- Epidermal growth factor receptor
-
This article is about a cell surface receptor. For estimated measure of kidney function (eGFR), see Glomerular filtration rate.
The epidermal growth factor receptor (EGFR; ErbB-1; HER1 in humans) is the cell-surface receptor for members of the epidermal growth factor family (EGF-family) of extracellular protein ligands.[1] The epidermal growth factor receptor is a member of the ErbB family of receptors, a subfamily of four closely related receptor tyrosine kinases: EGFR (ErbB-1), HER2/c-neu (ErbB-2), Her 3 (ErbB-3) and Her 4 (ErbB-4). Mutations affecting EGFR expression or activity could result in cancer.[2] Epidermal Growth Factor was discovered by Stanley Cohen of Vanderbilt University along with Rita Levi-Montalcini for which both received the Nobel prize in Physiology or Medicine in 1986.
Contents
Function
EGFR (epidermal growth factor receptor) exists on the cell surface and is activated by binding of its specific ligands, including epidermal growth factor and transforming growth factor α (TGFα) (note, a full list of the ligands able to activate EGFR and other members of the ErbB family is given in the ErbB article). ErbB2 has no known direct activating ligand, and may be in an activated state constitutively or become active upon heterodimerization with other family members such as EGFR. Upon activation by its growth factor ligands, EGFR undergoes a transition from an inactive monomeric form to an active homodimer - although there is some evidence that preformed inactive dimers may also exist before ligand binding[citation needed]. In addition to forming homodimers after ligand binding, EGFR may pair with another member of the ErbB receptor family, such as ErbB2/Her2/neu, to create an activated heterodimer. There is also evidence to suggest that clusters of activated EGFRs form, although it remains unclear whether this clustering is important for activation itself or occurs subsequent to activation of individual dimers[citation needed].
EGFR dimerization stimulates its intrinsic intracellular protein-tyrosine kinase activity. As a result, autophosphorylation of several tyrosine (Y) residues in the C-terminal domain of EGFR occurs. These include Y992, Y1045, Y1068, Y1148 and Y1173 as shown in the diagram to the left.[3] This autophosphorylation elicits downstream activation and signaling by several other proteins that associate with the phosphorylated tyrosines through their own phosphotyrosine-binding SH2 domains. These downstream signaling proteins initiate several signal transduction cascades, principally the MAPK, Akt and JNK pathways, leading to DNA synthesis and cell proliferation.[4] Such proteins modulate phenotypes such as cell migration, adhesion, and proliferation. Activation of the receptor is important for the innate immune response in human skin.[5] The kinase domain of EGFR can also cross-phosphorylate tyrosine residues of other receptors it is aggregated with, and can itself be activated in that manner.
Clinical applications
Mutations that lead to EGFR overexpression (known as upregulation) or overactivity have been associated with a number of cancers, including lung cancer, anal cancers[6] and glioblastoma multiforme. In this latter case a more or less specific mutation of EGFR, called EGFRvIII is often observed.[7] Mutations, amplifications or misregulations of EGFR or family members are implicated in about 30% of all epithelial cancers.
Mutations involving EGFR could lead to its constant activation, which could result in uncontrolled cell division – a predisposition for cancer.[8] Consequently, mutations of EGFR have been identified in several types of cancer, and it is the target of an expanding class of anticancer therapies.[2]
The identification of EGFR as an oncogene has led to the development of anticancer therapeutics directed against EGFR, including gefitinib[9] and erlotinib for lung cancer, and cetuximab for colon cancer.
Many therapeutic approaches are aimed at the EGFR. Cetuximab and panitumumab are examples of monoclonal antibody inhibitors. However the former is of the IgG1 type, the latter of the IgG2 type; consequences on antibody-dependent cellular cytotoxicity can be quite different.[10] Other monoclonals in clinical development are zalutumumab, nimotuzumab, and matuzumab. The monoclonal antibodies block the extracellular ligand binding domain. With the binding site blocked, signal molecules can no longer attach there and activate the tyrosine kinase.
Another method is using small molecules to inhibit the EGFR tyrosine kinase, which is on the cytoplasmic side of the receptor. Without kinase activity, EGFR is unable to activate itself, which is a prerequisite for binding of downstream adaptor proteins. Ostensibly by halting the signaling cascade in cells that rely on this pathway for growth, tumor proliferation and migration is diminished. Gefitinib, erlotinib, and lapatinib (mixed EGFR and ERBB2 inhibitor) are examples of small molecule kinase inhibitors.
There are several quantitative methods available that use protein phosphorylation detection to identify EGFR family inhibitors.[11]
EGFR and Lung Cancer
New drugs such as IRESSA and Tarceva directly target the EGFR. Patients have been divided into EGFR-positive and EGFR-negative, based upon whether a tissue test shows a mutation. EGFR-positive patients have shown an impressive 60% response rate, which exceeds the response rate for conventional chemotherapy.[12]
However, many patients develop resistance. Two primary sources of resistance are the T790M Mutation and MET oncogene.[12] However, as of 2010 there was no consensus of an accepted approach to combat resistance nor FDA approval of a specific combination. Preclinical results have been reported for AP26113 which targets the T790M mutation.
