- Phosphatidylethanolamine
-
Phosphatidylethanolamine 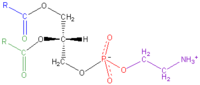 Other namescephalin
Other namescephalinIdentifiers PubChem 446872 ChemSpider 394115 
MeSH phosphatidylethanolamines Jmol-3D images Image 1 - O=C(OC[C@@H](OC(=O)CCCCCCCCCCCCCCCCCCC)COP(=O)(OCCN)O)CCCCCCCCCCCCCC
- InChI=1S/C40H80NO8P/c1-3-5-7-9-11-13-15-17-18-19-20-21-23-25-27-29-31-33-40(43)49-38(37-48-50(44,45)47-35-34-41)36-46-39(42)32-30-28-26-24-22-16-14-12-10-8-6-4-2/h38H,3-37,41H2,1-2H3,(H,44,45)/t38-/m1/s1

Key: NJGIRBISCGPRPF-KXQOOQHDSA-N
InChI=1/C40H80NO8P/c1-3-5-7-9-11-13-15-17-18-19-20-21-23-25-27-29-31-33-40(43)49-38(37-48-50(44,45)47-35-34-41)36-46-39(42)32-30-28-26-24-22-16-14-12-10-8-6-4-2/h38H,3-37,41H2,1-2H3,(H,44,45)/t38-/m1/s1
Key: NJGIRBISCGPRPF-KXQOOQHDBR
 (verify) (what is:
(verify) (what is:  /
/ ?)
?)
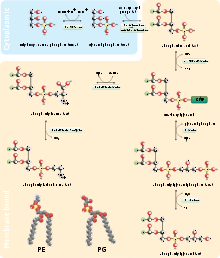
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Phosphatidylethanolamine (cephalin, sometimes abbreviated PE) is a lipid found in biological membranes. It is synthesized by the addition of CDP-ethanolamine to diglyceride, releasing CMP. S-adenosyl methionine can subsequently methylate the amine of phosphatidyl ethanolamine to yield phosphatidyl choline.
Cephalin is a phospholipid, which is a lipid derivative. It is not to be confused with the molecule of the same name that is an alkaloid constituent of Ipecac.
Contents
Function
Cephalin is found in all living cells, although in human physiology it is found particularly in nervous tissue such as the white matter of brain, nerves, neural tissue, and in spinal cord. Whereas lecithin is the principal phospholipid in animals, cephalin is the principal one in bacteria.
As a polar head group, phosphatidylethanolamine (PE) creates a more viscous lipid membrane compared to phosphatidylcholine (PC). For example, the melting temperature of di-oleoyl-PE is -16C while the melting temperature of di-oleoyl-PC is -20C. If the lipids had two palmitoyl chains, PE would melt at 63C while PC would melt already at 41C (See references in Wan et al. Biochemistry 47 2008). Lower melting temperatures correspond, in a simplistic view, to more fluid membranes.
Chemistry
In the chemical sense, cephalin is phosphatidylethanolamine. Like lecithin, it consists of a combination of glycerol esterified with two fatty acids and phosphoric acid. Whereas the phosphate group is combined with choline in Lecithin, it is combined with the ethanolamine in Cephalin.
The two fatty acids may be the same, or different, and are usually in the 1,2 positions (though can be in the 1,3 positions).
