- Donepezil
-
Donepezil 
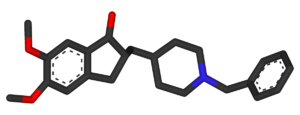
Systematic (IUPAC) name (RS)-2-[(1-benzyl-4-piperidyl)methyl]- 5,6-dimethoxy-2,3-dihydroinden-1-one Clinical data Trade names Aricept AHFS/Drugs.com monograph MedlinePlus a697032 Pregnancy cat. C Legal status ℞ Prescription only Routes Oral tablet, 5,10 & 23mg Pharmacokinetic data Bioavailability 100 (%) Protein binding 96% Half-life 70 hours Excretion 0,11-0,13 (l/h/kg) Identifiers CAS number 120014-06-4 
ATC code N06DA02 PubChem CID 3152 DrugBank APRD00039 ChemSpider 3040 
UNII 8SSC91326P 
KEGG D07869 
ChEBI CHEBI:53289 
ChEMBL CHEMBL502 
Chemical data Formula C24H29NO3 Mol. mass 379.492 g/mol SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Donepezil, marketed under the trade name Aricept by its developer Eisai and partner Pfizer, is a centrally acting reversible acetylcholinesterase inhibitor.[1] Its main therapeutic use is in the palliative treatment of mild to moderate Alzheimer's disease.[2] Common side effects include gastrointestinal upset. It has an oral bioavailability of 100% and easily crosses the blood-brain barrier. Because it has a half-life of about 70 hours, it can be taken once a day.
Contents
Medical uses
Alzheimer's disease
Currently, there is no definitive proof that use of donepezil or other similar agents alters the course or progression of Alzheimer's disease. However, 6-12 month controlled studies have shown modest benefits in cognition and/or behavior.[3] Pilot studies have reported that donepezil therapy may potentially have effects on markers of disease progression, such as hippocampal volume. Therefore, many neurologists, psychiatrists, and primary care physicians use donepezil in patients with Alzheimer's disease. In 2005, the UK National Institute for Clinical Excellence (NICE) withdrew its recommendation for use of the drug for mild-to-moderate AD, on the basis that there is no significant improvement in functional outcome, of quality of life, or of behavioral symptoms. However, NICE revised its guidelines to suggest that donepezil be used in moderate stage patients for whom the evidence is strongest. It is currently not licensed for use in the treatment of Alzheimer's disease in the UK at any other stage.[4]
While the drug is currently indicated for mild to moderate Alzheimer's, there is also evidence from 2 clinical trials that indicates it may be effective for moderate to severe disease. An example of this is a Karolinska Institute paper published in The Lancet in early 2006, which states that donepezil improves cognitive function even in patients with severe Alzheimer's disease symptoms.[5]
Dosage
Mild to Moderate Alzheimer’s Disease: Starter dose of 5mg once daily. In a minimum of 4 to 6 weeks, increase to 10mg is recommended. The usual dose is 5 to 10 mg once daily.
Moderate to Severe Alzheimer’s Disease: Starter dose of 5mg once daily. In a minimum of 4 to 6 weeks, increase to 10mg is recommended. In a minimum of 3 months on 10mg once daily, a patient may receive a dose of 23mg once daily.
Dementia: 5-10mg once daily.
Maximum daily dose: 23mg once daily. [6]
Contraindications
Donepezil (Aricept) should be used with caution in patient with; Cardiac Disease, Cardiac Conduction Disturbances, Chronic Obstructive Pulmonary Disease (COPD), Asthma, Severe Cardiac Arrhythmias and Sick Sinus Syndrome. Patient with gastrointestinal disorders should use caution because nausea/vomiting may occur. Nausea and vomiting symptoms may appear more frequent when initiating treatment or increasing the donepezil dose. Although occurrence of seizures is rare, patient who have a predisposition to seizures should be treated with caution. [7]
Other
Donepezil has been tested in other cognitive disorders (and is sometimes used off label) including Lewy body dementia[8] and vascular dementia,[9] but it is not currently approved for these indications. Donepezil has also been found to improve sleep apnea in Alzheimer's patients.[10]
Donepezil has also been studied in patients with mild cognitive impairment, schizophrenia, attention deficit disorder, post-coronary bypass cognitive impairment, cognitive impairment associated with multiple sclerosis, CADASIL syndrome, and Down syndrome. A 3 year National Institutes of Health (NIH) trial in patients with mild cognitive impairment reported that donepezil was superior to placebo in delaying rate of progression to dementia during the initial 18 months of the study but this was not sustained at 36 months. In a secondary analysis, a subgroup of individuals with the apolipoprotein E4 genotype showed sustained benefits with donepezil throughout the study(Citation? Petersen 2005). However at this time donepezil is not indicated for prevention of dementia.
