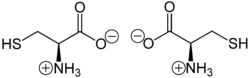
- Cysteine
-
L-Cysteine 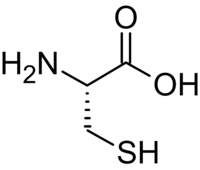
 CysteineOther names2-Amino-3-mercaptopropanoic acid
CysteineOther names2-Amino-3-mercaptopropanoic acidIdentifiers CAS number 52-90-4  ,
,
52-89-1 (hydrochloride)PubChem 5862 ChemSpider 574 (Racemic)  , 5653 (L-form)
, 5653 (L-form) 
UNII K848JZ4886 
EC-number 200-158-2 KEGG D00026 
ChEBI CHEBI:15356 
ChEMBL CHEMBL54943 
Jmol-3D images Image 1
Image 2- C([C@@H](C(=O)O)N)S
C(C(C(=O)O)N)S
Properties[1] Molecular formula C3H7NO2S Molar mass 121.16 g mol−1 Appearance white crystals or powder Melting point 240 °C decomp.
Solubility in water soluble Chiral rotation [α]D +9.4º (H2O, c = 1.3) Supplementary data page Structure and
propertiesn, εr, etc. Thermodynamic
dataPhase behaviour
Solid, liquid, gasSpectral data UV, IR, NMR, MS  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
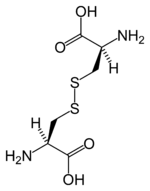
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Cysteine (abbreviated as Cys or C)[2] is an α-amino acid with the chemical formula HO2CCH(NH2)CH2SH. It is a non-essential amino acid, which means that it is biosynthesized in humans.[3] Its codons are UGU and UGC. The side chain on cysteine is thiol, which is polar and thus cysteine is usually classified as a hydrophilic amino acid.[4] The thiol side chain often participates in enzymatic reactions, serving as a nucleophile. The thiol is susceptible to oxidization to give the disulfide derivative cystine, which serves an important structural role in many proteins.
Contents
Sources
Dietary sources
Although classified as a non-essential amino acid, in rare cases, cysteine may be essential for infants, the elderly, and individuals with certain metabolic disease or who suffer from malabsorption syndromes. Cysteine can usually be synthesized by the human body under normal physiological conditions if a sufficient quantity of methionine is available. Cysteine is catabolized in the gastrointestinal tract and blood plasma[citation needed]. In contrast, cystine travels safely through the GI tract and blood plasma and is promptly reduced to the two cysteine molecules upon cell entry[citation needed].
Cysteine is found in most high-protein foods, including:
- Animal sources: pork, sausage meat, chicken, turkey, duck, luncheon meat, eggs, milk, whey protein, ricotta, cottage cheese, yogurt
- Plant sources: red peppers, garlic, onions, broccoli, brussels sprouts, oats, granola, wheat germ, lentil
As other amino acids, cysteine has an amphoteric character.
Industrial sources
The majority of L-Cysteine was once obtained industrially by hydrolysis of human hair, but in recent years 80% is produced from duck feathers. Due to marketing restraints with Jewish Kosher and Muslim Halal however, it is now possible to get synthetically produced material, albeit at a higher price.[5] The synthetic route involves fermentation utilizing a mutant of E. coli. Wacker Chemie introduced a route from substituted thiazolines.[6] Following this technology, L-cysteine is produced by the hydrolysis of racemic 2-amino-Δ2-thiazoline-4-carboxylic acid using Pseudomonas thiazolinophilum.[7]
Biosynthesis
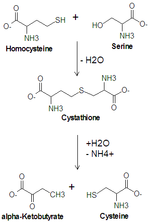 Cysteine synthesis. Cystathionine beta synthase catalyzes the upper reaction and cystathionine gamma-lyase catalyzes the lower reaction.
Cysteine synthesis. Cystathionine beta synthase catalyzes the upper reaction and cystathionine gamma-lyase catalyzes the lower reaction.
