- Phenylalanine hydroxylase
-
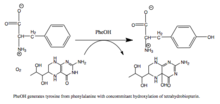
Phenylalanine hydroxylase (PheOH, alternatively PheH or PAH) (EC 1.14.16.1) is an enzyme that catalyzes the hydroxylation of the aromatic side chain of phenylalanine to generate tyrosine. PheOH is one of three members of the pterin-dependent amino acid hydroxylases, a class of monooxygenase that uses tetrahydrobiopterin (BH4, a pteridine cofactor) and a non-heme iron for catalysis. During the reaction, molecular oxygen is heterolytically cleaved with sequential incorporation of one oxygen atom into BH4 and phenylalanine substrate.[1]
Phenylalanine hydroxylase is the rate-limiting enzyme of the metabolic pathway which degrades excess phenylalanine. Research on phenylalanine hydroxylase by Seymour Kaufman led to the discovery of tetrahydrobiopterin as a biological cofactor.[2] The enzyme is also interesting from a human health perspective because mutations in PAH, the encoding gene, can lead to phenylketonuria, a severe metabolic disorder.
Contents
Enzyme Mechanism
The reaction is thought to proceed through the following steps:
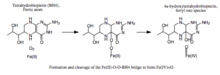
- formation of a Fe(II)-O-O-BH4 bridge.
- heterolytic cleavage of the O-O bond to yield the ferryl oxo hydroxylating intermediate Fe(IV)=O
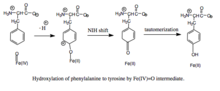
- attack on Fe(IV)=O to hydroxylate phenylalanine substrate to tyrosine.[3]
Formation and cleavage of the iron-peroxypterin bridge. Although evidence strongly supports Fe(IV)=O as the hydroxylating intermediate,[4] the mechanistic details underlying the formation of the Fe(II)-O-O-BH4 bridge prior to heterolytic cleavage remain controversial. Two pathways have been proposed based on models that differ in the proximity of the iron to the pterin cofactor and the number of water molecules assumed to be iron-coordinated during catalysis. According to one model, an iron dioxygen complex is initially formed and stabilized as a resonance hybrid of Fe2+O2 and Fe3+O2-. The activated O2 then attacks BH4, forming a transition state characterized by charge separation between the electron-deficient pterin ring and the electron-rich dioxygen species.[5] The Fe(II)-O-O-BH4 bridge is subsequently formed. On the other hand, formation of this bridge has been modeled assuming that BH4 is located in iron's first coordination shell and that the iron is not coordinated to any water molecules. This model predicts a different mechanism involving a pterin radical and superoxide as critical intermediates.[6] Once formed, the Fe(II)-O-O-BH4 bridge is broken through heterolytic cleavage of the O-O bond to Fe(IV)=O and 4a-hydroxytetrahydrobiopterin; thus, molecular oxygen is the source of both oxygen atoms used to hydroxylate the pterin ring and phenylalanine.
