gonad of the Common Mynah Acridtheres tristis". Australian Journal of Zoology 32 (6): 803–9. doi:10.1071/ZO9840803. http://www.publish.csiro.au/paper/ZO9840803.htm.
- ^ Chen, H.J. (1981). "Spontaneous and Melatonin-Induced Testicular Regression in Male Golden Hamsters: Augmented Sensitivity of the Old Male to Melatonin Inhibition". Neuroendocrinology 33 (1): 43–6. doi:10.1159/000123198. PMID 7254478.
- ^ Kaur C, Ling EA (2008). "Antioxidants and neuroprotection in the adult and developing central nervous system". Curr. Med. Chem. 15 (29): 3068–80. PMID 19075654.
- ^ Nayak, S. Kumar; Jegla, T.; Panda, S. (2006). "Role of a novel photopigment, melanopsin, in behavioral adaptation to light". Cellular and Molecular Life Sciences 64 (2): 144–54. doi:10.1007/s00018-006-5581-1. PMID 17160354.
- ^ Roberts, Joan E. (2005). "Update on the Positive Effects of Light in Humans". Photochemistry and Photobiology: 490–2. doi:10.1562/2004-12-02-IR-391.
- ^ Richardson, GS (2005). "The human circadian system in normal and disordered sleep". The Journal of clinical psychiatry 66 Suppl 9: 3–9; quiz 42–3. PMID 16336035.
- ^ Perreau-Lenz, StéPhanie; Pévet, Paul; Buijs, Ruud M.; Kalsbeek, Andries (2004). "The Biological Clock: The Bodyguard of Temporal Homeostasis". Chronobiology International 21 (1): 1–25. doi:10.1081/CBI-120027984. PMID 15129821.
- ^ Ardura, Julio; Gutierrez, Regina; Andres, Jesus; Agapito, Teresa (2003). "Emergence and Evolution of the Circadian Rhythm of Melatonin in Children". Hormone Research 59 (2): 66–72. doi:10.1159/000068571. PMID 12589109.
- ^ Sack, RL; Lewy, AJ; Erb, DL; Vollmer, WM; Singer, CM (1986). "Human melatonin production decreases with age". Journal of pineal research 3 (4): 379–88. doi:10.1111/j.1600-079X.1986.tb00760.x. PMID 3783419.
- ^ Gavin, Mary L.; Scavina, Mena T. (2009). "Why Aren't Teens Getting Enough Sleep?". How Much Sleep Do I Need?. http://kidshealth.org/teen/your_body/take_care/how_much_sleep.html.
- ^ Wirz-Justice, A; Benedetti, F; Terman, M (2009). Chronotherapeutics for Affective Disorders: A Clinician’s Manual for Light and Wake Therapy. Basel: Karger. ISBN 978-3-8055-9120-1.[page needed]
- ^ Brainard, GC; Hanifin, JP; Greeson, JM; Byrne, B; Glickman, G; Gerner, E; Rollag, MD (2001). "Action spectrum for melatonin regulation in humans: evidence for a novel circadian photoreceptor". The Journal of neuroscience : the official journal of the Society for Neuroscience 21 (16): 6405–12. PMID 11487664.
- ^ Kayumov, L. (2005). "Blocking Low-Wavelength Light Prevents Nocturnal Melatonin Suppression with No Adverse Effect on Performance during Simulated Shift Work". Journal of Clinical Endocrinology & Metabolism 90 (5): 2755–61. doi:10.1210/jc.2004-2062.
- ^ a b Tan, D. X.; Chen, L. D.; Poeggeler, B.; Manchester, L. C.; Reiter, R. J. (1993). "Melatonin: a potent, endogenous hydroxyl radical scavenger". Endocrine J 1: 57–60.
- ^ a b Tan, Dun-Xian; Manchester, Lucien C.; Terron, Maria P.; Flores, Luis J.; Reiter, Russel J. (2007). "One molecule, many derivatives: A never-ending interaction of melatonin with reactive oxygen and nitrogen species?". Journal of Pineal Research 42 (1): 28–42. doi:10.1111/j.1600-079X.2006.00407.x. PMID 17198536.