In animals, biosynthesis begins with the amino acid serine. The sulfur is derived from methionine, which is converted to homocysteine through the intermediate S-adenosylmethionine. Cystathionine beta-synthase then combines homocysteine and serine to form the asymmetrical thioether cystathionine. The enzyme cystathionine gamma-lyase converts the cystathionine into cysteine and alpha-ketobutyrate. In plants and bacteria, cysteine biosynthesis again starts from serine, which is converted to O-acetylserine by the enzyme serine transacetylase. The enzyme O-acetylserine (thiol)-lyase, using sulfide sources, converts this ester into cysteine, releasing acetate.[8]
Biological functions
The cysteine thiol group is nucleophilic and easily oxidized. The reactivity is enhanced when the thiol is ionized, and cysteine residues in proteins have pKa values close to neutrality, so are often in their reactive thiolate form in the cell.[9] Because of its high reactivity, the thiol group of cysteine has numerous biological functions.
Precursor to the antioxidant glutathione
Due to the ability of thiols to undergo redox reactions, cysteine has antioxidant properties. Cysteine's antioxidant properties are typically expressed in the tripeptide glutathione, which occurs in humans as well as other organisms. The systemic availability of oral glutathione (GSH) is negligible; so it must be biosynthesized from its constituent amino acids, cysteine, glycine, and glutamic acid. Glutamic acid and glycine are readily available in most Western diets, but the availability of cysteine can be the limiting substrate.[citation needed]
Disulfide bonds
Disulfide bonds play an important role in the folding and stability of some proteins, usually proteins secreted to the extracellular medium.[10] Since most cellular compartments are reducing environments, disulfide bonds are generally unstable in the cytosol with some exceptions as noted below.
Disulfide bonds in proteins are formed by oxidation of the thiol groups of cysteine residues. The other sulfur-containing amino acid, methionine, cannot form disulfide bonds. More aggressive oxidants convert cysteine to the corresponding sulfinic acid and sulfonic acid. Cysteine residues play a valuable role by crosslinking proteins, which increases the rigidity of proteins and also functions to confer proteolytic resistance (since protein export is a costly process, minimizing its necessity is advantageous). Inside the cell, disulfide bridges between cysteine residues within a polypeptide support the protein's tertiary structure. Insulin is an example of a protein with cystine crosslinking, wherein two separate peptide chains are connected by a pair of disulfide bonds.
Protein disulfide isomerases catalyze the proper formation of disulfide bonds; the cell transfers dehydroascorbic acid to the endoplasmic reticulum, which oxidises the environment. In this environment, cysteines are, in general, oxidized to cystine and are no longer functional as a nucleophiles.
Precursor to iron-sulfur clusters
Cysteine is an important source of sulfide in human metabolism. The sulfide in iron-sulfur clusters and in nitrogenase is extracted from cysteine, which is converted to alanine in the process.[11]
Metal ion binding
Beyond the iron-sulfur proteins, many other metal cofactors in enzymes are bound to the thiolate substituent of cysteinyl residues. Examples include zinc in zinc fingers and alcohol dehydrogenase, copper in the blue copper proteins, iron in cytochrome P450, and nickel in the [NiFe]-hydrogenases.[12] The thiol group also has a high affinity for heavy metals, so that proteins containing cysteine, such as metallothionein, will bind metals such as mercury, lead, and cadmium tightly.[13]
Post-translational modifications
Aside from its oxidation to cystine, cysteine participates in numerous posttranslational modifications. The nucleophilic thiol group allows cysteine to conjugate to other groups, e.g., in prenylation. Ubiquitin ligases transfer ubiquitin to its pendant, proteins, and caspases, which engage in proteolysis in the apoptotic cycle. Inteins often function with the help of a catalytic cysteine. These roles are typically limited to the intracellular milieu, where the environment is reducing, and cysteine is not oxidized to cystine.
Applications
Cysteine, mainly the L-enantiomer, is a precursor in the food, pharmaceutical, and personal care industries. One of the largest applications is the production of flavors. For example, the reaction of cysteine with sugars in a Maillard reaction yields meat flavors.[14] L-cysteine is also used as a processing aid for baking.[15]
In the field of personal care, cysteine is used for permanent wave applications predominantly in Asia. Again the cysteine is used for breaking up the disulfide bonds in the hair's keratin.
Cysteine is a very popular target for site-directed labeling experiments to investigate biomolecular structure and dynamics. Maleimides will selectively attach to cysteine using a covalent Michael addition. Site-directed spin labeling for EPR or paramagnetic relaxation enhanced NMR also uses cysteine extensively.