Hydroxylation of phenylalanine by ferryl oxo intermediate. Because the mechanism involves a Fe(IV)=O (as opposed to a peroxypterin) hydroxylating intermediate, oxidation of the BH4 cofactor and hydroxylation of phenylalanine can be decoupled, resulting in unproductive consumption of BH4 and formation of H2O2.[3] When productive, though, the Fe(IV)=O intermediate is added to phenylalanine in an electrophilic aromatic substitution reaction that reduces iron from the ferryl to the ferrous state.[3] Although initially an arene oxide or radical intermediate was proposed, analyses of the related tryptophan and tyrosine hydroxylases have suggested that the reaction instead proceeds through a cationic intermediate that requires Fe(IV)=O to be coordinated to a water ligand rather than a hydroxo group.[3][7] This cationic intermediate subsequently undergoes a 1,2-hydride NIH shift, yielding a dienone intermediate that then tautomerizes to form the tyrosine product.[8] The pterin cofactor is regenerated by hydration of the carbinolamine product of PheOH to quinonoid dihydrobiopterin (qBH2), which is then reduced to BH4.[9]
Structure
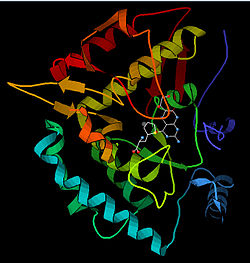
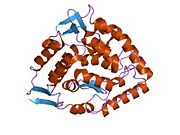
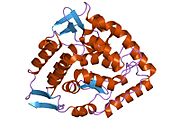
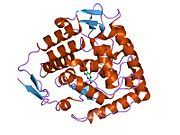
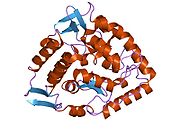
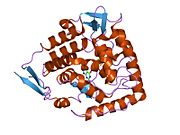
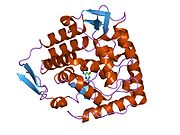
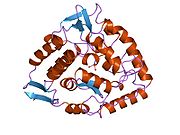
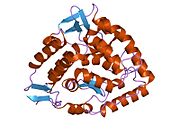
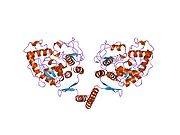
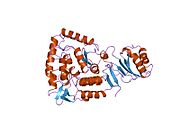
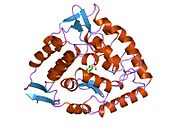
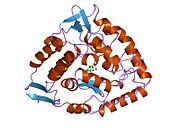
The PheOH monomer (51.9 kDa) consists of three distinct domains: a regulatory N-terminal domain (residues 1-117), the catalytic domain (residues 118-427), and a C-terminal domain (residues 428-453) responsible for oligomerization of identical monomers. Extensive crystallographic analysis has been performed, especially on the pterin- and iron-coordinated catalytic domain to examine the active site. The structure of the N-terminal regulatory domain has also been determined, and together with the solved structure of the homologous tyrosine hydroxylase C-terminal tetramerization domain, a structural model of tetrameric PheOH has been proposed.[9]
Catalytic domain
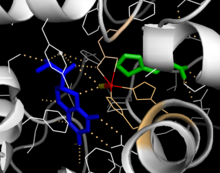
Solved crystal structures of the catalytic domain indicate that the active site consists of an open and spacious pocket lined primarily by hydrophobic residues, though three glutamic acid residues, two histidines, and a tyrosine are also present and critical for pterin- and iron-binding.[9] Contradictory evidence exists about the coordination state of the ferrous atom and its proximity to BH4 within the active site. According to crystallographic analysis, Fe(II) is coordinated by water, His285, His290 and Glu330 (a 2-his-1-carboxylate facial triad arrangement) with octahedral geometry.[10] Inclusion of a Phe analogue in the crystal structure both changes iron from a six- to a five-coordinated state involving a single water molecule and bidentate coordination to Glu330 and opening a site for oxygen to bind. BH4 is concommitantly shifted towards the iron atom, although the pterin cofactor remains in the second coordination sphere.[11] On the other hand, a competing model based on NMR and molecular modeling analyses suggests that all coordinated water molecules are forced out of the active site during the catalytic cycle while BH4 becomes directly coordinated to iron.[12] As discussed above, resolving this discrepancy will be important for determining the exact mechanism of PheOH catalysis.