External links
See also
Additional images
References
Glycerol backbone
(Glycerophospholipids/
Phosphoglycerides)Phosphatidyl-: -ethanolamine/cephalin (PE) · -choline/lechithin (PC) · -serine (PS) · -glycerol (PG) · -inositol (PI) (glyco- (GPI))
Phosphoinositides: PIP (PI(3)P, PI(4)P, PI(5)P) · PIP2 (PI(3,4)P2, PI(3,5)P2, PI(4,5)P2) · PIP3
Ether lipids: Plasmalogen (Platelet-activating factor)Sphingosine backbone Metabolites biochemical families: prot · nucl · carb (glpr, alco, glys) · lipd (fata/i, phld, strd, gllp, eico) · amac/i · ncbs/i · ttpy/iCholinergics Receptor ligands Agonists: 77-LH-28-1 • AC-42 • AC-260,584 • Aceclidine • Acetylcholine • AF30 • AF150(S) • AF267B • AFDX-384 • Alvameline • AQRA-741 • Arecoline • Bethanechol • Butyrylcholine • Carbachol • CDD-0034 • CDD-0078 • CDD-0097 • CDD-0098 • CDD-0102 • Cevimeline • cis-Dioxolane • Ethoxysebacylcholine • LY-593,039 • L-689,660 • LY-2,033,298 • McNA343 • Methacholine • Milameline • Muscarine • NGX-267 • Ocvimeline • Oxotremorine • PD-151,832 • Pilocarpine • RS86 • Sabcomeline • SDZ 210-086 • Sebacylcholine • Suberylcholine • Talsaclidine • Tazomeline • Thiopilocarpine • Vedaclidine • VU-0029767 • VU-0090157 • VU-0152099 • VU-0152100 • VU-0238429 • WAY-132,983 • Xanomeline • YM-796
Antagonists: 3-Quinuclidinyl Benzilate • 4-DAMP • Aclidinium Bromide • Anisodamine • Anisodine • Atropine • Atropine Methonitrate • Benactyzine • Benzatropine (Benztropine) • Benzydamine • BIBN 99 • Biperiden • Bornaprine • CAR-226,086 • CAR-301,060 • CAR-302,196 • CAR-302,282 • CAR-302,368 • CAR-302,537 • CAR-302,668 • CS-27349 • Cyclobenzaprine • Cyclopentolate • Darifenacin • DAU-5884 • Dimethindene • Dexetimide • DIBD • Dicyclomine (Dicycloverine) • Ditran • EA-3167 • EA-3443 • EA-3580 • EA-3834 • Elemicin • Etanautine • Etybenzatropine (Ethylbenztropine) • Flavoxate • Himbacine • HL-031,120 • Ipratropium bromide • J-104,129 • Hyoscyamine • Mamba Toxin 3 • Mamba Toxin 7 • Mazaticol • Mebeverine • Methoctramine • Metixene • Myristicin • N-Ethyl-3-Piperidyl Benzilate • N-Methyl-3-Piperidyl Benzilate • Orphenadrine • Otenzepad • Oxybutynin • PBID • PD-102,807 • PD-0298029 • Phenglutarimide • Phenyltoloxamine • Pirenzepine • Piroheptine • Procyclidine • Profenamine • RU-47,213 • SCH-57,790 • SCH-72,788 • SCH-217,443 • Scopolamine (Hyoscine) • Solifenacin • Telenzepine • Tiotropium bromide • Tolterodine • Trihexyphenidyl • Tripitamine • Tropatepine • Tropicamide • WIN-2299 • Xanomeline • Zamifenacin; Others: 1st Generation Antihistamines (Brompheniramine, chlorphenamine, cyproheptadine, dimenhydrinate, diphenhydramine, doxylamine, mepyramine/pyrilamine, phenindamine, pheniramine, tripelennamine, triprolidine, etc) • Tricyclic Antidepressants (Amitriptyline, doxepin, trimipramine, etc) • Tetracyclic Antidepressants (Amoxapine, maprotiline, etc) • Typical Antipsychotics (Chlorpromazine, thioridazine, etc) • Atypical Antipsychotics (Clozapine, olanzapine, quetiapine, etc)Agonists: 5-HIAA • A-84,543 • A-366,833 • A-582,941 • A-867,744 • ABT-202 • ABT-418 • ABT-560 • ABT-894 • Acetylcholine • Altinicline • Anabasine • Anatoxin-a • AR-R17779 • Butyrylcholine • Carbachol • Cotinine • Cytisine • Decamethonium • Desformylflustrabromine • Dianicline • Dimethylphenylpiperazinium • Epibatidine • Epiboxidine • Ethanol • Ethoxysebacylcholine • EVP-4473 • EVP-6124 • Galantamine • GTS-21 • Ispronicline • Lobeline • MEM-63,908 (RG-3487) • Nicotine • NS-1738 • PHA-543,613 • PHA-709,829 • PNU-120,596 • PNU-282,987 • Pozanicline • Rivanicline • Sazetidine A • Sebacylcholine • SIB-1508Y • SIB-1553A • SSR-180,711 • Suberylcholine • TC-1698 • TC-1734 • TC-1827 • TC-2216 • TC-5214 • TC-5619 • TC-6683 • Tebanicline • Tropisetron • UB-165 • Varenicline • WAY-317,538 • XY-4083
Antagonists: 18-Methoxycoronaridine • α-Bungarotoxin • α-Conotoxin • Alcuronium • Amantadine • Anatruxonium • Atracurium • Bupropion (Amfebutamone) • Chandonium • Chlorisondamine • Cisatracurium • Coclaurine • Coronaridine • Dacuronium • Decamethonium • Dextromethorphan • Dextropropoxyphene • Dextrorphan • Diadonium • DHβE • Dimethyltubocurarine (Metocurine) • Dipyrandium • Dizocilpine (MK-801) • Doxacurium • Duador • Esketamine • Fazadinium • Gallamine • Hexafluronium • Hexamethonium (Benzohexonium) • Ibogaine • Isoflurane • Ketamine • Kynurenic acid • Laudexium (Laudolissin) • Levacetylmethadol • Malouetine • Mecamylamine • Memantine • Methadone • Methorphan (Racemethorphan) • Methyllycaconitine • Metocurine • Mivacurium • Morphanol (Racemorphanol) • Neramexane • Nitrous Oxide • Pancuronium • Pempidine • Pentamine • Pentolinium • Phencyclidine • Pipecuronium • Radafaxine • Rapacuronium • Rocuronium • Surugatoxin • Suxamethonium (Succinylcholine) • Thiocolchicoside • Toxiferine • Trimethaphan • Tropeinium • Tubocurarine • Vecuronium • XenonReuptake inhibitors PlasmalemmalCHT InhibitorsHemicholinium-3 (Hemicholine; HC3) • TriethylcholineVAChT InhibitorsEnzyme inhibitors ChAT inhibitors1-(-Benzoylethyl)pyridinium • 2-(α-Naphthoyl)ethyltrimethylammonium • 3-Chloro-4-stillbazole • 4-(1-Naphthylvinyl)pyridine • Acetylseco hemicholinium-3 • Acryloylcholine • AF64A • B115 • BETA • CM-54,903 • CatabolismAChE inhibitorsReversible: Carbamates: Aldicarb • Bendiocarb • Bufencarb • Carbaryl • Carbendazim • Carbetamide • Carbofuran • Chlorbufam • Chloropropham • Ethienocarb • Ethiofencarb • Fenobucarb • Fenoxycarb • Formetanate • Furadan • Ladostigil • Methiocarb • Methomyl • Miotine • Oxamyl • Phenmedipham • Pinmicarb • Pirimicarb • Propamocarb • Propham • Propoxur; Stigmines: Ganstigmine • Neostigmine • Phenserine • Physostigmine • Pyridostigmine • Rivastigmine; Others: Acotiamide • Ambenonium • Donepezil • Edrophonium • Galantamine • Huperzine A • Minaprine • Tacrine • Zanapezil
