- Mebeverine
-
Mebeverine 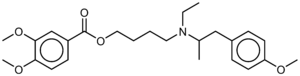
Systematic (IUPAC) name (RS)-4-(ethyl[1-(4-methoxyphenyl)propan-2-yl]amino)butyl 3,4-dimethoxybenzoate Clinical data AHFS/Drugs.com International Drug Names Pregnancy cat. ? Legal status POM (UK) Routes Oral Identifiers CAS number 2753-45-9 ATC code A03AA04 PubChem CID 4031 ChemSpider 3891 
UNII 7F80CC3NNV 
KEGG D04868 
ChEMBL CHEMBL282121 
Chemical data Formula C25H35NO5 Mol. mass 429,6[g/mol] SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Mebeverine is a musculotropic antispasmodic drug without atropic side-effects whose major therapeutic role is in the treatment of irritable bowel syndrome. It is also indicated for treatment of gastrointestinal spasm secondary to organic disorder. It was first registered in 1965. It is manufactured and marketed as Colofac, Duspatal and Duspatalin by Solvay Pharmaceuticals.
Mebeverine is presented in the United Kingdom as a (135 mg) tablet. Other strengths: 100 mg tablet and 200 mg capsule.
Contents
Mode of action
Mebeverine is an antimuscarinic. Mebeverine belongs to a group of compounds called musculotropic antispasmodics. These compounds act directly on the gut muscles at the cellular level to relax them. Mebeverine is also an inhibitor of calcium-depot replenishment. Therefore, mebeverine has dual mode of action which normalizes the small bowel motility.
Indications
Spastic functional disturbances of the colon:
- Irritable bowel syndrome in its primary form
- Irritable bowel syndrome associated with organic lesions of the gastrointestinal tract such as; diverticulosis and diverticulitis, regional enteritis, disease of the gall bladder and gall ducts, gastric and duodenal ulcers, dysentery, and aspecific or specific inflammation of the digestive tract.
- mebeverine should be taken 20 minutes before meals.
Adverse effects
Since 1978, 21 cases of severe adverse reactions to mebeverine were reported in the Netherlands.[1] Most reactions consisted of urticaria or maculopapular rash, sometimes accompanied by fever, polyarthritis, thrombopenia or angioedema.
Listed side effects
Very rarely, people taking this medicine may develop allergic reactions.[2]
References
- ^ in 't Veld BA, van Puyenbroek E, Stricker BH. (1997). Hypersensitivity reactions to use of mebeverine. pp. 1392–5. Ned Tijdschr Geneeskd. 1997 July 12;141(28):1392-5. Hypersensitivity reactions to use of mebeverine [1] pubmed: 9380201
- ^ Colofac Patient leaflet sheet http://emc.medicines.org.uk/medicine/2531/PIL/Colofac+Tablets+135mg/
Drugs for functional gastrointestinal disorders (A03) Drugs for
functional bowel disordersTertiary
amino groupOxyphencyclimine • Camylofin • Mebeverine • Trimebutine • Rociverine • Dicycloverine • Dihexyverine • Difemerine • PiperidolateQuaternary ammonium
compoundsBenzilone • Mepenzolate • Pipenzolate • Glycopyrronium • Oxyphenonium • Penthienate • Methantheline • Propantheline • Otilonium bromide • Tridihexethyl • Isopropamide • Hexocyclium • Poldine • Bevonium • Diphemanil • Tiemonium iodide • Prifinium bromide • Timepidium bromide • FenpiveriniumOtherFenpiprane • Diisopromine • Chlorbenzoxamine • Pinaverium • Fenoverine • Idanpramine • Proxazole • Alverine • Trepibutone • Isometheptene • Caroverine • Phloroglucinol • Silicones • TrimethyldiphenylpropylamineBelladonna and derivatives
(antimuscarinics)tertiary amines: Atropine • Hyoscyamine
quaternary ammonium compounds: Scopolamine (Butylscopolamine, Methylscopolamine) • Methylatropine • Fentonium • Cimetropium bromidePropulsives Cholinergics Receptor ligands Agonists: 77-LH-28-1 • AC-42 • AC-260,584 • Aceclidine • Acetylcholine • AF30 • AF150(S) • AF267B • AFDX-384 • Alvameline • AQRA-741 • Arecoline • Bethanechol • Butyrylcholine • Carbachol • CDD-0034 • CDD-0078 • CDD-0097 • CDD-0098 • CDD-0102 • Cevimeline • cis-Dioxolane • Ethoxysebacylcholine • LY-593,039 • L-689,660 • LY-2,033,298 • McNA343 • Methacholine • Milameline • Muscarine • NGX-267 • Ocvimeline • Oxotremorine • PD-151,832 • Pilocarpine • RS86 • Sabcomeline • SDZ 210-086 • Sebacylcholine • Suberylcholine • Talsaclidine • Tazomeline • Thiopilocarpine • Vedaclidine • VU-0029767 • VU-0090157 • VU-0152099 • VU-0152100 • VU-0238429 • WAY-132,983 • Xanomeline • YM-796