Preclinical
Efficient conversion of strongly absorbed light by plasmonic gold nanoparticles to heat energy and their easy bioconjugation suggest their use as selective photothermal agents in molecular cancer cell targeting. Two oral squamous carcinoma cell lines (HSC 313 and HOC 3 Clone 8) and one benign epithelial cell line (HaCaT) were incubated with anti-epithelial growth factor receptor (EGFR) antibody conjugated gold nanoparticles and then exposed to continuous visible argon ion laser at 514 nm. It is found that the malignant cells require less than half the laser energy to be killed than the benign cells after incubation with anti-EGFR antibody conjugated Au nanoparticles. No photothermal destruction is observed for all types of cells in the absence of nanoparticles at four times energy required to kill the malignant cells with anti-EGFR/Au conjugates bonded. Au nanoparticles thus offer a novel class of selective photothermal agents using a CW laser at low powers.[13]
Possible involvement in axonal regeneration
In 2005 it was shown that inhibitors of EGFR could enhance axonal regeneration on non-conducive substrates such as CNS myelin.[14] In July 2007 it was discovered that the blood clotting protein fibrinogen also activates EGFR, thereby inhibiting regeneration of axons.[15]
Natural EGFR inhibitors
Natural inhibitors include potato carboxypeptidase inhibitor (PCI), which contains a small cysteine-rich module, called a T-knot scaffold, that is shared by several different protein families, including the EGF family. Structural similarities with these factors can explain the antagonistic effect of PCI.[16]
Interactions
Epidermal growth factor receptor has been shown to interact with:
- Androgen receptor,[17][18]
- ARF4,[19]
- Beta-catenin,[20][21][22]
- Caveolin 1,[23]
- Caveolin 3,[23]
- Cbl gene,[24][25][26][27][28]
- CBLB,[29][25]
- CBLC,[30][31]
- CDC25A,[32]
- CRK,[29][33]
- Decorin,[34][35]
- Epidermal growth factor,[36][37]
- GRB14,[38]
- Grb2,[39][29][38][40][41][36][42][43][44][45]
- Janus kinase 2,[46]
- MUC1,[47][48]
- NCK1,[49][50][39]
- NCK2[39][51][52]
- PKC alpha,[53]
- PLCG1,[24][54]
- PLSCR1,[55]
- PTPN1,[56][57]
- PTPN11,[29][58]
- PTPN6,[58][59]
- SH2D3A,[60]
- SH3KBP1,[61][62]
- SHC1,[29][63]
- SOS1,[44][64][65]
- Src,[46][66][67]
- STAT1,[46][68]
- STAT3,[46][69]
- STAT5A,[46][29]
- Ubiquitin C,[26][27][70] and
- Wiskott-Aldrich syndrome protein.[71]
References
- ^ Herbst RS (2004). "Review of epidermal growth factor receptor biology". Int. J. Radiat. Oncol. Biol. Phys. 59 (2 Suppl): 21–6. doi:10.1016/j.ijrobp.2003.11.041. PMID 15142631.
- ^ a b Zhang H, Berezov A, Wang Q, Zhang G, Drebin J, Murali R, Greene MI (August 2007). "ErbB receptors: from oncogenes to targeted cancer therapies". J. Clin. Invest. 117 (8): 2051–8. doi:10.1172/JCI32278. PMC 1934579. PMID 17671639. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1934579.
- ^ Downward J, Parker P, Waterfield MD (1984). "Autophosphorylation sites on the epidermal growth factor receptor". Nature 311 (5985): 483–5. doi:10.1038/311483a0. PMID 6090945.
- ^ Oda K, Matsuoka Y, Funahashi A, Kitano H (2005). "A comprehensive pathway map of epidermal growth factor receptor signaling". Mol. Syst. Biol. 1 (1): 2005.0010. doi:10.1038/msb4100014. PMC 1681468. PMID 16729045. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1681468.
- ^ Sorensen OE, Tapa DR, Roupé KM, "et al." (2006). "Injury-induced innate immune response in human skin mediated by transactivation of the epidermal growth factor receptor". J Clin Invest. 116 (7): 1878–1885. doi:10.1172/JCI28422. PMC 1479426. PMID 16778986. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1479426.
- ^ Walker, F.; Abramowitz, L.; Benabderrahmane, D.; Duval, X.; Descatoire, V. �R.; Hénin, D.; Lehy, T. �R. �S.; Aparicio, T. (2009). "Growth factor receptor expression in anal squamous lesions: modifications associated with oncogenic human papillomavirus and human immunodeficiency virus☆". Human Pathology 40 (11): 1517–1527. doi:10.1016/j.humpath.2009.05.010. PMID 19716155.
- ^ Kuan CT, Wikstrand CJ, Bigner DD (June 2001). "EGF mutant receptor vIII as a molecular target in cancer therapy". Endocr. Relat. Cancer 8 (2): 83–96. doi:10.1677/erc.0.0080083. PMID 11397666.
- ^ Lynch TJ, Bell DW, Sordella R, Gurubhagavatula S, Okimoto RA, Brannigan BW, Harris PL, Haserlat SM, Supko JG, Haluska FG, Louis DN, Christiani DC, Settleman J, Haber DA (May 2004). "Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib". N. Engl. J. Med. 350 (21): 2129–39. doi:10.1056/NEJMoa040938. PMID 15118073.
- ^ Paez JG, Jänne PA, Lee JC, Tracy S, Greulich H, Gabriel S, Herman P, Kaye FJ, Lindeman N, Boggon TJ, Naoki K, Sasaki H, Fujii Y, Eck MJ, Sellers WR, Johnson BE, Meyerson M' (June 2004). "EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy". Science 304 (5676): 1497–500. doi:10.1126/science.1099314. PMID 15118125.
- ^ Yan L, Beckman RA (October 2005). "Pharmacogenetics and pharmacogenomics in oncology therapeutic antibody development". BioTechniques 39 (4): 565–8. doi:10.2144/000112043. PMID 16235569.
- ^ Olive DM (October 2004). "Quantitative methods for the analysis of protein phosphorylation in drug development". Expert Rev Proteomics 1 (3): 327–41. doi:10.1586/14789450.1.3.327. PMID 15966829. http://biosupport.licor.com/docs/2005/Olive.pdf.
