- Methylmethaqualone
-
Methylmethaqualone 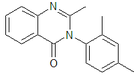
Systematic (IUPAC) name 3-(2,4-dimethylphenyl)-2-methylquinazolin-4(3H)-one Clinical data Pregnancy cat. ? Legal status ? Identifiers CAS number 3244-75-5 ATC code None PubChem CID 63382 ChemSpider 57045 
Chemical data Formula C17H16N2O Mol. mass 264.322 SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Methylmethaqualone (MMQ) is an analogue of methaqualone which has similar sedative and hypnotic properties to its parent compound, and is around the same potency. Methylmethaqualone differs from methaqualone by 4-methylation on the phenyl ring. It was made illegal in Germany in 1999 and listed by the DEA as a "drug of forensic interest" at about the same time, but little other information is available, it would appear that this compound was sold on the black market in Germany as a designer drug analogue of methaqualone.[1][2]
Animal studies of methylmethaqualone have shown it to produce convulsions at only slightly above the effective sedative dose,[3] and anecdotal reports from human users have confirmed that it can have a pro-convulsive effect, which could potentially make this compound particularly hazardous if taken in excessive doses.
References
- ^ Klein RFX, Hays PA (January–June; 2003). "Detection and Analysis of Drugs of Forensic Interest, 1992 - 2001; A Literature Review." (pdf). Microgram Journal (DEA) 1 (1–2): 60. http://www.justice.gov/dea/programs/forensicsci/microgram/journal_v1/2003_1-2.pdf.
- ^ Angelos, S. A.; Lankin, D. C.; Meyers, J. A.; Raney, J. K. (1993). "The structural identification of a methyl analog of methaqualone via 2-dimensional NMR techniques". Journal of forensic sciences 38 (2): 455–465. PMID 8455002.
- ^ Boltze, K. H.; Dell, H. D.; Lehwald, H.; Lorenz, D.; Rueberg-Schweer, M. (1963). "Substituted 4-Quinazolinone Derivatives As Hypnotics and Anticonvulsants". Arzneimittel-Forschung 13: 688–701. PMID 14085923.

This sedative-related article is a stub. You can help Wikipedia by expanding it.