- ^ Reiter RJ, Manchester LC, Tan DX (September 2010). "Neurotoxins: free radical mechanisms and melatonin protection". Curr Neuropharmacol 8 (3): 194–210. doi:10.2174/157015910792246236. PMC 3001213. PMID 21358970. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=3001213.
- ^ Poeggeler, Burkhard; Saarela, Seppo; Reiter, Russel J.; Tan, DUN-Xian; Chen, LI-DUN; Manchester, Lucien C.; Barlow-Walden, Lornell R. (2006). "Melatonin-A Highly Potent Endogenous Radical Scavenger and Electron Donor: New Aspects of the Oxidation Chemistry of this Indole Accessed in vitroa". Annals of the New York Academy of Sciences 738: 419–20. doi:10.1111/j.1749-6632.1994.tb21831.x. PMID 7832450.
- ^ Tan, Dun-Xian; Manchester, Lucien C.; Reiter, Russel J.; Qi, Wen-Bo; Karbownik, Malgorzata; Calvo, Juan R. (2000). "Significance of Melatonin in Antioxidative Defense System: Reactions and Products". Neurosignals 9 (3–4): 137–59. doi:10.1159/000014635. PMID 10899700.
- ^ Karbownik, M; Reiter, Russel J.; Cabrera, Javier; Garcia, Joaquin J. (2001). "Comparison of the protective effect of melatonin with other antioxidants in the hamster kidney model of estradiol-induced DNA damage". Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis 474: 87–92. doi:10.1016/S0027-5107(00)00164-0.
- ^ Tutunculer, Filiz; Eskiocak, Sevgi; Basaran, Umit Nusret; Ekuklu, Galip; Ayvaz, Suleyman; Vatansever, Ulfet (2005). "The protective role of melatonin in experimental hypoxic brain damage". Pediatrics International 47 (4): 434–9. doi:10.1111/j.1442-200x.2005.02085.x. PMID 16091083.
- ^ Ward Dean, John Morgenthaler, Steven William Fowkes (1993). Smart Drugs II: The Next Generation : New Drugs and Nutrients to Improve Your Memory and Increase Your Intelligence (Smart Drug Series, V. 2). Smart Publications. ISBN 0-9627418-7-6.[page needed]
- ^ Anisimov, V; Alimova, IN; Baturin, DA; Popovich, IG; Zabezhinski, MA; Rosenfeld, SV; Manton, KG; Semenchenko, AV et al. (2003). "Dose-dependent effect of melatonin on life span and spontaneous tumor incidence in female SHR mice". Experimental Gerontology 38 (4): 449–61. doi:10.1016/S0531-5565(02)00240-1. PMID 12670632.
- ^ Oaknin-Bendahan, Sol; Anis, Yossi; Nir, Isaac; Zisapel, Nava (1995). "Effects of long-term administration of melatonin and a putative antagonist on the ageing rat". NeuroReport 6 (5): 785–8. doi:10.1097/00001756-199503270-00020. PMID 7605949.
- ^ Carrillo-Vico, Antonio; Guerrero, Juan M.; Lardone, Patricia J.; Reiter, Russel J. (2005). "A Review of the Multiple Actions of Melatonin on the Immune System". Endocrine 27 (2): 189–200. doi:10.1385/ENDO:27:2:189. PMID 16217132.
- ^ Arushanian, EB; Beĭer, EV (2002). "Immunotropic properties of pineal melatonin". Eksperimental'naia i klinicheskaia farmakologiia 65 (5): 73–80. PMID 12596522.
- ^ Carrillo-Vico, A; Reiter, RJ; Lardone, PJ; Herrera, JL; Fernández-Montesinos, R; Guerrero, JM; Pozo, D (2006). "The modulatory role of melatonin on immune responsiveness". Current opinion in investigational drugs (London, England : 2000) 7 (5): 423–31. PMID 16729718.