N-terminal regulatory domain
The regulatory nature of the N-terminal domain (residues 1-117) is conferred by its structural flexibility.[13] Hydrogen/deuterium exchanges analysis indicates that allosteric binding of Phe globally alters the conformation of PheOH such that the active site is less occluded as the interface between the regulatory and catalytic domains is increasingly exposed to solvent.[13][14][15] This observation is consistent with kinetic studies, which show an initially low rate of tyrosine formation for full-length PheOH. This lag time is not observed, however, for a truncated PheOH lacking the N-terminal domain or if the full-length enzyme is pre-incubated with Phe. Deletion of the N-terminal domain also eliminates the lag time while increasing the affinity for Phe by nearly two-fold; no difference is observed in the Vmax or Km for the tetrahydrobiopterin cofactor.[16] Additional regulation is provided by Ser16; phosphorylation of this residue does not alter enzyme conformation but does reduce the concentration of Phe required for allosteric activation.[15] This N-terminal regulatory domain is not observed in bacterial PheOHs but shows considerable structural homology to the regulatory domain of phosphogylcerate dehydrogenase, an enzyme in the serine biosynthetic pathway.[15]
Tetramerization domain
Prokaryotic PheOH is monomeric, whereas eukaryotic PheOH exists in an equilibrium between homotetrameric and homodimeric forms.[3][9] The dimerization interface is composed of symmetry-related loops that link identical monomers, while the overlapping C-terminal tetramerization domain mediates the association of conformationally distinct dimers that are characterized by a different relative orientation of the catalytic and tetramerization domains (Flatmark, Erlandsen). The resulting distortion of the tetramer symmetry is evident in the differential surface area of the dimerization interfaces and distinguishes PheOH from the tetramerically symmetrical tyrosine hydroxylase.[9] A domain swapping mechanism has been proposed to mediate formation of the tetramer from dimers, in which C-terminal alpha-helixes mutually alter their conformation around a flexible C-terminal five-residue hinge region to form a coiled-coil structure, shifting equilibrium towards the tetrameric form.[3][9][17] Although both the homodimeric and homotetrameric forms of PheOH are catalytically active, the two exhibit differential kinetics and regulation. In addition to reduced catalytic efficiency, the dimer does not display positive cooperativity towards L-Phe (which at high concentrations activates the enzyme), suggesting that L-Phe allosterically regulates PheOH by influencing dimer-dimer interaction.[17]
Biological function
PheOH is a critical enzyme in phenylalanine metabolism and catalyzes the rate-limiting step in its complete catabolism to carbon dioxide and water.[9][18] Regulation of flux through phenylalanine-associated pathways is critical in mammalian metabolism, as evidenced by the toxicity of high plasma levels of this amino acid observed in phenylketonuria (see below.) The principal source of phenylalanine is ingested proteins but relatively little of this pool is used for protein synthesis.[18] Instead, the majority of ingested phenylalanine is catabolized through PheOH to form tyrosine; addition of the hydroxyl group allows for the benzene ring to be broken in subsequent catabolic steps. Transamination to phenylpyruvate, whose metabolites are excreted in the urine, represents another pathway of phenylalanine turnover, but catabolism through PheOH predominates.[18]
In humans, this enzyme is expressed both in the liver and the kidney, and there is some indication that it may be differentially regulated in these tissues.[19] PheOH is unusual among the aromatic amino acid hydroxylases for its involvement in catabolism; tyrosine and tryptophan hydroxylases, on the other hand, are primarily expressed in the central nervous system and catalyze rate-limiting steps in neurotransmitter/hormone biosynthesis.[9]
Disease relevance
Deficiency in PheOH activity due to mutations in the PAH gene causes hyperphenylalaninemia (HPA), and when blood phenylalanine levels increase above 20 times the normal concentration, the metabolic disease phenylketonuria (PKU) results.[18] PKU is both genotypically and phenotypically heterogeneous: over 300 distinct pathological mutants have been identified, the majority of which correspond to missense mutations that map to the catalytic domain.[9][10] When a cohort of identified PheOH mutants were expressed in recombinant systems, the enzymes displayed altered kinetic behavior and/or reduced stability, consistent with structural mapping of these mutations to both the catalytic and tetramerization domains of the enzyme.[9] Interestingly, BH44 has been administered as a pharmacological treatment and has been shown to reduce blood levels of phenylalanine for a segment of PKU patients whose genotypes lead to some residual PAH activity but have no defect in BH44 synthesis or regeneration. Follow-up studies suggest that in the case of certain PheOH mutants, excess BH44 acts as a pharmacological chaperone to stabilize mutant enzymes with disrupted tetramer assembly and increased sensitivity to proteolytic cleavage and aggregation.[20] Mutations that have been identified in the PAH locus are documented at the Phenylalanine Hydroxylase Locus Knowledgbase (PAHdb, http://www.pahdb.mcgill.ca/).
Related enzymes
Phenylalanine hydroxylase is closely related to two other enzymes:
- tryptophan hydroxylase (EC number 1.14.16.4), which controls levels of serotonin in the brain and the gastrointestinal tract
- tyrosine hydroxylase (EC number 1.14.16.2), which controls levels of dopamine, epinephrine, and norepinephrine in the brain and the adrenal medulla.
The three enzymes are homologous, that is, are thought to have evolved from the same ancient hydroxylase.