Recent studies suggest that donepezil can improve speech in children with autism. The studies found that speech of autistic children who were mild to moderately affected appeared to improve from the use of donepezil.[11][12]
Adverse effects
Common side effects include bradycardia, nausea, diarrhea, anorexia, abdominal pain, and vivid dreams. In 2006, Eisai, the manufacturer issued a statement that a single vascular dementia study found a difference in the percent of study participants who died in the donepezil group (1.7%) versus the placebo group (0%) and that this could be due to an unusually low death rate on the placebo group. An analysis of all three vascular dementia trials, according to Eisai, "shows no statistically significant differences in observed mortality rates between the donepezil and placebo groups". A physician has reported several cases of mania.[13]
Development and marketing
Research leading to the development of donepezil began in 1983 at Eisai and the first Phase I clinical trial took place in 1989.[14] In 1996, Eisai received approval from the United States Food and Drug Administration (USFDA) for donepezil under the brand Aricept, which it co-marketed with Pfizer. As of 2011, Aricept was the world's best-selling Alzheimer's treatment.[15] The first generic donepezil became available in November 2010 with the USFDA approval of a formulation prepared by Ranbaxy Labs.[16] In April 2011 a second generic formulation, from Wockhardt, received tentative USFDA marketing approval.[17]
Combinations
References
- ^ Birks J, Harvey RJ (2006). Birks, Jacqueline. ed. "Donepezil for dementia due to Alzheimer's disease". Cochrane Database Syst Rev (1): CD001190. doi:10.1002/14651858.CD001190.pub2. PMID 16437430.
- ^ "aricept". The American Society of Health-System Pharmacists. http://www.drugs.com/monograph/aricept.html. Retrieved 3 April 2011.
- ^ Steele LS, Glazier RH (April 1999). "Is donepezil effective for treating Alzheimer's disease?". Can Fam Physician 45: 917–9. PMC 2328349. PMID 10216789. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2328349.
- ^ Xiong G, Doraiswamy PM (2005). "Combination drug therapy for Alzheimer's disease: what is evidence-based, and what is not?". Geriatrics 60 (6): 22–6. PMID 15948662.
- ^ "Drug 'treats severe Alzheimer's'". BBC News. 2006-03-23. http://news.bbc.co.uk/2/hi/health/4832574.stm. Retrieved 2007-07-24.
- ^ Aricept (donepezil hydrochloride) package insert. Woodcliff Lake, NJ: Eisai Co., Ltd.; 2010 Nov.
- ^ Aricept (donepezil hydrochloride) package insert. Woodcliff Lake, NJ: Eisai Co., Ltd.; 2010 Nov.
- ^ Rojas-Fernandez CH (February 2001). "Successful use of donepezil for the treatment of dementia with Lewy bodies". Ann Pharmacother 35 (2): 202–5. doi:10.1345/aph.10192. PMID 11215841. http://www.theannals.com/cgi/pmidlookup?view=long&pmid=11215841.
- ^ Malouf R, Birks J (2004). Malouf, Reem. ed. "Donepezil for vascular cognitive impairment". Cochrane Database Syst Rev (1): CD004395. doi:10.1002/14651858.CD004395.pub2. PMID 14974068.
- ^ Moraes W, Poyares D, Sukys-Claudino L, Guilleminault C, Tufik S (March 2008). "Donepezil improves obstructive sleep apnea in Alzheimer disease: a double-blind, placebo-controlled study". Chest 133 (3): 677–83. doi:10.1378/chest.07-1446. PMID 18198262. http://www.chestjournal.org/cgi/pmidlookup?view=long&pmid=18198262.
- ^ "Alzheimer's Drug Shows Promise As Treatment for Autism -- Arehart-Treichel". Psychiatric News (pn.psychiatryonline.org). 2001-11-16. http://pn.psychiatryonline.org/cgi/content/full/36/22/16-a. Retrieved 2009-08-18.
- ^ Donepezil hydrochloride: a double-blind study in autistic children
- ^ Benazzi F (November 1999). "Mania associated with donepezil". J Psychiatry Neurosci 24 (5): 468–9. PMC 1189062. PMID 10586539. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1189062.