- ^ Maestroni, GJ (1999). "Therapeutic potential of melatonin in immunodeficiency states, viral diseases, and cancer". Advances in experimental medicine and biology 467: 217–26. PMID 10721059.
- ^ Carrillo-Vico, A. (2004). "Human Lymphocyte-Synthesized Melatonin Is Involved in the Regulation of the Interleukin-2/Interleukin-2 Receptor System". Journal of Clinical Endocrinology & Metabolism 90 (2): 992–1000. doi:10.1210/jc.2004-1429.
- ^ Cutolo, M; Maestroni, GJ (2005). "The melatonin-cytokine connection in rheumatoid arthritis". Annals of the Rheumatic Diseases 64 (8): 1109–11. doi:10.1136/ard.2005.038588. PMC 1755599. PMID 16014678. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1755599.
- ^ Arias, J; Melean, E; Valero, N; Pons, H; Chacín-Bonilla, L; Larreal, Y; Bonilla, E (2003). "Effect of melatonin on lymphocyte proliferation and production of interleukin-2 (IL-2) and interleukin-1 beta (IL-1 beta) in mice splenocytes". Investigacion clinica 44 (1): 41–50. PMID 12703182.
- ^ a b c Lewis, Alan (1999). Melatonin and the Biological Clock. McGraw-Hill. p. 23. ISBN 0-87983-734-9.
- ^ Melke, J; Goubran Botros, H; Chaste, P; Betancur, C; Nygren, G; Anckarsäter, H; Rastam, M; Ståhlberg, O et al. (2007). "Abnormal melatonin synthesis in autism spectrum disorders". Molecular Psychiatry 13 (1): 90–8. doi:10.1038/sj.mp.4002016. PMC 2199264. PMID 17505466. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2199264.
- ^ Wright, Barry; Sims, David; Smart, Siobhan; Alwazeer, Ahmed; Alderson-Day, Ben; Allgar, Victoria; Whitton, Clare; Tomlinson, Heather et al. (2010). "Melatonin Versus Placebo in Children with Autism Spectrum Conditions and Severe Sleep Problems Not Amenable to Behaviour Management Strategies: A Randomised Controlled Crossover Trial". Journal of Autism and Developmental Disorders 41 (2): 175–84. doi:10.1007/s10803-010-1036-5. PMID 20535539.
- ^ University of Maryland Medical Center. “Melatonin.” 2011. http://www.umm.edu/altmed/articles/melatonin-000315.htm
- ^ Sharman, Edward H.; Sharman, Kaizhi G.; Ge, Yuan-Wen; Lahiri, Debomoy K.; Bondy, Stephen C. (2004). "Age-related changes in murine CNS mRNA gene expression are modulated by dietary melatonin". Journal of Pineal Research 36 (3): 165–70. doi:10.1046/j.1600-079X.2003.00112.x. PMID 15009506.
- ^ Acuna-Castroviejo, Dario; Martin, Miguel; MacIas, Manuel; Escames, Germaine; Leon, Josefa; Khaldy, Huda; Reiter, Russel J. (2001). "Melatonin, mitochondria, and cellular bioenergetics". Journal of Pineal Research 30 (2): 65–74. doi:10.1034/j.1600-079X.2001.300201.x. PMID 11270481. Lay summary – Science Daily (April 24, 2007).
- ^ OxfordUniversity. "A Dictionary of Science: Fifth Edition". Oxford University Press, p.513
- ^ Turek, F; Gillette, M (2004). "Melatonin, sleep, and circadian rhythms: rationale for development of specific melatonin agonists". Sleep Medicine 5 (6): 523–32. doi:10.1016/j.sleep.2004.07.009. PMID 15511698.
- ^ Wade, Alan G; Ford, Ian; Crawford, Gordon; McMahon, Alex D; Nir, Tali; Laudon, Moshe; Zisapel, Nava (2007). "Efficacy of prolonged release melatonin in insomnia patients aged 55–80 years: quality of sleep and next-day alertness outcomes". Current Medical Research and Opinion 23 (10): 2597–605. doi:10.1185/030079907X233098. PMID 17875243.