In a 1994 report released by five top cigarette companies, cysteine is one of the 599 additives to cigarettes. Like most cigarette additives, however, its use or purpose is unknown.[16] Its inclusion in cigarettes could offer two benefits: Acting as an expectorant, since smoking increases mucus production in the lungs; and increasing the beneficial antioxidant glutathione (which is diminished in smokers).
Sheep
Cysteine is required by sheep in order to produce wool: It is an essential amino acid that must be taken in as food from grass. As a consequence, during drought conditions, sheep stop producing wool; however, transgenic sheep that can make their own cysteine have been developed.[17]
Reducing toxic effects of alcohol
Cysteine has been proposed as a preventative or antidote for some of the negative effects of alcohol, including liver damage and hangover. It counteracts the poisonous effects of acetaldehyde, which is the major by-product of alcohol metabolism and is responsible for most of the negative aftereffects and long-term damage associated with alcohol use (but not the immediate effects of drunkenness). Cysteine supports the next step in metabolism, which turns acetaldehyde into the relatively harmless acetic acid. In a rat study, test animals received an LD50 dose of acetaldehyde (the amount that normally kills half of all animals). Those that received cysteine had an 80% survival rate; when both cysteine and thiamine were administered, all animals survived.[18] There is not yet direct evidence for or against its effectiveness in humans who consume alcohol at normal levels.
N-acetylcysteine (NAC)
N-acetyl-L-cysteine (NAC) is a derivative of cysteine wherein an acetyl group is attached to the nitrogen atom. This compound is sold as a dietary supplement commonly claiming antioxidant and liver-protecting effects. NAC is often used as a cough medicine because it breaks up the disulfide bonds in the mucus and thus liquefies it, making it easier to cough up. It is also this action of breaking disulfide bonds that makes it useful in thinning the abnormally thick mucus in Cystic Fibrosis patients. NAC is also used as a specific antidote in cases of acetaminophen overdose.
See also
References
- ^ Weast, Robert C., ed (1981). CRC Handbook of Chemistry and Physics (62nd ed.). Boca Raton, FL: CRC Press. p. C-259. ISBN 0-8493-0462-8..
- ^ "Nomenclature and symbolism for amino acids and peptides (IUPAC-IUB Recommendations 1983)", Pure Appl. Chem. 56 (5): 595–624, 1984, doi:10.1351/pac198456050595
- ^ "The primary structure of proteins is the amino acid sequence". The Microbial World. University of Wisconsin-Madison Bacteriology Department. http://www.microbiologytext.com/index.php?module=Book&func=displayarticle&art_id=40. Retrieved 15 August 2011.
- ^ <a href="http://www.microbiologytext.com/index.php?module=Book&func=displayarticle&art_id=40">The primary structure of proteins is the amino acid sequence</a>; The Microbial World, The University of Wisconsin-Madison Biology Department
- ^ http://www.vrg.org/nutshell/faqingredients.htm#cystine
- ^ Martens, Jürgen; Offermanns, Heribert; Scherberich, Paul (1981), "Facile Synthesis of Racemic Cysteine", Angew. Chem. Int. Ed. Engl. 20 (8): 668, doi:10.1002/anie.198106681
- ^ Karlheinz Drauz, Ian Grayson, Axel Kleemann, Hans-Peter Krimmer, Wolfgang Leuchtenberger, Christoph Weckbecker "Amino Acids"in Ullmann's Encyclopedia of Industrial Chemistry 2007, Wiley-VCH, Weinheim. doi:10.1002/14356007.a02_057.pub2
- ^ Hell, Rüdiger (1997), "Molecular physiology of plant sulfur metabolism", Planta 202 (2): 138–48, doi:10.1007/s004250050112, PMID 9202491.
- ^ Bulaj, Grzegorz; Kortemme, Tanja; Goldenberg, David P. (1998), "Ionization-reactivity relationships for cysteine thiols in polypeptides.", Biochemistry 37 (25): 8965–72, doi:10.1021/bi973101r, PMID 9636038.
- ^ Sevier, Carolyn S.; Kaiser, Chris A. (2002), "Formation and transfer of disulphide bonds in living cells", Nature Rev. Mol. Cell. Biol. 3 (11): 836–47, doi:10.1038/nrm954, PMID 12415301.