References
- ^ Fitzpatrick PF (1999). "Tetrahydropterin-dependent amino acid hydroxylases". Annu. Rev. Biochem. 68: 355–81. doi:10.1146/annurev.biochem.68.1.355. PMID 10872454.
- ^ KAUFMAN S (February 1958). "A new cofactor required for the enzymatic conversion of phenylalanine to tyrosine". J. Biol. Chem. 230 (2): 931–9. PMID 13525410.
- ^ a b c d e f Fitzpatrick PF (December 2003). "Mechanism of aromatic amino acid hydroxylation". Biochemistry 42 (48): 14083–91. doi:10.1021/bi035656u. PMC 1635487. PMID 14640675. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1635487.
- ^ Panay AJ, Lee M, Krebs C, Bollinger JM, Fitzpatrick PF (March 2011). "Evidence for a high-spin Fe(IV) species in the catalytic cycle of a bacterial phenylalanine hydroxylase". Biochemistry 50 (11): 1928–33. doi:10.1021/bi1019868. PMID 21261288.
- ^ Bassan A, Blomberg MR, Siegbahn PE (January 2003). "Mechanism of dioxygen cleavage in tetrahydrobiopterin-dependent amino acid hydroxylases". Chemistry 9 (1): 106–15. doi:10.1002/chem.200390006. PMID 12506369.
- ^ Olsson E, Martinez A, Teigen K, Jensen VR (March 2011). "Formation of the iron-oxo hydroxylating species in the catalytic cycle of aromatic amino acid hydroxylases". Chemistry 17 (13): 3746–58. doi:10.1002/chem.201002910. PMID 21351297.
- ^ Bassan A, Blomberg MR, Siegbahn PE (September 2003). "Mechanism of aromatic hydroxylation by an activated FeIV=O core in tetrahydrobiopterin-dependent hydroxylases". Chemistry 9 (17): 4055–67. doi:10.1002/chem.200304768. PMID 12953191.
- ^ Pavon JA, Fitzpatrick PF (September 2006). "Insights into the catalytic mechanisms of phenylalanine and tryptophan hydroxylase from kinetic isotope effects on aromatic hydroxylation". Biochemistry 45 (36): 11030–7. doi:10.1021/bi0607554. PMC 1945167. PMID 16953590. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1945167.
- ^ a b c d e f g h i j Flatmark T, Stevens RC (August 1999). "Structural Insight into the Aromatic Amino Acid Hydroxylases and Their Disease-Related Mutant Forms". Chem. Rev. 99 (8): 2137–2160. doi:10.1021/cr980450y. PMID 11849022.
- ^ a b Erlandsen H, Fusetti F, Martinez A, Hough E, Flatmark T, Stevens RC (December 1997). "Crystal structure of the catalytic domain of human phenylalanine hydroxylase reveals the structural basis for phenylketonuria". Nat. Struct. Biol. 4 (12): 995–1000. doi:10.1038/nsb1297-995. PMID 9406548.
- ^ Andersen OA, Flatmark T, Hough E (July 2002). "Crystal structure of the ternary complex of the catalytic domain of human phenylalanine hydroxylase with tetrahydrobiopterin and 3-(2-thienyl)-L-alanine, and its implications for the mechanism of catalysis and substrate activation". J. Mol. Biol. 320 (5): 1095–108. doi:10.1016/S0022-2836(02)00560-0. PMID 12126628.
- ^ Teigen K, Frøystein NA, Martínez A (December 1999). "The structural basis of the recognition of phenylalanine and pterin cofactors by phenylalanine hydroxylase: implications for the catalytic mechanism". J. Mol. Biol. 294 (3): 807–23. doi:10.1006/jmbi.1999.3288. PMID 10610798.
- ^ a b Li J, Dangott LJ, Fitzpatrick PF (April 2010). "Regulation of phenylalanine hydroxylase: conformational changes upon phenylalanine binding detected by hydrogen/deuterium exchange and mass spectrometry". Biochemistry 49 (15): 3327–35. doi:10.1021/bi1001294. PMC 2855537. PMID 20307070. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2855537.