- ^ a b Lewy, Alfred J.; Emens, Jonathan S.; Sack, Robert L.; Hasler, Brant P.; Bernert, Rebecca A. (2002). "Low, but not high, doses of melatonin entrained a free-running blind person with a long circadian period". Chronobiology International 19 (3): 649–58. doi:10.1081/CBI-120004546. PMID 12069043.
- ^ Sack, Robert L.; Brandes, Richard W.; Kendall, Adam R.; Lewy, Alfred J. (2000). "Entrainment of Free-Running Circadian Rhythms by Melatonin in Blind People". New England Journal of Medicine 343 (15): 1070–77. doi:10.1056/NEJM200010123431503. PMID 11027741.
- ^ Filadelfi, Ana Maria Caliman; Castrucci, Ana Maria de Lauro (1996). "Comparative aspects of the pineal/melatonin system of poikilothermic vertebrates". Journal of Pineal Research 20 (4): 175–86. doi:10.1111/j.1600-079X.1996.tb00256.x. PMID 8836950.
- ^ Sugden, David; Davidson, Kathryn; Hough, Kate A.; Teh, Muy-Teck (2004). "Melatonin, Melatonin Receptors and Melanophores: A Moving Story". Pigment Cell Research 17 (5): 454–60. doi:10.1111/j.1600-0749.2004.00185.x. PMID 15357831.
- ^ Lerner, AB; Case, JD; Takahashi, Y (1960). "Isolation of melatonin and 5-methoxyindole-3-acetic acid from bovine pineal glands". The Journal of biological chemistry 235: 1992–7. PMID 14415935.
- ^ Lynch, H.; Wurtman, R.; Moskowitz, M.; Archer, M.; Ho, M. (1975). "Daily rhythm in human urinary melatonin". Science 187 (4172): 169–71. doi:10.1126/science.1167425. PMID 1167425.
- ^ Arendt, J. (2005). "Melatonin: Characteristics, Concerns, and Prospects". Journal of Biological Rhythms 20 (4): 291–303. doi:10.1177/0748730405277492. PMID 16077149. "There is very little evidence in the short term for toxicity or undesirable effects in humans. The extraordinary “hype” of the miraculous powers of melatonin in the recent past did a disservice to acceptance of its genuine benefits."
- ^ Arendt, Josephine (2000). "Melatonin, Circadian Rhythms, and Sleep". New England Journal of Medicine 343 (15): 1114–6. doi:10.1056/NEJM200010123431510. PMID 11027748.
- ^ "FDA Issues Dietary Supplements Final Rule" (Press release). U.S. Food and Drug Administration. 2007-06-22. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2007/ucm108938.htm. Retrieved 2009-08-04.
- ^ url = http://www.nytimes.com/2011/05/15/us/15lazycakes.html?_r=1&scp=1&sq=lazy%20cakes&st=cse
- ^ Hoebert, Michel; Van Der Heijden, Kristiaan B.; Van Geijlswijk, Ingeborg M.; Smits, Marcel G. (2009). "Long-term follow-up of melatonin treatment in children with ADHD and chronic sleep onset insomnia". Journal of Pineal Research 47 (1): 1–7. doi:10.1111/j.1600-079X.2009.00681.x. PMID 19486273.
- ^ Medical News Today Circadin (Prolonged-Release Melatonin) For Primary Insomnia Recommended For Approval In The EU (27 Apr 2007)
External links
- Melatonin information from MedlinePlus
- Melatonin entry in TiHKAL • info
- Wade, Alan G; Ford, Ian; Crawford, Gordon; McConnachie, Alex; Nir, Tali; Laudon, Moshe; Zisapel, Nava (2010). "Nightly treatment of primary insomnia with prolonged release melatonin for 6 months: a randomized placebo controlled trial on age and endogenous melatonin as predictors of efficacy and safety". BMC Medicine 8: 51. doi:10.1186/1741-7015-8-51. PMC 2933606. PMID 20712869. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2933606.