- ^ a b Jackman DM, Miller VA, Cioffredi LA, Yeap BY, Jänne PA, Riely GJ, Ruiz MG, Giaccone G, Sequist LV, Johnson BE (August 2009). "Impact of epidermal growth factor receptor and KRAS mutations on clinical outcomes in previously untreated non-small cell lung cancer patients: results of an online tumor registry of clinical trials". Clin. Cancer Res. 15 (16): 5267–73. doi:10.1158/1078-0432.CCR-09-0888. PMID 19671843.
- ^ El-Sayed IH, Huang X, El-Sayed MA (July 2006). "Selective laser photo-thermal therapy of epithelial carcinoma using anti-EGFR antibody conjugated gold nanoparticles". Cancer Lett. 239 (1): 129–35. doi:10.1016/j.canlet.2005.07.035. PMID 16198049.
- ^ Koprivica V, Cho KS, Park JB, Yiu G, Atwal J, Gore B, Kim JA, Lin E, Tessier-Lavigne M, Chen DF, He Z (October 2005). "EGFR activation mediates inhibition of axon regeneration by myelin and chondroitin sulfate proteoglycans". Science 310 (5745): 106–10. doi:10.1126/science.1115462. PMID 16210539.
- ^ Schachtrup C, Lu P, Jones LL, et al. (2007). "Fibrinogen inhibits neurite outgrowth via beta 3 integrin-mediated phosphorylation of the EGF receptor". Proc. Natl. Acad. Sci. U.S.A. 104 (28): 11814–9. doi:10.1073/pnas.0704045104. PMC 1913857. PMID 17606926. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1913857. Lay summary.
- ^ Blanco-Aparicio C, Molina MA, Fernández-Salas E, Frazier ML, Mas JM, Querol E, Avilés FX, de Llorens R (May 1998). "Potato carboxypeptidase inhibitor, a T-knot protein, is an epidermal growth factor antagonist that inhibits tumor cell growth". J. Biol. Chem. 273 (20): 12370–7. doi:10.1074/jbc.273.20.12370. PMID 9575190. http://www.jbc.org/cgi/content/full/273/20/12370.
- ^ Bonaccorsi, Lorella; Carloni Vinicio, Muratori Monica, Formigli Lucia, Zecchi Sandra, Forti Gianni, Baldi Elisabetta (Oct. 2004). "EGF receptor (EGFR) signaling promoting invasion is disrupted in androgen-sensitive prostate cancer cells by an interaction between EGFR and androgen receptor (AR)". Int. J. Cancer 112 (1): 78–86. doi:10.1002/ijc.20362. ISSN 0020-7136. PMID 15305378.
- ^ Bonaccorsi, L; Muratori M, Carloni V, Marchiani S, Formigli L, Forti G, Baldi E (Aug. 2004). "The androgen receptor associates with the epidermal growth factor receptor in androgen-sensitive prostate cancer cells". Steroids 69 (8-9): 549–52. doi:10.1016/j.steroids.2004.05.011. ISSN 0039-128X. PMID 15288768.
- ^ Kim, Sung-Woo; Hayashi Masaaki, Lo Jeng-Fan, Yang Young, Yoo Jin-San, Lee Jiing-Dwan (Jan. 2003). "ADP-ribosylation factor 4 small GTPase mediates epidermal growth factor receptor-dependent phospholipase D2 activation". J. Biol. Chem. 278 (4): 2661–8. doi:10.1074/jbc.M205819200. ISSN 0021-9258. PMID 12446727.
- ^ Hazan, R B; Norton L (Apr. 1998). "The epidermal growth factor receptor modulates the interaction of E-cadherin with the actin cytoskeleton". J. Biol. Chem. 273 (15): 9078–84. doi:10.1074/jbc.273.15.9078. ISSN 0021-9258. PMID 9535896.
- ^ Schroeder, Joyce A; Adriance Melissa C, McConnell Elizabeth J, Thompson Melissa C, Pockaj Barbara, Gendler Sandra J (Jun. 2002). "ErbB-beta-catenin complexes are associated with human infiltrating ductal breast and murine mammary tumor virus (MMTV)-Wnt-1 and MMTV-c-Neu transgenic carcinomas". J. Biol. Chem. 277 (25): 22692–8. doi:10.1074/jbc.M201975200. ISSN 0021-9258. PMID 11950845.
- ^ Takahashi, K; Suzuki K, Tsukatani Y (Jul. 1997). "Induction of tyrosine phosphorylation and association of beta-catenin with EGF receptor upon tryptic digestion of quiescent cells at confluence". Oncogene 15 (1): 71–8. doi:10.1038/sj.onc.1201160. ISSN 0950-9232. PMID 9233779.
- ^ a b Couet, J; Sargiacomo M, Lisanti M P (Nov. 1997). "Interaction of a receptor tyrosine kinase, EGF-R, with caveolins. Caveolin binding negatively regulates tyrosine and serine/threonine kinase activities". J. Biol. Chem. 272 (48): 30429–38. doi:10.1074/jbc.272.48.30429. ISSN 0021-9258. PMID 9374534.
- ^ a b Tvorogov, Denis; Carpenter Graham (Jul. 2002). "EGF-dependent association of phospholipase C-gamma1 with c-Cbl". Exp. Cell Res. 277 (1): 86–94. doi:10.1006/excr.2002.5545. ISSN 0014-4827. PMID 12061819.
- ^ a b Ettenberg, S A; Keane M M, Nau M M, Frankel M, Wang L M, Pierce J H, Lipkowitz S (Mar. 1999). "cbl-b inhibits epidermal growth factor receptor signaling". Oncogene 18 (10): 1855–66. doi:10.1038/sj.onc.1202499. ISSN 0950-9232. PMID 10086340.
- ^ a b Pennock, Steven; Wang Zhixiang (May. 2008). "A tale of two Cbls: interplay of c-Cbl and Cbl-b in epidermal growth factor receptor downregulation". Mol. Cell. Biol. 28 (9): 3020–37. doi:10.1128/MCB.01809-07. PMC 2293090. PMID 18316398. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2293090.
