- Methocarbamol
-
Methocarbamol 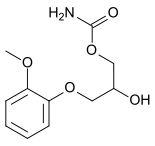
Systematic (IUPAC) name 2-hydroxy-3-(2-methoxyphenoxy)propyl carbamate Clinical data AHFS/Drugs.com monograph MedlinePlus a682579 Pregnancy cat. C Legal status ℞-only (US) OTC(Canada) Routes Oral, intravenous Pharmacokinetic data Metabolism Hepatic Half-life 1.14–1.24 hours[1] Identifiers CAS number 532-03-6 
ATC code M03BA03 PubChem CID 4107 DrugBank APRD01102 ChemSpider 3964 
UNII 125OD7737X 
KEGG D00402 
ChEMBL CHEMBL1201117 
Chemical data Formula C11H15NO5 Mol. mass 241.241 g/mol SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Methocarbamol (trade name Robaxin, which is marketed by Actient Pharmaceuticals LLC in the United States and Pfizer in Canada) is a central muscle relaxant used to treat skeletal muscle spasms. It is the carbamate of guaifenesin, but does not produce guaifenesin as a metabolite, since the carbamate bond is not hydrolyzed metabolically; metabolism is by Phase I ring hydroxylation and O-demethylation, followed by Phase II conjugation. All the major metabolites are unhydrolyzed carbamates.[2][3]
Methocarbamol is marketed under different names when presented in combination with other active ingredients. In combination with acetaminophen (Paracetamol), its trade name is Robaxacet, whereas Robax Platinum is the trade name for a formulation of methocarbamol and ibuprofen.[4] A combination of methocarbamol and aspirin is marketed as Robaxisal. Unlike other carbamates such as carisoprodol and meprobamate, Methocarbamol has greatly reduced abuse potential. Studies comparing it to lorazepam (Ativan) and diphenhydramine (Benadryl), along with placebo, find that methocarbamol produces increased 'liking' responses and some sedative-like effects, however, at higher doses dysphoria is reported. It is considered to have an abuse profile similar to, but weaker than, lorazepam. [5]
Potential side-effects include: drowsiness, dizziness, upset stomach, flushing, blurred vision, and fever. Serious side-effects include the development of a severe skin rash or itching, slow heart rate, fainting, jaundice, persistent nausea/vomiting, stomach/abdominal pain, mental/mood changes, clumsiness, trouble urinating, signs of infection. [6] In addition, methocarbamol may cause urine to turn black, blue, or green. However, this effect is harmless.[7]
Because of potential for side-effects, this drug is on the list for High-Risk Medications in the elderly. (See NCQA’s HEDIS Measure: Use of High Risk Medications in the Elderly, http://www.ncqa.org/Portals/0/Newsroom/SOHC/Drugs_Avoided_Elderly.pdf).
Chemistry
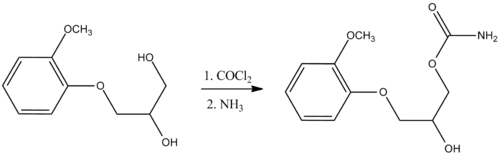
Methocarbamol, 3-(2-methoxyphenoxy)-1,2-propanediol-1 carbamate, is synthesized by successive reaction with phosgene and then ammonia into 3-(2-methoxyphenoxy)propane-1,2-diol.
- R.S. Murphey, U.S. Patent 2,770,649 (1956).
- Yale, H. L.; Pribyl, E. J.; Braker, W.; Bergeim, F. H.; Lott, W. A. (1950). Journal of the American Chemical Society 72 (8): 3710. doi:10.1021/ja01164a107.
References
- ^ Sica DA, Comstock TJ, Davis J, Manning L, Powell R, Melikian A, Wright G. (1990). "Pharmacokinetics and protein binding of methocarbamol in renal insufficiency and normals". European Journal of Clinical Pharmacology 39 (2): 193–4. doi:10.1007/BF00280060. PMID 2253675.
- ^ Methocarbamol. In: DRUGDEX System [intranet database]. Greenwood Village, Colorado: Thomson Healthcare; c1974–2009 [cited 2009 Feb 10].
- ^ Bruce RB, Turnbull LB, Newman JH. (1971 Jan). "Metabolism of methocarbamol in the rat, dog, and human". J Pharm Sci 60 (1): 104–106. doi:10.1002/jps.2600600120. PMID 5548215.
- ^ "New Drugs and Indications Reviewed at the May 2003 DEC Meeting" (PDF). ESI Canada. http://www.esi-canada.com/aboutus/news/health_newsflashes/volume5_issue9_newdrugsmay2003.pdf. Retrieved 2008-11-14.
- ^ "Subjective and behavioral effects of diphenhydramine, lorazepam and methocarbamol: evaluation of abuse liability". Journal of Pharmacology and Experimental Therapeutics. http://www.ncbi.nlm.nih.gov/pubmed/1501118. Retrieved 2011-05-06.
- ^ METHOCARBAMOL – ORAL (Robaxin) side effects, medical uses, and drug interactions. Medicinenet.com. Retrieved on 2011-11-09.
- ^ Methocarbamol: MedlinePlus Drug Information. Nlm.nih.gov. Retrieved on 2011-11-09.
Skeletal muscle relaxants (M03) Peripherally acting
(primarily antinicotinic,
NMJ block)Curare alkaloidsultra-short duration: Gantacurium
short duration: Mivacurium • Chandonium
intermediate duration: Atracurium • Cisatracurium • Fazadinium • Rocuronium • Vecuronium
long duration: Doxacurium • Dimethyltubocurarine • Pancuronium • Pipecuronium • Laudexium • Gallamine
unsorted: Hexafluronium (Hexafluorenium)Choline derivatives: Suxamethonium (Succinylcholine)
Polyalkylene derivatives: HexamethoniumCentrally acting Carbamic acid estersBenzodiazepinesAnticholinergics (Antimuscarinics)OtherBaclofen • Chlormezanone • Chlorphenesin • Chlorzoxazone • Donepezil • Eperisone • Flopropione • Mephenesin • Mephenoxalone • Metaxalone • Phenyramidol • Pridinol • Promoxolane • Quinine • Thiocolchicoside • Tizanidine • Tolperisone • TrazodoneDirectly acting Categories:- Muscle relaxants
- Veterinary drugs
- Carbamates
- Phenol ethers
- Alcohols
Wikimedia Foundation. 2010.