- ^ Sugimoto, Hachiro; Ogura, Hiroo; Arai, Yasuo; Iimura, Youichi; Yamanishi, Yoshiharu (25 January 2002), "Research and Development of Donepezil Hydrochloride, a New Type of Acetylcholinesterase Inhibitor", The Japanese Journal of Pharmacology 89 (1): 7–20, 2002, doi:10.1254/jjp.89.7, http://www.jstage.jst.go.jp/article/jjp/89/1/7/_pdf, retrieved 25 April 2011
- ^ Kanoko Matsuyama (25 April 2011). "Eisai Aricept Patch for Alzheimer’s Isn’t Ready for Approval". Bloomberg. http://www.bloomberg.com/news/2011-04-25/eisai-says-fda-indicates-aricept-nda-not-ready-for-approval.html. Retrieved 25 April 2011.
- ^ "Ranbaxy gets FDA nod for Alzheimer's drug". The Indian Express (New Delhi, India: Indian Express Group): IndianExpress.com. 30 November 2010. http://www.indianexpress.com/news/ranbaxy-gets-fda-nod-for-alzheimers-drug/718059/. Retrieved 25 April 2011.
- ^ Staff Writer (25 April 2011). "Wockhardt Obtains US FDA Nod For Generic Version Of Aricept Tablets". RTTNews. http://www.rttnews.com/ArticleView.aspx?Id=1604675. Retrieved 25 April 2011.
External links
- Brenner, George D.; George M., PhD. Brenner (2000). Pharmacology. Philadelphia: W. B. Saunders. ISBN 0-7216-7757-6.
- Acting Editor-in-Chief Louise Welbanks. (2000). Compendium of Pharmaceuticals and Specialities, 2000 (25th ed.). Canadian Pharmaceutical Assn. ISBN 0-919115-76-4.
- Official Aricept product site
- Aricept entry at Drugs.com
- http://www.websciences.org/cftemplate/NAPS/archives/indiv.cfm?ID=20081395
- 3D Molecular structure of Donepezil
Cholinergics Receptor ligands Agonists: 77-LH-28-1 • AC-42 • AC-260,584 • Aceclidine • Acetylcholine • AF30 • AF150(S) • AF267B • AFDX-384 • Alvameline • AQRA-741 • Arecoline • Bethanechol • Butyrylcholine • Carbachol • CDD-0034 • CDD-0078 • CDD-0097 • CDD-0098 • CDD-0102 • Cevimeline • cis-Dioxolane • Ethoxysebacylcholine • LY-593,039 • L-689,660 • LY-2,033,298 • McNA343 • Methacholine • Milameline • Muscarine • NGX-267 • Ocvimeline • Oxotremorine • PD-151,832 • Pilocarpine • RS86 • Sabcomeline • SDZ 210-086 • Sebacylcholine • Suberylcholine • Talsaclidine • Tazomeline • Thiopilocarpine • Vedaclidine • VU-0029767 • VU-0090157 • VU-0152099 • VU-0152100 • VU-0238429 • WAY-132,983 • Xanomeline • YM-796
Antagonists: 3-Quinuclidinyl Benzilate • 4-DAMP • Aclidinium Bromide • Anisodamine • Anisodine • Atropine • Atropine Methonitrate • Benactyzine • Benzatropine (Benztropine) • Benzydamine • BIBN 99 • Biperiden • Bornaprine • CAR-226,086 • CAR-301,060 • CAR-302,196 • CAR-302,282 • CAR-302,368 • CAR-302,537 • CAR-302,668 • CS-27349 • Cyclobenzaprine • Cyclopentolate • Darifenacin • DAU-5884 • Dimethindene • Dexetimide • DIBD • Dicyclomine (Dicycloverine) • Ditran • EA-3167 • EA-3443 • EA-3580 • EA-3834 • Elemicin • Etanautine • Etybenzatropine (Ethylbenztropine) • Flavoxate • Himbacine • HL-031,120 • Ipratropium bromide • J-104,129 • Hyoscyamine • Mamba Toxin 3 • Mamba Toxin 7 • Mazaticol • Mebeverine • Methoctramine • Metixene • Myristicin • N-Ethyl-3-Piperidyl Benzilate • N-Methyl-3-Piperidyl Benzilate • Orphenadrine • Otenzepad • Oxybutynin • PBID • PD-102,807 • PD-0298029 • Phenglutarimide • Phenyltoloxamine • Pirenzepine • Piroheptine • Procyclidine • Profenamine • RU-47,213 • SCH-57,790 • SCH-72,788 • SCH-217,443 • Scopolamine (Hyoscine) • Solifenacin • Telenzepine • Tiotropium bromide • Tolterodine • Trihexyphenidyl • Tripitamine • Tropatepine • Tropicamide • WIN-2299 • Xanomeline • Zamifenacin; Others: 1st