- ^ a b Umebayashi, Kyohei; Stenmark Harald, Yoshimori Tamotsu (Aug. 2008). "Ubc4/5 and c-Cbl continue to ubiquitinate EGF receptor after internalization to facilitate polyubiquitination and degradation". Mol. Biol. Cell 19 (8): 3454–62. doi:10.1091/mbc.E07-10-0988. PMC 2488299. PMID 18508924. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2488299.
- ^ Ng, Cherlyn; Jackson Rebecca A, Buschdorf Jan P, Sun Qingxiang, Guy Graeme R, Sivaraman J (Mar. 2008). "Structural basis for a novel intrapeptidyl H-bond and reverse binding of c-Cbl-TKB domain substrates". EMBO J. 27 (5): 804–16. doi:10.1038/emboj.2008.18. PMC 2265755. PMID 18273061. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2265755.
- ^ a b c d e f Schulze, Waltraud X; Deng Lei, Mann Matthias (2005). "Phosphotyrosine interactome of the ErbB-receptor kinase family". Mol. Syst. Biol. 1 (1): 2005.0008. doi:10.1038/msb4100012. PMC 1681463. PMID 16729043. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1681463.
- ^ Kim, M; Tezuka T, Suziki Y, Sugano S, Hirai M, Yamamoto T (Oct. 1999). "Molecular cloning and characterization of a novel cbl-family gene, cbl-c". Gene 239 (1): 145–54. doi:10.1016/S0378-1119(99)00356-X. ISSN 0378-1119. PMID 10571044.
- ^ Keane, M M; Ettenberg S A, Nau M M, Banerjee P, Cuello M, Penninger J, Lipkowitz S (Jun. 1999). "cbl-3: a new mammalian cbl family protein". Oncogene 18 (22): 3365–75. doi:10.1038/sj.onc.1202753. ISSN 0950-9232. PMID 10362357.
- ^ Wang, Ziqiu; Wang Meifang, Lazo John S, Carr Brian I (May. 2002). "Identification of epidermal growth factor receptor as a target of Cdc25A protein phosphatase". J. Biol. Chem. 277 (22): 19470–5. doi:10.1074/jbc.M201097200. ISSN 0021-9258. PMID 11912208.
- ^ Hashimoto, Y; Katayama H, Kiyokawa E, Ota S, Kurata T, Gotoh N, Otsuka N, Shibata M, Matsuda M (Jul. 1998). "Phosphorylation of CrkII adaptor protein at tyrosine 221 by epidermal growth factor receptor". J. Biol. Chem. 273 (27): 17186–91. doi:10.1074/jbc.273.27.17186. ISSN 0021-9258. PMID 9642287.
- ^ Santra, Manoranjan; Reed Charles C, Iozzo Renato V (Sep. 2002). "Decorin binds to a narrow region of the epidermal growth factor (EGF) receptor, partially overlapping but distinct from the EGF-binding epitope". J. Biol. Chem. 277 (38): 35671–81. doi:10.1074/jbc.M205317200. ISSN 0021-9258. PMID 12105206.
- ^ Iozzo, R V; Moscatello D K, McQuillan D J, Eichstetter I (Feb. 1999). "Decorin is a biological ligand for the epidermal growth factor receptor". J. Biol. Chem. 274 (8): 4489–92. doi:10.1074/jbc.274.8.4489. ISSN 0021-9258. PMID 9988678.
- ^ a b Wong, L; Deb T B, Thompson S A, Wells A, Johnson G R (Mar. 1999). "A differential requirement for the COOH-terminal region of the epidermal growth factor (EGF) receptor in amphiregulin and EGF mitogenic signaling". J. Biol. Chem. 274 (13): 8900–9. doi:10.1074/jbc.274.13.8900. ISSN 0021-9258. PMID 10085134.
- ^ Stortelers, Catelijne; Souriau Christelle, van Liempt Ellis, van de Poll Monique L M, van Zoelen Everardus J J (Jul. 2002). "Role of the N-terminus of epidermal growth factor in ErbB-2/ErbB-3 binding studied by phage display". Biochemistry 41 (27): 8732–41. doi:10.1021/bi025878c. ISSN 0006-2960. PMID 12093292.
- ^ a b Daly, R J; Sanderson G M, Janes P W, Sutherland R L (May. 1996). "Cloning and characterization of GRB14, a novel member of the GRB7 gene family". J. Biol. Chem. 271 (21): 12502–10. doi:10.1074/jbc.271.21.12502. ISSN 0021-9258. PMID 8647858.
- ^ a b c Braverman, L E; Quilliam L A (Feb. 1999). "Identification of Grb4/Nckbeta, a src homology 2 and 3 domain-containing adapter protein having similar binding and biological properties to Nck". J. Biol. Chem. 274 (9): 5542–9. doi:10.1074/jbc.274.9.5542. ISSN 0021-9258. PMID 10026169.
- ^ Blagoev, Blagoy; Kratchmarova Irina, Ong Shao-En, Nielsen Mogens, Foster Leonard J, Mann Matthias (Mar. 2003). "A proteomics strategy to elucidate functional protein-protein interactions applied to EGF signaling". Nat. Biotechnol. 21 (3): 315–8. doi:10.1038/nbt790. ISSN 1087-0156. PMID 12577067.
- ^ Oneyama, Chitose; Nakano Hirofumi, Sharma Sreenath V (Mar. 2002). "UCS15A, a novel small molecule, SH3 domain-mediated protein-protein interaction blocking drug". Oncogene 21 (13): 2037–50. doi:10.1038/sj.onc.1205271. ISSN 0950-9232. PMID 11960376.