- ^ Lill, Roland; Mühlenhoff, Ulrich (2006), "Iron-Sulfur Protein Biogenesis in Eukaryotes: Components and Mechanisms", Ann. Rev. Cell Dev. Biol. 22: 457–86, doi:10.1146/annurev.cellbio.22.010305.104538, PMID 16824008.
- ^ Lippard, Stephen J.; Berg, Jeremy M. (1994), Principles of Bioinorganic Chemistry, Mill Valley, CA: University Science Books, ISBN 0-935702-73-3.
- ^ Baker, David H.; Czarnecki-Maulden, Gail L. (1987), "Pharmacologic role of cysteine in ameliorating or exacerbating mineral toxicities", J. Nutr. 117 (6): 1003–10, PMID 3298579, http://jn.nutrition.org/cgi/reprint/117/6/1003.
- ^ Hui, Nip W.; Rogers, R. (2001), Hui, Y., ed., Meat science and applications, CRC Press, p. 74, ISBN 0824705483.
- ^ Food Ingredients and Colors, U.S. Food and Drug Administration, November 2004, http://www.cfsan.fda.gov/~dms/foodic.html, retrieved 2009-09-06.
- ^ Martin, Terry (2009-06-25), The List of Additives in Cigarettes, about.com, http://quitsmoking.about.com/cs/nicotineinhaler/a/cigingredients.htm, retrieved 2009-09-06.
- ^ Powell BC, Walker SK, Bawden CS, Sivaprasad AV, Rogers GE (1994), "Transgenic sheep and wool growth: possibilities and current status.", Reprod Fertil Dev 6 (5): 615–23, doi:10.1071/RD9940615, PMID 7569041, http://www.ncbi.nlm.nih.gov/pubmed/7569041.
- ^ Sprince, Herbert; Parker, Clarence M.; Smith, George G.; Gonzales, Leon J. (1974), "Protection against Acetaldehyde Toxicity in the rat by L-cysteine, thiamine and L-2-Methylthiazolidine-4-carboxylic acid", Inflam. Res. 4 (2): 125–30, doi:10.1007/BF01966822, PMID 4842541.
External links
- International Kidney Stone Institute
- http://www.chemie.fu-berlin.de/chemistry/bio/aminoacid/cystein en.html
- On the hydrophobic nature of cysteine.
- 952-10-3056-9 Interaction of alcohol and smoking in the pathogenesis of upper digestive tract cancers - possible chemoprevention with cysteine
- Cystine Kidney Stones
The 20 common amino acids By properties AliphaticBranched-chain amino acids (Valine · Isoleucine · Leucine) · Methionine · Alanine · Proline · GlycineAromaticPolar, unchargedPositive charge (pKa)Negative charge (pKa)GeneralOther classifications biochemical families: prot · nucl · carb (glpr, alco, glys) · lipd (fata/i, phld, strd, gllp, eico) · amac/i · ncbs/i · ttpy/iE numbers Colours (E100–199) • Preservatives (E200–299) • Antioxidants & acidity regulators (E300–399) • Thickeners, stabilisers & emulsifiers (E400–499) • pH regulators & anticaking agents (E500–599) • Flavour enhancers (E600–699) • Miscellaneous (E900–999) • Additional chemicals (E1100–1599)
Waxes (E900–909) • Synthetic glazes (E910–919) • Improving agents (E920–929) • Packaging gases (E930–949) • Sweeteners (E950–969) • Foaming agents (E990–999)
L-cysteine (E920) • L-cystine (E921) • Potassium persulfate (E922) • Ammonium persulfate (E923) • Potassium bromate (E924) • Chlorine (E925) • Chlorine dioxide (E926) • Azodicarbonamide (E927) • Carbamide (E927b) • Benzoyl peroxide (E928)
K→acetyl-CoA G otherα-Ketoisovaleric acid · Isobutyryl-CoA · Methacrylyl-CoA · 3-Hydroxyisobutyryl-CoA · 3-Hydroxyisobutyric acid · 2-Methyl-3-oxopropanoic acidG→fumarateG→oxaloacetatesee urea cycleOther biochemical families: prot · nucl · carb (glpr, alco, glys) · lipd (fata/i, phld, strd, gllp, eico) · amac/i · ncbs/i · ttpy/iCategories:- Proteinogenic amino acids
- Glucogenic amino acids
- Sulfur amino acids
- Thiols
- C([C@@H](C(=O)O)N)S
Wikimedia Foundation. 2010.