Generation Antihistamines (Brompheniramine, chlorphenamine, cyproheptadine, dimenhydrinate, diphenhydramine, doxylamine, mepyramine/pyrilamine, phenindamine, pheniramine, tripelennamine, triprolidine, etc) • Tricyclic Antidepressants (Amitriptyline, doxepin, trimipramine, etc) • Tetracyclic Antidepressants (Amoxapine, maprotiline, etc) • Typical Antipsychotics (Chlorpromazine, thioridazine, etc) • Atypical Antipsychotics (Clozapine, olanzapine, quetiapine, etc)Agonists: 5-HIAA • A-84,543 • A-366,833 • A-582,941 • A-867,744 • ABT-202 • ABT-418 • ABT-560 • ABT-894 • Acetylcholine • Altinicline • Anabasine • Anatoxin-a • AR-R17779 • Butyrylcholine • Carbachol • Cotinine • Cytisine • Decamethonium • Desformylflustrabromine • Dianicline • Dimethylphenylpiperazinium • Epibatidine • Epiboxidine • Ethanol • Ethoxysebacylcholine • EVP-4473 • EVP-6124 • Galantamine • GTS-21 • Ispronicline • Lobeline • MEM-63,908 (RG-3487) • Nicotine • NS-1738 • PHA-543,613 • PHA-709,829 • PNU-120,596 • PNU-282,987 • Pozanicline • Rivanicline • Sazetidine A • Sebacylcholine • SIB-1508Y • SIB-1553A • SSR-180,711 • Suberylcholine • TC-1698 • TC-1734 • TC-1827 • TC-2216 • TC-5214 • TC-5619 • TC-6683 • Tebanicline • Tropisetron • UB-165 • Varenicline • WAY-317,538 • XY-4083
Antagonists: 18-Methoxycoronaridine • α-Bungarotoxin • α-Conotoxin • Alcuronium • Amantadine • Anatruxonium • Atracurium • Bupropion (Amfebutamone) • Chandonium • Chlorisondamine • Cisatracurium • Coclaurine • Coronaridine • Dacuronium • Decamethonium • Dextromethorphan • Dextropropoxyphene • Dextrorphan • Diadonium • DHβE • Dimethyltubocurarine (Metocurine) • Dipyrandium • Dizocilpine (MK-801) • Doxacurium • Duador • Esketamine • Fazadinium • Gallamine • Hexafluronium • Hexamethonium (Benzohexonium) • Ibogaine • Isoflurane • Ketamine • Kynurenic acid • Laudexium (Laudolissin) • Levacetylmethadol • Malouetine • Mecamylamine • Memantine • Methadone • Methorphan (Racemethorphan) • Methyllycaconitine • Metocurine • Mivacurium • Morphanol (Racemorphanol) • Neramexane • Nitrous Oxide • Pancuronium • Pempidine • Pentamine • Pentolinium • Phencyclidine • Pipecuronium • Radafaxine • Rapacuronium • Rocuronium • Surugatoxin • Suxamethonium (Succinylcholine) • Thiocolchicoside • Toxiferine • Trimethaphan • Tropeinium • Tubocurarine • Vecuronium • XenonReuptake inhibitors PlasmalemmalCHT InhibitorsHemicholinium-3 (Hemicholine; HC3) • TriethylcholineVAChT InhibitorsEnzyme inhibitors ChAT inhibitors1-(-Benzoylethyl)pyridinium • 2-(α-Naphthoyl)ethyltrimethylammonium • 3-Chloro-4-stillbazole • 4-(1-Naphthylvinyl)pyridine • Acetylseco hemicholinium-3 • Acryloylcholine • AF64A • B115 • BETA • CM-54,903 • CatabolismAChE inhibitorsReversible: Carbamates: Aldicarb • Bendiocarb • Bufencarb • Carbaryl • Carbendazim • Carbetamide • Carbofuran • Chlorbufam • Chloropropham • Ethienocarb • Ethiofencarb • Fenobucarb • Fenoxycarb • Formetanate • Furadan • Ladostigil • Methiocarb • Methomyl • Miotine • Oxamyl • Phenmedipham • Pinmicarb • Pirimicarb • Propamocarb • Propham • Propoxur; Stigmines: Ganstigmine • Neostigmine • Phenserine • Physostigmine • Pyridostigmine • Rivastigmine; Others: Acotiamide • Ambenonium • Donepezil • Edrophonium • Galantamine • Huperzine A • Minaprine • Tacrine • Zanapezil