- ^ Okutani, T; Okabayashi Y, Kido Y, Sugimoto Y, Sakaguchi K, Matuoka K, Takenawa T, Kasuga M (Dec. 1994). "Grb2/Ash binds directly to tyrosines 1068 and 1086 and indirectly to tyrosine 1148 of activated human epidermal growth factor receptors in intact cells". J. Biol. Chem. 269 (49): 31310–4. ISSN 0021-9258. PMID 7527043.
- ^ Tortora, G; Damiano V, Bianco C, Baldassarre G, Bianco A R, Lanfrancone L, Pelicci P G, Ciardiello F (Feb. 1997). "The RIalpha subunit of protein kinase A (PKA) binds to Grb2 and allows PKA interaction with the activated EGF-receptor". Oncogene 14 (8): 923–8. doi:10.1038/sj.onc.1200906. ISSN 0950-9232. PMID 9050991.
- ^ a b Buday, L; Egan S E, Rodriguez Viciana P, Cantrell D A, Downward J (Mar. 1994). "A complex of Grb2 adaptor protein, Sos exchange factor, and a 36-kDa membrane-bound tyrosine phosphoprotein is implicated in ras activation in T cells". J. Biol. Chem. 269 (12): 9019–23. ISSN 0021-9258. PMID 7510700.
- ^ Lowenstein, E J; Daly R J, Batzer A G, Li W, Margolis B, Lammers R, Ullrich A, Skolnik E Y, Bar-Sagi D, Schlessinger J (Aug. 1992). "The SH2 and SH3 domain-containing protein GRB2 links receptor tyrosine kinases to ras signaling". Cell 70 (3): 431–42. doi:10.1016/0092-8674(92)90167-B. ISSN 0092-8674. PMID 1322798.
- ^ a b c d e Olayioye, M A; Beuvink I, Horsch K, Daly J M, Hynes N E (Jun. 1999). "ErbB receptor-induced activation of stat transcription factors is mediated by Src tyrosine kinases". J. Biol. Chem. 274 (24): 17209–18. doi:10.1074/jbc.274.24.17209. ISSN 0021-9258. PMID 10358079.
- ^ Schroeder, J A; Thompson M C, Gardner M M, Gendler S J (Apr. 2001). "Transgenic MUC1 interacts with epidermal growth factor receptor and correlates with mitogen-activated protein kinase activation in the mouse mammary gland". J. Biol. Chem. 276 (16): 13057–64. doi:10.1074/jbc.M011248200. ISSN 0021-9258. PMID 11278868.
- ^ Li, Y; Ren J, Yu W, Li Q, Kuwahara H, Yin L, Carraway K L, Kufe D (Sep. 2001). "The epidermal growth factor receptor regulates interaction of the human DF3/MUC1 carcinoma antigen with c-Src and beta-catenin". J. Biol. Chem. 276 (38): 35239–42. doi:10.1074/jbc.C100359200. ISSN 0021-9258. PMID 11483589.
- ^ Tang, J; Feng G S, Li W (Oct. 1997). "Induced direct binding of the adapter protein Nck to the GTPase-activating protein-associated protein p62 by epidermal growth factor". Oncogene 15 (15): 1823–32. doi:10.1038/sj.onc.1201351. ISSN 0950-9232. PMID 9362449.
- ^ Li, W; Hu P, Skolnik E Y, Ullrich A, Schlessinger J (Dec. 1992). "The SH2 and SH3 domain-containing Nck protein is oncogenic and a common target for phosphorylation by different surface receptors". Mol. Cell. Biol. 12 (12): 5824–33. ISSN 0270-7306. PMC 360522. PMID 1333047. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=360522.
- ^ Chen, M; She H, Davis E M, Spicer C M, Kim L, Ren R, Le Beau M M, Li W (Sep. 1998). "Identification of Nck family genes, chromosomal localization, expression, and signaling specificity". J. Biol. Chem. 273 (39): 25171–8. doi:10.1074/jbc.273.39.25171. ISSN 0021-9258. PMID 9737977.
- ^ Tu, Y; Li F, Wu C (Dec. 1998). "Nck-2, a novel Src homology2/3-containing adaptor protein that interacts with the LIM-only protein PINCH and components of growth factor receptor kinase-signaling pathways". Mol. Biol. Cell 9 (12): 3367–82. ISSN 1059-1524. PMC 25640. PMID 9843575. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=25640.
- ^ Gauthier, Mona L; Torretto Cheryl, Ly John, Francescutti Valerie, O'Day Danton H (Aug. 2003). "Protein kinase Calpha negatively regulates cell spreading and motility in MDA-MB-231 human breast cancer cells downstream of epidermal growth factor receptor". Biochem. Biophys. Res. Commun. 307 (4): 839–46. doi:10.1016/S0006-291X(03)01273-7. ISSN 0006-291X. PMID 12878187.
- ^ Bedrin, M S; Abolafia C M, Thompson J F (Jul. 1997). "Cytoskeletal association of epidermal growth factor receptor and associated signaling proteins is regulated by cell density in IEC-6 intestinal cells". J. Cell. Physiol. 172 (1): 126–36. doi:10.1002/(SICI)1097-4652(199707)172:1<126::AID-JCP14>3.0.CO;2-A. ISSN 0021-9541. PMID 9207933.
- ^ Sun, Jun; Nanjundan Meera, Pike Linda J, Wiedmer Therese, Sims Peter J (May. 2002). "Plasma membrane phospholipid scramblase 1 is enriched in lipid rafts and interacts with the epidermal growth factor receptor". Biochemistry 41 (20): 6338–45. doi:10.1021/bi025610l. ISSN 0006-2960. PMID 12009895.
- ^ Sarmiento, M; Puius Y A, Vetter S W, Keng Y F, Wu L, Zhao Y, Lawrence D S, Almo S C, Zhang Z Y (Jul. 2000). "Structural basis of plasticity in protein tyrosine phosphatase 1B substrate recognition". Biochemistry 39 (28): 8171–9. doi:10.1021/bi000319w. ISSN 0006-2960. PMID 10889023.