Irreversible: Organophosphates: Acephate • Azinphos-methyl • Bensulide • Cadusafos • Chlorethoxyfos • Chlorfenvinphos • Chlorpyrifos • Chlorpyrifos-Methyl • Coumaphos • Cyclosarin (GF) • Demeton • Demeton-S-Methyl • Diazinon • Dichlorvos • Dicrotophos • Diisopropyl fluorophosphate (Guthion) • Diisopropylphosphate • Dimethoate • Dioxathion • Disulfoton • EA-3148 • Echothiophate • Ethion • Ethoprop • Fenamiphos • Fenitrothion • Fenthion • Fosthiazate • GV • Isofluorophate • Isoxathion • Malaoxon • Malathion • Methamidophos • Methidathion • Metrifonate • Mevinphos • Monocrotophos • Naled • Novichok agent • Omethoate • Oxydemeton-Methyl • Paraoxon • Parathion • Parathion-Methyl • Phorate • Phosalone • Phosmet • Phostebupirim • Phoxim • Pirimiphos-Methyl • Sarin (GB) • Soman (GD) • Tabun (GA) • Temefos • Terbufos • Tetrachlorvinphos • Tribufos • Trichlorfon • VE • VG • VM • VR • VX; Others: Demecarium • Onchidal (Onchidella binneyi)BChE inhibitorsCymserine * Many of the acetylcholinesterase inhibitors listed above act as butyrylcholinesterase inhibitors.Others Choline (Lecithin) • Citicoline • Cyprodenate • Dimethylethanolamine (DMAE, deanol) • Glycerophosphocholine • Meclofenoxate (Centrophenoxine) • Phosphatidylcholine • Phosphatidylethanolamine • Phosphorylcholine • PirisudanolOthersAcetylcholine releasing agents: α-Latrotoxin • β-Bungarotoxin; Acetylcholine release inhibitors: Botulinum toxin (Botox); Acetylcholinesterase reactivators: Asoxime • Obidoxime • PralidoximeCategories:- Phospholipids
- Membrane biology
- Biochemistry stubs
Wikimedia Foundation. 2010.
Look at other dictionaries:
Phosphatidyléthanolamine — Général Synonymes Céphaline No CAS … Wikipédia en Français
Phosphatidylethanolamine — Strukturschema von Phosphatidylethanolaminen Phosphatidylethanolamine (kurz PE, auch Kephaline) zählen zu den Lipiden, genauer zu den Phosphoglyceriden. Sie sind eines der wichtigsten am Aufbau von Biomembranen beteiligten Phospholipide.… … Deutsch Wikipedia
Phosphatidylethanolamine binding protein 1 — PDB rendering based on 1a44 … Wikipedia
Phosphatidylethanolamine N-methyltransferase — protein name=phosphatidylethanolamine N methyltransferase caption= width= HGNCid=8830 Symbol=PEMT AltSymbols= EntrezGene=10400 OMIM= RefSeq=NM 007169 UniProt= PDB= ECnumber=2.1.1.17 Chromosome=17 Arm=p Band=11.2 LocusSupplementaryData=In… … Wikipedia
phosphatidylethanolamine — noun Date: 1942 any of a group of phospholipids that occur especially in blood plasma and the white matter of the central nervous system called also cephalin … New Collegiate Dictionary
phosphatidylethanolamine — noun Any of a class of phospholipid containing ethanolamine Syn: cephalin … Wiktionary
phosphatidylethanolamine — The condensation product of a phosphatidic acid and ethanolamine; found in biomembranes. SEE ALSO: cephalin. p. cytidylyltransferase a key enzyme in the biosynthesis of cephalin; it catalyzes the reaction of phosphoethanolamine and CTP to … Medical dictionary
phosphatidylethanolamine — ¦ ̷ ̷ ̷ ̷ ¦ ̷ ̷ ̷ ̷ ˌ ̷ ̷ ̷ ̷ ¦ ̷ ̷ ̷ ̷ ˌ ̷ ̷ , ̷ ̷ ¦ ̷ ̷ ̷ ̷ ̷ ̷ ˌ noun Etymology: phosphatidyl + ethanolamine : cephalin II … Useful english dictionary
N-acyl phosphatidylethanolamine-specific phospholipase D — N acyl phosphatidylethanolamine phospholipase D Identifiers Symbol NAPEPLD Entrez 222236 HUGO … Wikipedia
39382-08-6 — Phosphatidyléthanolamine Phosphatidyléthanolamine Général Synonymes Céphaline No CA … Wikipédia en Français