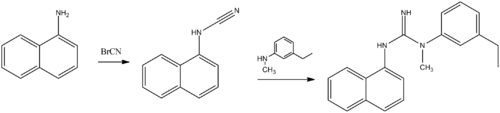
- Aptiganel
-
Aptiganel 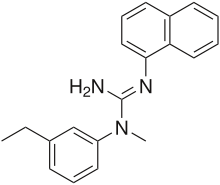
Systematic (IUPAC) name 1-(3-ethylphenyl)-1-methyl-2-naphthalen-1-ylguanidine Clinical data Pregnancy cat. ? Legal status Uncontrolled Identifiers CAS number 137159-92-3 ATC code None PubChem CID 60840 UNII 46475LV84I 
Chemical data Formula C20H21N3 Mol. mass 303.401 g/mol SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Aptiganel (Cerestat; CNS-1102) is a drug which acts as a noncompetitive NMDA antagonist.[1] It has neuroprotective effects and was researched for potential use in the treatment of stroke,[2] but despite positive results in animal studies,[3] human trials showed limited efficacy,[4] as well as undesirable side effects such as sedation and hallucinations,[5][6] and clinical development was ultimately not continued.[7]
References
- ^ Reddy NL, Hu LY, Cotter RE, Fischer JB, Wong WJ, McBurney RN, Weber E, Holmes DL, Wong ST, Prasad R, et al. Synthesis and structure-activity studies of N,N'-diarylguanidine derivatives. N-(1-naphthyl)-N'-(3-ethylphenyl)-N'-methylguanidine: a new, selective noncompetitive NMDA receptor antagonist. Journal of Medicinal Chemistry. 1994 Jan 21;37(2):260-7. PMID 8295213
- ^ Muir KW, Grosset DG, Gamzu E, Lees KR. Pharmacological effects of the non-competitive NMDA antagonist CNS 1102 in normal volunteers. British Journal of Clinical Pharmacology. 1994 Jul;38(1):33-8. PMID 7946934
- ^ Schäbitz WR, Li F, Fisher M. The N-methyl-D-aspartate antagonist CNS 1102 protects cerebral gray and white matter from ischemic injury following temporary focal ischemia in rats. Stroke. 2000 Jul;31(7):1709-14. PMID 10884477
- ^ Albers GW, Goldstein LB, Hall D, Lesko LM; Aptiganel Acute Stroke Investigators. Aptiganel hydrochloride in acute ischemic stroke: a randomized controlled trial. Journal of the American Medical Association. 2001 Dec 5;286(21):2673-82. PMID 11730442
- ^ Muir KW, Grosset DG, Lees KR. Effects of prolonged infusions of the NMDA antagonist aptiganel hydrochloride (CNS 1102) in normal volunteers. Clinical Neuropharmacology. 1997 Aug;20(4):311-21. PMID 9260729
- ^ Lees KR. Cerestat and other NMDA antagonists in ischemic stroke. Neurology. 1997 Nov;49(5 Suppl 4):S66-9. PMID 9371155
- ^ Hoyte L, Barber PA, Buchan AM, Hill MD. The rise and fall of NMDA antagonists for ischemic stroke. Current Molecular Medicine. 2004 Mar;4(2):131-6. PMID 15032709
Glutamatergics Ionotropic Agonists: 5-Fluorowillardiine • AMPA • Domoic acid • Quisqualic acid; Positive allosteric modulators: Aniracetam • Cyclothiazide • CX-516 • CX-546 • CX-614 • CX-691 • CX-717 • Diazoxide • HCTZ • IDRA-21 • LY-392,098 • LY-404,187 • LY-451,395 • LY-451,646 • LY-503,430 • Oxiracetam • PEPA • Piracetam • Pramiracetam • S-18986 • Sunifiram • Unifiram
Antagonists: ATPO • Barbiturates • Caroverine • CNQX • DNQX • GYKI-52466 • NBQX • Perampanel • Talampanel • Tezampanel • Topiramate; Negative allosteric modulators: GYKI-53,655Agonists: Glutamate/acite site competitive agonists: Aspartate • Glutamate • Homoquinolinic acid • Ibotenic acid • NMDA • Quinolinic acid • Tetrazolylglycine; Glycine site agonists: ACBD • ACPC • ACPD • Alanine • CCG • Cycloserine • DHPG • Fluoroalanine • Glycine • HA-966 • L-687,414 • Milacemide • Sarcosine • Serine • Tetrazolylglycine; Polyamine site agonists: Acamprosate • Spermidine • Spermine