Melatonergics Receptor
ligandsAfobazole • LuzindoleEnzyme
inhibitorsAANAT inhibitorsASMT inhibitorsOthers Ferrous Iron (Fe2+) • Magnesium (Mg2+) • S-Adenosyl-L-methionine • Tetrahydrobiopterin • Vitamin B3 (Niacin, nicotinamide → NADPH) • Vitamin B6 (Pyridoxine, Pyridoxamine, Pyridoxal → Pyridoxal phosphate) • Vitamin B9 (Folic acid → Tetrahydrofolic acid) • Vitamin C (Ascorbic acid) • Zinc (Zn2+)Neurotransmitters Amino acids Alanine · Aspartate · Cycloserine · DMG · GABA · Glutamate · Glycine · Hypotaurine · Kynurenic acid (Transtorine) · NAAG (Spaglumic acid) · NMG (Sarcosine) · Serine · Taurine · TMG (Betaine)
Endocannabinoids 2-AG · 2-AGE (Noladin ether) · AEA (Anandamide) · NADA · OAE (Virodhamine) · Oleamide · PEA (Palmitoylethanolamide) · RVD-Hpα · Hp (Hemopressin)
Gasotransmitters Monoamines Dopamine · Epinephrine (Adrenaline) · Melatonin · NAS (Normelatonin) · Norepinephrine (Noradrenaline) · Serotonin (5-HT)
Purines Trace amines 3-ITA · 5-MeO-DMT · Bufotenin · DMT · NMT · Octopamine · Phenethylamine · Synephrine · Thyronamine · Tryptamine · Tyramine
Others 1,4-BD · Acetylcholine · GBL · GHB · Histamine
Antioxidants Acetyl-L-Carnitine (ALCAR) • Alpha-Lipoic Acid (ALA) • Ascorbic Acid (Vitamin C) • Carotenoids (Vitamin A) • Curcumin • Edaravone • Polyphenols • Glutathione • Hydroxytyrosol • L-Carnitine • Ladostigil • Melatonin • N-Acetylcysteine (NAC) • N-Acetylserotonin (NAS) • Oleocanthal • Oleuropein • Rasagiline • Resveratrol • Selegiline • Selenium • Tocopherols (Vitamin E) • Tocotrienols (Vitamin E) • Tyrosol • Ubiquinone (Coenzyme Q) • Uric AcidEndocrine system: hormones (Peptide hormones · Steroid hormones) Endocrine
glandsTestis: testosterone · AMH · inhibin
Ovary: estradiol · progesterone · activin and inhibin · relaxin (pregnancy)
Placenta: hCG · HPL · estrogen · progesteroneIslet-Acinar
AxisPancreas: glucagon · insulin · amylin · somatostatin · pancreatic polypeptide
Pineal gland: melatoninNon-end.