- ^ Li J, Ilangovan U, Daubner SC, Hinck AP, Fitzpatrick PF (January 2011). "Direct evidence for a phenylalanine site in the regulatory domain of phenylalanine hydroxylase". Arch. Biochem. Biophys. 505 (2): 250–5. doi:10.1016/j.abb.2010.10.009. PMID 20951114.
- ^ a b c Kobe B, Jennings IG, House CM, Michell BJ, Goodwill KE, Santarsiero BD, Stevens RC, Cotton RG, Kemp BE (May 1999). "Structural basis of autoregulation of phenylalanine hydroxylase". Nat. Struct. Biol. 6 (5): 442–8. doi:10.1038/8247. PMID 10331871.
- ^ Daubner SC, Hillas PJ, Fitzpatrick PF (December 1997). "Expression and characterization of the catalytic domain of human phenylalanine hydroxylase". Arch. Biochem. Biophys. 348 (2): 295–302. doi:10.1006/abbi.1997.0435. PMID 9434741.
- ^ a b Bjørgo E, de Carvalho RM, Flatmark T (February 2001). "A comparison of kinetic and regulatory properties of the tetrameric and dimeric forms of wild-type and Thr427-->Pro mutant human phenylalanine hydroxylase: contribution of the flexible hinge region Asp425-Gln429 to the tetramerization and cooperative substrate binding". Eur. J. Biochem. 268 (4): 997–1005. doi:10.1046/j.1432-1327.2001.01958.x. PMID 11179966.
- ^ a b c d Kaufman S (March 1999). "A model of human phenylalanine metabolism in normal subjects and in phenylketonuric patients". Proc. Natl. Acad. Sci. U.S.A. 96 (6): 3160–4. doi:10.1073/pnas.96.6.3160. PMC 15912. PMID 10077654. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=15912.
- ^ Lichter-Konecki U, Hipke CM, Konecki DS (August 1999). "Human phenylalanine hydroxylase gene expression in kidney and other nonhepatic tissues". Mol. Genet. Metab. 67 (4): 308–16. doi:10.1006/mgme.1999.2880. PMID 10444341.
- ^ Muntau AC, Gersting SW (December 2010). "Phenylketonuria as a model for protein misfolding diseases and for the development of next generation orphan drugs for patients with inborn errors of metabolism". J. Inherit. Metab. Dis. 33 (6): 649–58. doi:10.1007/s10545-010-9185-4. PMID 20824346.
Further reading
- Eisensmith RC, Woo SL (1993). "Molecular basis of phenylketonuria and related hyperphenylalaninemias: mutations and polymorphisms in the human phenylalanine hydroxylase gene.". Hum. Mutat. 1 (1): 13–23. doi:10.1002/humu.1380010104. PMID 1301187.
- Konecki DS, Lichter-Konecki U (1991). "The phenylketonuria locus: current knowledge about alleles and mutations of the phenylalanine hydroxylase gene in various populations.". Hum. Genet. 87 (4): 377–88. doi:10.1007/BF00197152. PMID 1679029.
- Cotton RG (1991). "Heterogeneity of phenylketonuria at the clinical, protein and DNA levels.". J. Inherit. Metab. Dis. 13 (5): 739–50. doi:10.1007/BF01799577. PMID 2246858.
- Erlandsen H, Fusetti F, Martinez A, et al. (1998). "Crystal structure of the catalytic domain of human phenylalanine hydroxylase reveals the structural basis for phenylketonuria.". Nat. Struct. Biol. 4 (12): 995–1000. doi:10.1038/nsb1297-995. PMID 9406548.
- Waters PJ, Parniak MA, Nowacki P, Scriver CR (1998). "In vitro expression analysis of mutations in phenylalanine hydroxylase: linking genotype to phenotype and structure to function.". Hum. Mutat. 11 (1): 4–17. doi:10.1002/(SICI)1098-1004(1998)11:1<4::AID-HUMU2>3.0.CO;2-L. PMID 9450897.
- Waters PJ (2003). "How PAH gene mutations cause hyper-phenylalaninemia and why mechanism matters: insights from in vitro expression.". Hum. Mutat. 21 (4): 357–69. doi:10.1002/humu.10197. PMID 12655545.