Irreversible: Organophosphates: Acephate • Azinphos-methyl • Bensulide • Cadusafos • Chlorethoxyfos • Chlorfenvinphos • Chlorpyrifos • Chlorpyrifos-Methyl • Coumaphos • Cyclosarin (GF) • Demeton • Demeton-S-Methyl • Diazinon • Dichlorvos • Dicrotophos • Diisopropyl fluorophosphate (Guthion) • Diisopropylphosphate • Dimethoate • Dioxathion • Disulfoton • EA-3148 • Echothiophate • Ethion • Ethoprop • Fenamiphos • Fenitrothion • Fenthion • Fosthiazate • GV • Isofluorophate • Isoxathion • Malaoxon • Malathion • Methamidophos • Methidathion • Metrifonate • Mevinphos • Monocrotophos • Naled • Novichok agent • Omethoate • Oxydemeton-Methyl • Paraoxon • Parathion • Parathion-Methyl • Phorate • Phosalone • Phosmet • Phostebupirim • Phoxim • Pirimiphos-Methyl • Sarin (GB) • Soman (GD) • Tabun (GA) • Temefos • Terbufos • Tetrachlorvinphos • Tribufos • Trichlorfon • VE • VG • VM • VR • VX; Others: Demecarium • Onchidal (Onchidella binneyi)BChE inhibitorsCymserine * Many of the acetylcholinesterase inhibitors listed above act as butyrylcholinesterase inhibitors.Others Choline (Lecithin) • Citicoline • Cyprodenate • Dimethylethanolamine (DMAE, deanol) • Glycerophosphocholine • Meclofenoxate (Centrophenoxine) • Phosphatidylcholine • Phosphatidylethanolamine • Phosphorylcholine • PirisudanolOthersAcetylcholine releasing agents: α-Latrotoxin • β-Bungarotoxin; Acetylcholine release inhibitors: Botulinum toxin (Botox); Acetylcholinesterase reactivators: Asoxime • Obidoxime • PralidoximePsychoanaleptics: Antidementia agents (N06D) Anticholinesterases Cymserine • Donepezil • Galantamine • Huperzine A (Huperzia serrata) • Ladostigil • Rivastigmine • TacrineOthers Bifemelane • Bilobalide (Ginkgo biloba) • Cerlapirdine • Ensaculin • Latrepirdine • Lecozotan • Leteprinim • Memantine • SemagacestatCategories:- Acetylcholinesterase inhibitors
- Antidementia agents
- Alzheimer's disease
- Eisai
- Pfizer
- Piperidines
- Phenol ethers
- Indanes
Wikimedia Foundation. 2010.
Look at other dictionaries:
Donépézil — Structure du donépézil Général Nom IUPAC 2 [(1 benzyl 4 piperidyl)methyl] 5,6 dimethoxy 2,3 dihydroinden 1 one … Wikipédia en Français
donepezil — /dō nepˈə zil/ noun A cholinesterase inhibitor used in the treatment of Alzheimer s disease … Useful english dictionary
Donepezil — Strukturformel Allgemeines Freiname Donepezil Andere Namen … Deutsch Wikipedia
donepezil — don·ep·e·zil .dän ep ə zil n a reversible acetylcholinesterase inhibitor used as the hydrochloride C24H29NO3·HCl in the palliative treatment of mild to moderate dementia of the type occurring in Alzheimer s disease see aricept * * * n. see… … Medical dictionary
donepezil — n.; see acetylcholinesterase inhibitor … The new mediacal dictionary
donepezil — A drug that is used to treat Alzheimer disease and is being studied in the treatment of side effects caused by radiation therapy to the brain. It is a type of cholinesterase inhibitor … English dictionary of cancer terms
Chlorhydrate de donépézil — Donépézil Donézépil Structure du donézépil Général Nom IUPAC 2 [(1 benzyl 4 piperidyl)methyl] 5,6 dimethoxy 2,3 dihy … Wikipédia en Français
donepezil hydrochloride — do·nep·e·zil hy·dro·chlo·ride (do nepґə zil) a reversible acetylcholinesterase inhibitor used for the treatment of mild to moderate symptoms of dementia of the Alzheimer type; administered orally … Medical dictionary
120014-06-4 — Donépézil Donézépil Structure du donézépil Général Nom IUPAC 2 [(1 benzyl 4 piperidyl)methyl] 5,6 dimethoxy 2,3 dihy … Wikipédia en Français
C24H29NO3 — Donépézil Donézépil Structure du donézépil Général Nom IUPAC 2 [(1 benzyl 4 piperidyl)methyl] 5,6 dimethoxy 2,3 dihy … Wikipédia en Français