- ^ Zhang, Z Y; Walsh A B, Wu L, McNamara D J, Dobrusin E M, Miller W T (Mar. 1996). "Determinants of substrate recognition in the protein-tyrosine phosphatase, PTP1". J. Biol. Chem. 271 (10): 5386–92. doi:10.1074/jbc.271.10.5386. ISSN 0021-9258. PMID 8621392.
- ^ a b Tomic, S; Greiser U, Lammers R, Kharitonenkov A, Imyanitov E, Ullrich A, Böhmer F D (Sep. 1995). "Association of SH2 domain protein tyrosine phosphatases with the epidermal growth factor receptor in human tumor cells. Phosphatidic acid activates receptor dephosphorylation by PTP1C". J. Biol. Chem. 270 (36): 21277–84. doi:10.1074/jbc.270.36.21277. ISSN 0021-9258. PMID 7673163.
- ^ Keilhack, H; Tenev T, Nyakatura E, Godovac-Zimmermann J, Nielsen L, Seedorf K, Böhmer F D (Sep. 1998). "Phosphotyrosine 1173 mediates binding of the protein-tyrosine phosphatase SHP-1 to the epidermal growth factor receptor and attenuation of receptor signaling". J. Biol. Chem. 273 (38): 24839–46. doi:10.1074/jbc.273.38.24839. ISSN 0021-9258. PMID 9733788.
- ^ Lu, Y; Brush J, Stewart T A (Apr. 1999). "NSP1 defines a novel family of adaptor proteins linking integrin and tyrosine kinase receptors to the c-Jun N-terminal kinase/stress-activated protein kinase signaling pathway". J. Biol. Chem. 274 (15): 10047–52. doi:10.1074/jbc.274.15.10047. ISSN 0021-9258. PMID 10187783.
- ^ Soubeyran, Philippe; Kowanetz Katarzyna, Szymkiewicz Iwona, Langdon Wallace Y, Dikic Ivan (Mar. 2002). "Cbl-CIN85-endophilin complex mediates ligand-induced downregulation of EGF receptors". Nature 416 (6877): 183–7. doi:10.1038/416183a. ISSN 0028-0836. PMID 11894095.
- ^ Szymkiewicz, Iwona; Kowanetz Katarzyna, Soubeyran Philippe, Dinarina Ana, Lipkowitz Stanley, Dikic Ivan (Oct. 2002). "CIN85 participates in Cbl-b-mediated down-regulation of receptor tyrosine kinases". J. Biol. Chem. 277 (42): 39666–72. doi:10.1074/jbc.M205535200. ISSN 0021-9258. PMID 12177062.
- ^ Sakaguchi, K; Okabayashi Y, Kido Y, Kimura S, Matsumura Y, Inushima K, Kasuga M (Apr. 1998). "Shc phosphotyrosine-binding domain dominantly interacts with epidermal growth factor receptors and mediates Ras activation in intact cells". Mol. Endocrinol. 12 (4): 536–43. doi:10.1210/me.12.4.536. ISSN 0888-8809. PMID 9544989.
- ^ Qian, X; Esteban L, Vass W C, Upadhyaya C, Papageorge A G, Yienger K, Ward J M, Lowy D R, Santos E (Feb. 2000). "The Sos1 and Sos2 Ras-specific exchange factors: differences in placental expression and signaling properties". EMBO J. 19 (4): 642–54. doi:10.1093/emboj/19.4.642. ISSN 0261-4189. PMC 305602. PMID 10675333. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=305602.
- ^ Qian, X; Vass W C, Papageorge A G, Anborgh P H, Lowy D R (Feb. 1998). "N terminus of Sos1 Ras exchange factor: critical roles for the Dbl and pleckstrin homology domains". Mol. Cell. Biol. 18 (2): 771–8. ISSN 0270-7306. PMC 108788. PMID 9447973. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=108788.
- ^ Keely, S J; Calandrella S O, Barrett K E (Apr. 2000). "Carbachol-stimulated transactivation of epidermal growth factor receptor and mitogen-activated protein kinase in T(84) cells is mediated by intracellular ca(2+), PYK-2, and p60(src)". J. Biol. Chem. 275 (17): 12619–25. doi:10.1074/jbc.275.17.12619. ISSN 0021-9258. PMID 10777553.
- ^ Sato, K; Kimoto M, Kakumoto M, Horiuchi D, Iwasaki T, Tokmakov A A, Fukami Y (Sep. 2000). "Adaptor protein Shc undergoes translocation and mediates up-regulation of the tyrosine kinase c-Src in EGF-stimulated A431 cells". Genes Cells 5 (9): 749–64. doi:10.1046/j.1365-2443.2000.00358.x. ISSN 1356-9597. PMID 10971656.
- ^ Xia, Ling; Wang Lijuan, Chung Alicia S, Ivanov Stanimir S, Ling Mike Y, Dragoi Ana M, Platt Adam, Gilmer Tona M, Fu Xin-Yuan, Chin Y Eugene (Aug. 2002). "Identification of both positive and negative domains within the epidermal growth factor receptor COOH-terminal region for signal transducer and activator of transcription (STAT) activation". J. Biol. Chem. 277 (34): 30716–23. doi:10.1074/jbc.M202823200. ISSN 0021-9258. PMID 12070153.
- ^ Yuan, Zheng-Long; Guan Ying-Jie, Wang Lijuan, Wei Wenyi, Kane Agnes B, Chin Y Eugene (Nov. 2004). "Central role of the threonine residue within the p+1 loop of receptor tyrosine kinase in STAT3 constitutive phosphorylation in metastatic cancer cells". Mol. Cell. Biol. 24 (21): 9390–400. doi:10.1128/MCB.24.21.9390-9400.2004. ISSN 0270-7306. PMC 522220. PMID 15485908. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=522220.