Antagonists: 3-Quinuclidinyl Benzilate • 4-DAMP • Aclidinium Bromide • Anisodamine • Anisodine • Atropine • Atropine Methonitrate • Benactyzine • Benzatropine (Benztropine) • Benzydamine • BIBN 99 • Biperiden • Bornaprine • CAR-226,086 • CAR-301,060 • CAR-302,196 • CAR-302,282 • CAR-302,368 • CAR-302,537 • CAR-302,668 • CS-27349 • Cyclobenzaprine • Cyclopentolate • Darifenacin • DAU-5884 • Dimethindene • Dexetimide • DIBD • Dicyclomine (Dicycloverine) • Ditran • EA-3167 • EA-3443 • EA-3580 • EA-3834 • Elemicin • Etanautine • Etybenzatropine (Ethylbenztropine) • Flavoxate • Himbacine • HL-031,120 • Ipratropium bromide • J-104,129 • Hyoscyamine • Mamba Toxin 3 • Mamba Toxin 7 • Mazaticol • Mebeverine • Methoctramine • Metixene • Myristicin • N-Ethyl-3-Piperidyl Benzilate • N-Methyl-3-Piperidyl Benzilate • Orphenadrine • Otenzepad • Oxybutynin • PBID • PD-102,807 • PD-0298029 • Phenglutarimide • Phenyltoloxamine • Pirenzepine • Piroheptine • Procyclidine • Profenamine • RU-47,213 • SCH-57,790 • SCH-72,788 • SCH-217,443 • Scopolamine (Hyoscine) • Solifenacin • Telenzepine • Tiotropium bromide • Tolterodine • Trihexyphenidyl • Tripitamine • Tropatepine • Tropicamide • WIN-2299 • Xanomeline • Zamifenacin; Others: 1st Generation Antihistamines (Brompheniramine, chlorphenamine, cyproheptadine, dimenhydrinate, diphenhydramine, doxylamine, mepyramine/pyrilamine, phenindamine, pheniramine, tripelennamine, triprolidine, etc) • Tricyclic Antidepressants (Amitriptyline, doxepin, trimipramine, etc) • Tetracyclic Antidepressants (Amoxapine, maprotiline, etc) • Typical Antipsychotics (Chlorpromazine, thioridazine, etc) • Atypical Antipsychotics (Clozapine, olanzapine, quetiapine, etc)Agonists: 5-HIAA • A-84,543 • A-366,833 • A-582,941 • A-867,744 • ABT-202 • ABT-418 • ABT-560 • ABT-894 • Acetylcholine • Altinicline • Anabasine • Anatoxin-a • AR-R17779 • Butyrylcholine • Carbachol • Cotinine • Cytisine • Decamethonium • Desformylflustrabromine • Dianicline • Dimethylphenylpiperazinium • Epibatidine • Epiboxidine • Ethanol • Ethoxysebacylcholine • EVP-4473 • EVP-6124 • Galantamine • GTS-21 • Ispronicline • Lobeline • MEM-63,908 (RG-3487) • Nicotine • NS-1738 • PHA-543,613 • PHA-709,829 • PNU-120,596 • PNU-282,987 • Pozanicline • Rivanicline • Sazetidine A • Sebacylcholine • SIB-1508Y • SIB-1553A • SSR-180,711 • Suberylcholine • TC-1698 • TC-1734 • TC-1827 • TC-2216 • TC-5214 • TC-5619 • TC-6683 • Tebanicline • Tropisetron • UB-165 • Varenicline • WAY-317,538 • XY-4083
Antagonists: 18-Methoxycoronaridine • α-Bungarotoxin • α-Conotoxin • Alcuronium • Amantadine • Anatruxonium • Atracurium • Bupropion (Amfebutamone) • Chandonium • Chlorisondamine • Cisatracurium • Coclaurine • Coronaridine • Dacuronium • Decamethonium • Dextromethorphan • Dextropropoxyphene • Dextrorphan • Diadonium • DHβE • Dimethyltubocurarine (Metocurine) • Dipyrandium • Dizocilpine (MK-801) • Doxacurium • Duador • Esketamine • Fazadinium • Gallamine • Hexafluronium • Hexamethonium (Benzohexonium) • Ibogaine • Isoflurane • Ketamine • Kynurenic acid • Laudexium (Laudolissin) • Levacetylmethadol • Malouetine • Mecamylamine • Memantine • Methadone • Methorphan (Racemethorphan) • Methyllycaconitine • Metocurine • Mivacurium • Morphanol (Racemorphanol) • Neramexane • Nitrous Oxide • Pancuronium • Pempidine • Pentamine • Pentolinium • Phencyclidine • Pipecuronium • Radafaxine • Rapacuronium • Rocuronium • Surugatoxin • Suxamethonium (Succinylcholine) • Thiocolchicoside • Toxiferine • Trimethaphan • Tropeinium • Tubocurarine • Vecuronium • XenonReuptake inhibitors PlasmalemmalCHT InhibitorsVAChT InhibitorsEnzyme inhibitors ChAT inhibitors1-(-Benzoylethyl)pyridinium • 2-(α-Naphthoyl)ethyltrimethylammonium • 3-Chloro-4-stillbazole • 4-(1-Naphthylvinyl)pyridine • Acetylseco hemicholinium-3 • Acryloylcholine • AF64A • B115 • BETA • CM-54,903 • CatabolismAChE inhibitorsReversible: Carbamates: Aldicarb • Bendiocarb • Bufencarb • Carbaryl • Carbendazim • Carbetamide • Carbofuran • Chlorbufam • Chloropropham • Ethienocarb • Ethiofencarb • Fenobucarb • Fenoxycarb • Formetanate • Furadan • Ladostigil • Methiocarb • Methomyl • Miotine • Oxamyl • Phenmedipham • Pinmicarb • Pirimicarb • Propamocarb • Propham • Propoxur; Stigmines: Ganstigmine • Neostigmine • Phenserine • Physostigmine • Pyridostigmine • Rivastigmine; Others: Acotiamide • Ambenonium • Donepezil • Edrophonium • Galantamine • Huperzine A • Minaprine • Tacrine • Zanapezil
Irreversible: Organophosphates: Acephate • Azinphos-methyl • Bensulide • Cadusafos • Chlorethoxyfos • Chlorfenvinphos • Chlorpyrifos • Chlorpyrifos-Methyl • Coumaphos • Cyclosarin (GF) • Demeton • Demeton-S-Methyl • Diazinon • Dichlorvos • Dicrotophos • Diisopropyl fluorophosphate (Guthion) • Diisopropylphosphate • Dimethoate • Dioxathion • Disulfoton • EA-3148 • Echothiophate • Ethion • Ethoprop • Fenamiphos • Fenitrothion • Fenthion • Fosthiazate • GV • Isofluorophate • Isoxathion • Malaoxon • Malathion • Methamidophos • Methidathion • Metrifonate • Mevinphos • Monocrotophos • Naled • Novichok agent • Omethoate • Oxydemeton-Methyl • Paraoxon • Parathion • Parathion-Methyl • Phorate • Phosalone • Phosmet • Phostebupirim • Phoxim • Pirimiphos-Methyl • Sarin (GB) • Soman (GD) • Tabun (GA) • Temefos • Terbufos • Tetrachlorvinphos • Tribufos • Trichlorfon • VE • VG • VM • VR • VX; Others: Demecarium • Onchidal (Onchidella binneyi)BChE inhibitorsCymserine * Many of the acetylcholinesterase inhibitors listed above act as butyrylcholinesterase inhibitors.Others Choline (Lecithin) • Citicoline • Cyprodenate • Dimethylethanolamine (DMAE, deanol) • Glycerophosphocholine • Meclofenoxate (Centrophenoxine) • Phosphatidylcholine • Phosphatidylethanolamine • Phosphorylcholine • PirisudanolOthersAcetylcholine releasing agents: α-Latrotoxin • β-Bungarotoxin; Acetylcholine release inhibitors: Botulinum toxin (Botox); Acetylcholinesterase reactivators: Asoxime • Obidoxime • PralidoximeCategories:- Motility stimulants
- Benzoates
- Phenol ethers
- Amines
Wikimedia Foundation. 2010.
Look at other dictionaries:
mebeverine — n.; see antispasmodic … The new mediacal dictionary
mebeverine hydrochloride — An intestinal antispasmodic. * * * me·bev·er·ine hy·dro·chlo·ride (mə bevґər ēn) a smooth muscle relaxant, used as an antispasmodic in the treatment of irritable bowel syndrome; administered orally … Medical dictionary
Мебеверин — … Википедия
Colofac — Mebeverine (Colona) 135mg, Bought U.K. Colofac is a medicine belonging to a group of medicines known as antispasmodic agents. It usually contains 135 mg of Mebeverine Hydrochloride with the minor ingredients of lactose, potato starch,… … Wikipedia
Muscarinic antagonist — In neurochemistry, a muscarinic receptor antagonist is an agent that reduces the activity of the muscarinic acetylcholine receptor. Acetylcholine (often abbreviated ACh) is a neurotransmitter, whose receptor is a protein found in synapses and… … Wikipedia
Atropine — Systematic (IUPAC) name … Wikipedia
Hyoscyamine — Systematic (IUPAC) name (8 methyl 8 az … Wikipedia
Anticholinergic — An anticholinergic agent is a substance that blocks the neurotransmitter acetylcholine in the central and the peripheral nervous system. An example of an anticholinergic is dicycloverine, and the classic example is atropine. Anticholinergics are… … Wikipedia
Dicycloverine — Systematic (IUPAC) name 2 (diethylamino)ethyl bi(cyclohexane) 1 carboxylate Clinical data Trade names Byclomi … Wikipedia
Glycopyrrolate — Systematic (IUPAC) name 3 (2 cyclopentyl 2 hydroxy 2 phenylacetoxy) 1,1 dimethylpyrrolidinium Clinical data AHFS/Drugs.com … Wikipedia