External links
- GeneReviews/NCBI/NIH/UW entry on Phenylalanine Hydroxylase Deficiency
- PAHdb - online locus-specific mutation database of the human phenylalanine hydroxylase gene
PDB gallery 1dmw: CRYSTAL STRUCTURE OF DOUBLE TRUNCATED HUMAN PHENYLALANINE HYDROXYLASE WITH BOUND 7,8-DIHYDRO-L-BIOPTERIN1j8t: Catalytic Domain of Human Phenylalanine Hydroxylase Fe(II)1j8u: Catalytic Domain of Human Phenylalanine Hydroxylase Fe(II) in Complex with Tetrahydrobiopterin1kw0: Catalytic Domain of Human Phenylalanine Hydroxylase (Fe(II)) in Complex with Tetrahydrobiopterin and Thienylalanine1lrm: Crystal structure of binary complex of the catalytic domain of human phenylalanine hydroxylase with dihydrobiopterin (BH2)1mmk: Crystal structure of ternary complex of the catalytic domain of human phenylalanine hydroxylase ((FeII)) complexed with tetrahydrobiopterin and thienylalanine1mmt: Crystal structure of ternary complex of the catalytic domain of human phenylalanine hydroxylase (Fe(II)) complexed with tetrahydrobiopterin and norleucine1pah: HUMAN PHENYLALANINE HYDROXYLASE DIMER, RESIDUES 117-4241phz: STRUCTURE OF PHOSPHORYLATED PHENYLALANINE HYDROXYLASE1tdw: Crystal structure of double truncated human phenylalanine hydroxylase BH4-responsive PKU mutant A313T.1tg2: Crystal structure of phenylalanine hydroxylase A313T mutant with 7,8-dihydrobiopterin bound2pah: TETRAMERIC HUMAN PHENYLALANINE HYDROXYLASE2phm: STRUCTURE OF PHENYLALANINE HYDROXYLASE DEPHOSPHORYLATED3pah: HUMAN PHENYLALANINE HYDROXYLASE CATALYTIC DOMAIN DIMER WITH BOUND ADRENALINE INHIBITOR4pah: HUMAN PHENYLALANINE HYDROXYLASE CATALYTIC DOMAIN DIMER WITH BOUND NOR-ADRENALINE INHIBITOR5pah: HUMAN PHENYLALANINE HYDROXYLASE CATALYTIC DOMAIN DIMER WITH BOUND DOPAMINE INHIBITOR6pah: HUMAN PHENYLALANINE HYDROXYLASE CATALYTIC DOMAIN DIMER WITH BOUND L-DOPA (3,4-DIHYDROXYPHENYLALANINE) INHIBITOROxidoreductases: dioxygenases, including steroid hydroxylases (EC 1.14) 1.14.11: 2-oxoglutarate 1.14.13: NADH or NADPH Flavin-containing monooxygenase (FMO1, FMO2, FMO3, FMO4, FMO5) - Nitric oxide synthase (NOS1, NOS2, NOS3) - Cholesterol 7 alpha-hydroxylase - Methane monooxygenase - 3A4 - Lanosterol 14 alpha-demethylase1.14.14: reduced flavin or flavoprotein 1.14.15: reduced iron-sulfur protein 1.14.16: reduced pteridine (BH4 dependent) 1.14.17: reduced ascorbate 1.14.18-19: other 1.14.99 - miscellaneous B enzm: 1.1/2/3/4/5/6/7/8/10/11/13/14/15-18, 2.1/2/3/4/5/6/7/8, 2.7.10, 2.7.11-12, 3.1/2/3/4/5/6/7, 3.1.3.48, 3.4.21/22/23/24, 4.1/2/3/4/5/6, 5.1/2/3/4/99, 6.1-3/4/5-6 Metabolism: amino acid metabolism · synthesis and catabolism enzymes (essential in CAPS) K→acetyl-CoA (see below)G →alpha-ketoglutarate→TCAOther→succinyl-CoA→TCAG→fumaratePhenylalanine hydroxylase · Tyrosine aminotransferase · 4-Hydroxyphenylpyruvate dioxygenase · Homogentisate 1,2-dioxygenase · Fumarylacetoacetate hydrolase
tyrosine→melanin: Tyrosinaseasparagine→aspartate→Categories:- Human proteins
- EC 1.14.16
Wikimedia Foundation. 2010.