glandsThymus: Thymosin (Thymosin α1, Thymosin beta) · Thymopoietin · Thymulin
Digestive system: Stomach: gastrin · ghrelin · Duodenum: CCK · GIP · secretin · motilin · VIP · Ileum: enteroglucagon · peptide YY · Liver/other: Insulin-like growth factor (IGF-1, IGF-2)
Adipose tissue: leptin · adiponectin · resistin
Kidney: JGA (renin) · peritubular cells (EPO) · calcitriol · prostaglandin
Heart: Natriuretic peptide (ANP, BNP)Hypnotics/Sedatives (N05C) GABAA Agonists/PAMs Barbiturates: Allobarbital • Amobarbital • Aprobarbital • Barbital • Butabarbital • Butobarbital • Cyclobarbital • Ethallobarbital • Heptabarbital • Hexobarbital • Mephobarbital • Methohexital • Pentobarbital • Phenobarbital • Proxibarbal • Reposal • Secobarbital • Talbutal • Thiamylal • Thiopental • Vinbarbital • Vinylbital; Benzodiazepines: Brotizolam • Cinolazepam • Climazolam • Doxefazepam • Estazolam • Flunitrazepam • Flurazepam • Flutoprazepam • Haloxazolam • Loprazolam • Lormetazepam • Midazolam • Nimetazepam • Nitrazepam • Quazepam • Temazepam • Triazolam; Carbamates: Carisoprodol • Ethinamate • Hexapropymate • Meprobamate • Methocarbamol • Procymate • Tybamate; Neuroactive Steroids: Acebrochol • Allopregnanolone • Alphadolone • Alphaxolone • Eltanolone • Ganaxolone • Hydroxydione • Minaxolone • Org 20599 • Org 21465 • Tetrahydrodeoxycorticosterone; Nonbenzodiazepines: CL-218,872 • Eszopiclone • Indiplon • JM-1232 • Lirequinil • Necopidem • Pazinaclone • ROD-188 • Saripidem • Suproclone • Suriclone • SX-3228 • U-89843A • U-90042 • Zaleplon • Zolpidem • Zopiclone; Phenols: Fospropofol • Propofol; Piperidinediones: Glutethimide • Methyprylon • Pyrithyldione • Piperidione; Quinazolinones: Afloqualone • Cloroqualone • Diproqualone • Etaqualone • Mebroqualone • Mecloqualone • Methaqualone • Methylmethaqualone • Nitromethaqualone; Others: 2-Methyl-2-butanol • Acetophenone • Acetylglycinamide chloral hydrate • Bromide (Lithium bromide, Potassium bromide, Sodium bromide) • Centalun • Chloral hydrate • Chloralose • Chloralodol • Clomethiazole • Dichloralphenazone • Ethanol (Alcohol) • Ethchlorvynol • Etomidate • Gaboxadol • Loreclezole • Methylpentynol • Metomidate • Paraldehyde • Petrichloral • Sulfonmethane • Trichloroethanol • Triclofos • Valerenic acid (Valerian)GABAB Agonists H1 Inverse agonists Antihistamines: Captodiame • Cyproheptadine • Dimenhydrinate • Diphenhydramine • Doxylamine • Hydroxyzine • Methapyrilene • Pheniramine • Promethazine • Propiomazine; Others: Tricyclic antidepressants (Amitriptyline, Doxepin, Trimipramine, etc.) • Tetracyclic antidepressants (Mianserin, Mirtazapine, etc.) • Typical antipsychotics (Chlorpromazine, Thioridazine, etc.) • Atypical antipsychotics (Olanzapine, Quetiapine, Risperidone, etc.)α1-Adrenergic Antagonists Mianserin • Niaprazine • Trazodone; Others: Tricyclic antidepressants (Amitriptyline, Doxepin, Trimipramine, etc.) • Typical antipsychotics (Chlorpromazine, Thioridazine, etc.) • Atypical antipsychotics (Olanzapine, Quetiapine, Risperidone, etc.)