- ^ Sehat, Bita; Andersson Sandra, Girnita Leonard, Larsson Olle (Jul. 2008). "Identification of c-Cbl as a new ligase for insulin-like growth factor-I receptor with distinct roles from Mdm2 in receptor ubiquitination and endocytosis". Cancer Res. 68 (14): 5669–77. doi:10.1158/0008-5472.CAN-07-6364. PMID 18632619.
- ^ She, H Y; Rockow S, Tang J, Nishimura R, Skolnik E Y, Chen M, Margolis B, Li W (Sep. 1997). "Wiskott-Aldrich syndrome protein is associated with the adapter protein Grb2 and the epidermal growth factor receptor in living cells". Mol. Biol. Cell 8 (9): 1709–21. ISSN 1059-1524. PMC 305731. PMID 9307968. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=305731.
Further reading
- Carpenter G (1987). "Receptors for epidermal growth factor and other polypeptide mitogens". Annu. Rev. Biochem. 56 (1): 881–914. doi:10.1146/annurev.bi.56.070187.004313. PMID 3039909.
- Boonstra J, Rijken P, Humbel B, et al. (1995). "The epidermal growth factor". Cell Biol. Int. 19 (5): 413–30. doi:10.1006/cbir.1995.1086. PMID 7640657.
- Carpenter G (2000). "The EGF receptor: a nexus for trafficking and signaling". Bioessays 22 (8): 697–707. doi:10.1002/1521-1878(200008)22:8<697::AID-BIES3>3.0.CO;2-1. PMID 10918300.
- Filardo EJ (2002). "Epidermal growth factor receptor (EGFR) transactivation by estrogen via the G-protein-coupled receptor, GPR30: a novel signaling pathway with potential significance for breast cancer". J. Steroid Biochem. Mol. Biol. 80 (2): 231–8. doi:10.1016/S0960-0760(01)00190-X. PMID 11897506.
- Tiganis T (2002). "Protein tyrosine phosphatases: dephosphorylating the epidermal growth factor receptor". IUBMB Life 53 (1): 3–14. doi:10.1080/15216540210811. PMID 12018405.
- Di Fiore PP, Scita G (2002). "Eps8 in the midst of GTPases". Int. J. Biochem. Cell Biol. 34 (10): 1178–83. doi:10.1016/S1357-2725(02)00064-X. PMID 12127568.
- Benaim G, Villalobo A (2002). "Phosphorylation of calmodulin. Functional implications". Eur. J. Biochem. 269 (15): 3619–31. doi:10.1046/j.1432-1033.2002.03038.x. PMID 12153558.
- Leu TH, Maa MC (2004). "Functional implication of the interaction between EGF receptor and c-Src". Front. Biosci. 8 (1-3): s28–38. doi:10.2741/980. PMID 12456372.
- Anderson NL, Anderson NG (2003). "The human plasma proteome: history, character, and diagnostic prospects". Mol. Cell Proteomics 1 (11): 845–67. doi:10.1074/mcp.R200007-MCP200. PMID 12488461.
- Kari C, Chan TO, Rocha de Quadros M, Rodeck U (2003). "Targeting the epidermal growth factor receptor in cancer: apoptosis takes center stage". Cancer Res. 63 (1): 1–5. PMID 12517767.
- Bonaccorsi L, Muratori M, Carloni V, et al. (2003). "Androgen receptor and prostate cancer invasion". Int. J. Androl. 26 (1): 21–5. doi:10.1046/j.1365-2605.2003.00375.x. PMID 12534934.
- Reiter JL, Maihle NJ (2003). "Characterization and expression of novel 60-kDa and 110-kDa EGFR isoforms in human placenta". Ann. N. Y. Acad. Sci. 995 (1): 39–47. doi:10.1111/j.1749-6632.2003.tb03208.x. PMID 12814937.
- Adams TE, McKern NM, Ward CW (2005). "Signalling by the type 1 insulin-like growth factor receptor: interplay with the epidermal growth factor receptor". Growth Factors 22 (2): 89–95. doi:10.1080/08977190410001700998. PMID 15253384.
- Ferguson KM (2005). "Active and inactive conformations of the epidermal growth factor receptor". Biochem. Soc. Trans. 32 (Pt 5): 742–5. doi:10.1042/BST0320742. PMID 15494003.
- Chao C, Hellmich MR (2005). "Bi-directional signaling between gastrointestinal peptide hormone receptors and epidermal growth factor receptor". Growth Factors 22 (4): 261–8. doi:10.1080/08977190412331286900. PMID 15621729.
- Carlsson J, Ren ZP, Wester K, et al. (2006). "Planning for intracavitary anti-EGFR radionuclide therapy of gliomas. Literature review and data on EGFR expression". J. Neurooncol. 77 (1): 33–45. doi:10.1007/s11060-005-7410-z. PMID 16200342.
- Scartozzi M, Pierantoni C, Berardi R, et al. (2006). "Epidermal growth factor receptor: a promising therapeutic target for colorectal cancer". Anal. Quant. Cytol. Histol. 28 (2): 61–8. PMID 16637508.
- Prudkin L, Wistuba II (2006). "Epidermal growth factor receptor abnormalities in lung cancer. Pathogenetic and clinical implications". Annals of diagnostic pathology 10 (5): 306–15. doi:10.1016/j.anndiagpath.2006.06.011. PMID 16979526.
- Ahmed SM, Salgia R (2007). "Epidermal growth factor receptor mutations and susceptibility to targeted therapy in lung cancer". Respirology 11 (6): 687–92. doi:10.1111/j.1440-1843.2006.00887.x. PMID 17052295.
- Zhang X, Chang A (2007). "Somatic mutations of the epidermal growth factor receptor and non-small-cell lung cancer". J. Med. Genet. 44 (3): 166–72. doi:10.1136/jmg.2006.046102. PMC 2598028. PMID 17158592. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2598028.