Antagonists: Competitive antagonists: AP5 (APV) • AP7 • CGP-37849 • CGP-39551 • CGP-39653 • CGP-40116 • CGS-19755 • CPP • LY-233,053 • LY-235,959 • LY-274,614 • MDL-100,453 • Midafotel (d-CPPene) • NPC-12,626 • NPC-17,742 • PBPD • PEAQX • Perzinfotel • PPDA • SDZ-220581 • Selfotel; Noncompetitive antagonists: ARR-15,896 • Caroverine • Dexanabinol • FPL-12495 • FR-115,427 • Hodgkinsine • Magnesium • MDL-27,266 • NPS-1506 • Psychotridine • Zinc; Uncompetitive pore blockers: 2-MDP • 3-MeO-PCP • 8A-PDHQ • Alaproclate • Amantadine • Aptiganel • ARL-12,495 • ARL-15,896-AR • ARL-16,247 • Budipine • Delucemine • Dexoxadrol • Dextrallorphan • Dieticyclidine • Dizocilpine • Endopsychosin • Esketamine • Etoxadrol • Eticyclidine • Gacyclidine • Ibogaine • Indantadol • Ketamine • Ketobemidone • Loperamide • Memantine • Meperidine (Pethidine) • Methadone • Methorphan (Dextromethorphan, Levomethorphan) • Methoxetamine • Milnacipran • Morphanol (Dextrorphan, Levorphanol) • NEFA • Neramexane • Nitrous oxide • Noribogaine • Orphenadrine • PCPr • Phencyclamine • Phencyclidine • Propoxyphene • Remacemide • Rhynchophylline • Riluzole • Rimantadine • Rolicyclidine • Sabeluzole • Tenocyclidine • Tiletamine • Tramadol • Xenon; Glycine site antagonists: ACEA-1021 • ACEA-1328 • ACPC • Carisoprodol • CGP-39653 • CKA • DCKA • Felbamate • Gavestinel • GV-196,771 • Kynurenic acid • L-689,560 • L-701,324 • Lacosamide • Licostinel • LU-73,068 • MDL-105,519 • Meprobamate • MRZ 2/576 • PNQX • ZD-9379; NR2B subunit antagonists: Besonprodil • CO-101,244 (PD-174,494) • CP-101,606 • Eliprodil • Haloperidol • Ifenprodil • Isoxsuprine • Nylidrin • Ro8-4304 • Ro25-6981 • Traxoprodil; Polyamine site antagonists: Arcaine • Co 101676 • Diaminopropane • Acamprosate • Diethylenetriamine • Huperzine A • Putrescine • Ro 25-6981; Unclassified/unsorted antagonists: Chloroform • Diethyl ether • Enflurane • Ethanol (Alcohol) • Halothane • Isoflurane • Methoxyflurane • Toluene • Trichloroethane • Trichloroethanol • Trichloroethylene • XyleneAgonists: 5-Iodowillardiine • ATPA • Domoic acid • Kainic acid • LY-339,434 • SYM-2081
Antagonists: CNQX • DNQX • LY-382,884 • NBQX • NS102 • Tezampanel • Topiramate • UBP-302; Negative allosteric modulators: NS-3763Metabotropic Agonists: Unselective: ACPD • DHPG • Quisqualic acid; mGlu1-selective: Ro01-6128 • Ro67-4853 • Ro67-7476 • VU-71; mGlu5-selective: ADX-47273 • CDPPB • CHPG • DFB • VU-1545
Antagonists: Unselective: MCPG • NPS-2390; mGlu1-selective: BAY 36-7620 • CPCCOEt • LY-367,385 • LY-456,236; mGlu5-selective: Dipraglurant • DMeOB • Fenobam • LY-344,545 • MPEP • MTEP • SIB-1757 • SIB-1893Agonists: Unselective: L-AP4; mGlu4-selective: PHCCC • VU-001,171 • VU-0155,041; mGlu7-selective: AMN082; mGlu8-selective: DCPG
Antagonists: Unselective: CPPG • MAP4 • MSOP • MPPG • MTPG • UBP-1112; mGlu7-selective: MMPIPTransporter
inhibitorsDHKA • PDC • WAY-213,613vGluTs7-CKA • Evans blueCategories:- NMDA receptor antagonists
- Guanidines
- 1-Naphthylamines
- Anilines
- Hallucinogen stubs
Wikimedia Foundation. 2010.