α2-Adrenergic Agonists 4-NEMD • Clonidine • Detomidine • Dexmedetomidine • Lofexidine • Medetomidine • Romifidine • Tizanidine • Xylazine5-HT2A Antagonists Eplivanserin • Niaprazine • Pruvanserin • Trazodone • Volinanserin; Others: Tricyclic antidepressants (Amitriptyline, Doxepin, Trimipramine, etc.) • Tetracyclic antidepressants (Mianserin, Mirtazapine, etc.) • Typical antipsychotics (Chlorpromazine, Thioridazine, etc.) • Atypical antipsychotics (Olanzapine, Quetiapine, Risperidone, etc.)Melatonin Agonists Orexin Antagonists Others Acecarbromal • Apronal • Bromisoval • Cannabidiol (Cannabis) • Carbromal • Embutramide • Evoxine • Fenadiazole • Gabapentin • Kavalactones (Kava) • Mephenoxalone • Opiates/Opioids (Hydrocodone, Morphine (Opium), etc.) • Passion flower • Scopolamine (Mandrake) • ValnoctamideAntidepressants (N06A) Specific reuptake inhibitors (RIs), enhancers (REs), and releasing agents (RAs) Alaproclate • Citalopram • Escitalopram • Femoxetine • Fluoxetine# • Fluvoxamine • Indalpine • Ifoxetine • Litoxetine • Lubazodone • Panuramine • Paroxetine • Pirandamine • Seproxetine • Sertraline# • Vilazodone • Zimelidine‡Bicifadine • Clovoxamine • Desvenlafaxine • Duloxetine • Levomilnacipran • Eclanamine • Milnacipran • Sibutramine • VenlafaxineSerotonin–norepinephrine–dopamine reuptake inhibitors (SNDRIs)Brasofensine • BTS-74,398 • Cocaine • Diclofensine • DOV-21,947 • DOV-102,677 • DOV-216,303 • EXP-561 • Fezolamine • JNJ-7925476 • NS-2359 • PRC200-SS • Pridefine • SEP-225,289 • SEP-227,162 • TesofensineAmedalin • Atomoxetine/Tomoxetine • Binedaline • Ciclazindol • Daledalin • Esreboxetine • Lortalamine • Mazindol • Nisoxetine • Reboxetine • Talopram • Talsupram • Tandamine • ViloxazineDopamine reuptake inhibitors (DRIs)Amineptine • Bupropion/Amfebutamone# • Cilobamine • Manifaxine • Methylphenidate • Nomifensine • Radafaxine • TametralineNorepinephrine-dopamine releasing agents (NDRAs)Serotonin-norepinephrine-dopamine releasing agents (SNDRAs)OthersReceptor antagonists and/or reuptake inhibitors Serotonin antagonists and reuptake inhibitors (SARIs)Serotonin modulators and stimulators (SMSs)VortioxetineTricyclic and tetracyclic antidepressants (TCAs/TeCAs) TricyclicsAmezepine • Amineptine • Amitriptyline# • Amitriptylinoxide • Azepindole • Butriptyline • Cianopramine • Clomipramine • Cotriptyline • Cyanodothiepin • Demexiptiline • Depramine/Balipramine • Desipramine • Dibenzepin • Dimetacrine • Dosulepin/Dothiepin • Doxepin • Enprazepine • Fluotracen • Hepzidine • Homopipramol • Imipramine • Imipraminoxide • Intriptyline • Iprindole • Ketipramine • Litracen • Lofepramine • Losindole • Mariptiline • Melitracen • Metapramine • Mezepine • Naranol • Nitroxazepine • Nortriptyline • Noxiptiline • Octriptyline • Opipramol • Pipofezine • Propizepine • Protriptyline • Quinupramine • Tampramine • Tianeptine • Tienopramine • Trimipramine;7-OH-Amoxapine • Amoxapine • Aptazapine • Azipramine • Ciclazindol • Ciclopramine • Esmirtazapine • Loxapine • Maprotiline • Mazindol • Mianserin • Mirtazapine • Oxaprotiline • Setiptiline/TeciptilineMonoamine oxidase inhibitors (MAOIs) NonselectiveIrreversible: Benmoxin • Echinopsidine • Iproclozide • Iproniazid • Isocarboxazid • Mebanazine • Metfendrazine • Nialamide • Octamoxin • Phenelzine • Pheniprazine • Phenoxypropazine • Pivalylbenzhydrazine • Safrazine • Tranylcypromine; Reversible: Caroxazone • Paraxazone;MAOA-SelectiveIrreversible: Clorgiline; Reversible: Amiflamine • Bazinaprine • Befloxatone • Befol • Brofaromine • Cimoxatone • Esuperone • Harmala Alkaloids (Harmine, Harmaline, Tetrahydroharmine, Harman, Norharman, etc) • Methylene Blue • Metralindole • Minaprine • Moclobemide • Pirlindole • Sercloremine • Tetrindole • Toloxatone • Tyrima;MAOB-SelectiveIrreversible: Ladostigil • Mofegiline • Pargyline • Rasagiline • Selegiline; Reversible: Lazabemide • MilacemideAzapirones and other 5-HT1A receptor agonists Alnespirone • Aripiprazole • Befiradol • Buspirone • Eptapirone • Flesinoxan • Flibanserin • Gepirone • Ipsapirone • Oxaflozane • Tandospirone • Vilazodone • ZalospironeDrugs from TiHKAL AL-LAD • DBT • DET • DiPT • 5-MeO-α-MT • DMT • 2,α-DMT • α,N-DMT • DPT • EiPT • α-ET • ETH-LAD • Harmaline • Harmine • 4-HO-DBT • 4-HO-DET • 4-HO-DiPT • 4-HO-DMT • 5-HO-DMT • 4-HO-DPT • 4-HO-MET • 4-HO-MiPT • 4-HO-MPT • 4-HO-pyr-T • Ibogaine • LSD • MBT • 4,5-MDO-DiPT • 5,6-MDO-DiPT • 4,5-MDO-DMT • 5,6-MDO-DMT • 5,6-MDO-MiPT • 2-Me-DET • 2-Me-DMT • Melatonin • 5-MeO-DET • 5-MeO-DiPT • 5-MeO-DMT • 4-MeO-MiPT • 5-MeO-MiPT • 5,6-MeO-MiPT • 5-MeO-NMT • 5-MeO-pyr-T • 6-MeO-THH • 5-MeO-TMT • 5-MeS-DMT • MiPT • α-MT • NET • NMT • PRO-LAD • pyr-T • Tryptamine • Tetrahydroharmine • α,N,O-TMS
Tryptamines 1-(2-Dimethylaminoethyl)dihydropyrano(3,2-e)indole • 2-Methyl-5-HT • 4-Acetoxy-DET • 4-Acetoxy-DIPT • 4-Acetoxy-DMT • 4-HO-αMT • 4-HO-DIPT • 4-HO-MET • 4-MeO-DMT • 4-Methyl-αET • 4-Methyl-αMT • 5-Benzyloxytryptamine • 5-Bromo-DMT • 5-Carboxamidotryptamine • 5-Ethoxy-αMT • 5-Fluoro-αMT • 5-HO-αMT • 5-HTP • 5-Ethoxy-DMT • 5-Ethyl-DMT • 5-Fluoro-DMT • 5-Methyl-DMT • 5-Methoxytryptamine • 5-MeO-7,N,N-TMT • 5-Methyl-αET • 5-MeO-αET • 5-MeO-αMT • 5-MeO-DALT • 5-MeO-DET • 5-MeO-DIPT • 5-MeO-DMT • 5-MeO-DPT • 5-MeO-MALT • 5-MeO-MIPT • 5,7-Dihydroxytryptamine • 5-(Nonyloxy)tryptamine • 6-Fluoro-αMT • 7-Methyl-αET • 7-Methyl-DMT • αET • αMT • Aeruginascin • AL-37350A • BW-723C86 • Baeocystin • Bufotenidine • Bufotenin • DALT • Desformylflustrabromine • DET • DiPT • DMT • DPT • Ethocybin • EiPT • EMDT • EPT • Ethocin • FGIN-127 • FGIN-143 • Ibogaine • Indorenate • Iprocin • MET • MiPT • Miprocin • Melatonin • MS-245 • NAS • NMT • Norbaeocystin • Normelatonin • Oxypertine • PiPT • Psilocin • Psilocybin • Rizatriptan • Serotonin • Sumatriptan • Tryptamine • Tryptophan • Yohimbine • Yuremamine • Zolmitriptan
Categories:- Antioxidants
- Circadian rhythms
- Hormones of the pineal gland
- Natural tryptamine alkaloids
- Treatment of bipolar disorder
- Acetamides
- Phenol ethers
- Drugs acting on the nervous system
Wikimedia Foundation. 2010.