- Cohenuram M, Saif MW (2007). "Epidermal growth factor receptor inhibition strategies in pancreatic cancer: past, present and the future". JOP 8 (1): 4–15. PMID 17228128.
- Mellinghoff IK, Cloughesy TF, Mischel PS (2007). "PTEN-mediated resistance to epidermal growth factor receptor kinase inhibitors". Clin. Cancer Res. 13 (2 Pt 1): 378–81. doi:10.1158/1078-0432.CCR-06-1992. PMID 17255257.
- Nakamura JL (2007). "The epidermal growth factor receptor in malignant gliomas: pathogenesis and therapeutic implications". Expert Opin. Ther. Targets 11 (4): 463–72. doi:10.1517/14728222.11.4.463. PMID 17373877.
External links
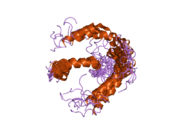
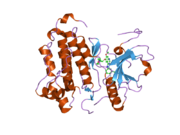
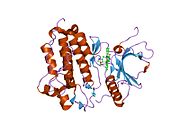
PDB gallery 1ivo: Crystal Structure of the Complex of Human Epidermal Growth Factor and Receptor Extracellular Domains.1m14: Tyrosine Kinase Domain from Epidermal Growth Factor Receptor1m17: Epidermal Growth Factor Receptor tyrosine kinase domain with 4-anilinoquinazoline inhibitor erlotinib1mox: Crystal Structure of Human Epidermal Growth Factor Receptor (residues 1-501) in complex with TGF-alpha1nql: Structure of the extracellular domain of human epidermal growth factor (EGF) receptor in an inactive (low pH) complex with EGF.1xkk: EGFR kinase domain complexed with a quinazoline inhibitor- GW5720161yy9: Structure of the extracellular domain of the epidermal growth factor receptor in complex with the Fab fragment of cetuximab/Erbitux/IMC-C2251z9i: A Structural Model for the Membrane-Bound Form of the Juxtamembrane Domain of the Epidermal Growth Factor Receptor2gs2: Crystal Structure of the active EGFR kinase domain2gs6: Crystal Structure of the active EGFR kinase domain in complex with an ATP analog-peptide conjugate2gs7: Crystal Structure of the inactive EGFR kinase domain in complex with AMP-PNP2itn: CRYSTAL STRUCTURE OF EGFR KINASE DOMAIN G719S MUTATION IN COMPLEX WITH AMP-PNP2ito: CRYSTAL STRUCTURE OF EGFR KINASE DOMAIN G719S MUTATION IN COMPLEX WITH IRESSA2itp: CRYSTAL STRUCTURE OF EGFR KINASE DOMAIN G719S MUTATION IN COMPLEX WITH AEE7882itq: CRYSTAL STRUCTURE OF EGFR KINASE DOMAIN G719S MUTATION IN COMPLEX WITH AFN9412itt: CRYSTAL STRUCTURE OF EGFR KINASE DOMAIN L858R MUTATION IN COMPLEX WITH AEE7882itu: CRYSTAL STRUCTURE OF EGFR KINASE DOMAIN L858R MUTATION IN COMPLEX WITH AFN9412itv: CRYSTAL STRUCTURE OF EGFR KINASE DOMAIN L858R MUTATION IN COMPLEX WITH AMP-PNP2itw: CRYSTAL STRUCTURE OF EGFR KINASE DOMAIN IN COMPLEX WITH AFN9412itx: CRYSTAL STRUCTURE OF EGFR KINASE DOMAIN IN COMPLEX WITH AMP-PNP2ity: CRYSTAL STRUCTURE OF EGFR KINASE DOMAIN IN COMPLEX WITH IRESSA2itz: CRYSTAL STRUCTURE OF EGFR KINASE DOMAIN L858R MUTATION IN COMPLEX WITH IRESSA2j5e: CRYSTAL STRUCTURE OF EGFR KINASE DOMAIN IN COMPLEX WITH AN IRREVERSIBLE INHIBITOR 13-JAB2j5f: CRYSTAL STRUCTURE OF EGFR KINASE DOMAIN IN COMPLEX WITH AN IRREVERSIBLE INHIBITOR 34-JAB2j6m: CRYSTAL STRUCTURE OF EGFR KINASE DOMAIN IN COMPLEX WITH AEE788Protein kinases: tyrosine kinases (EC 2.7.10) Receptor tyrosine kinases (EC 2.7.10.1) Insulin receptor familyPDGF receptor familyFGF receptor familyVEGF receptors familyHGF receptor familyTrk receptor familyEPH receptor familyLTK receptor familyLTK · ALKTIE receptor familyROR receptor familyROR1 · ROR2DDR receptor familyPTK7 receptor familyRYK receptor familyMuSK receptor familyROS receptor familyAATYK receptor familyAXL receptor familyRET receptor familyuncatagorisedNon-receptor tyrosine kinases (EC 2.7.10.2) ABL familyACK familyACK1 · TNK1CSK familyFAK familyFES familyFRK familyJAK familySRC-A familySRC-B familyTEC familySYK familyNeoplasm: Tumor suppressor genes/proteins and Oncogenes/Proto-oncogenes Ligand Receptor TSP: CDH1TSP: PTCH1TSP: TGF beta receptor 2Intracellular signaling P+Ps ONCO: Beta-catenin · TSP: APCHippo signaling pathwayOther/unknownNucleus TSP: VHL · ONCO: CBL - MDM2Mitochondria Other/ungrouped M: NEO
tsoc, mrkr
tumr, epon, para
drug (L1i/1e/V03)
Receptors: growth factor receptors Type I cytokine receptor Receptor protein serine/threonine kinase Receptor tyrosine kinase Fibroblast growth factor (1, 2, 3, 4)
Nerve growth factors: high affinity Trk (TrkA, TrkB, TrkC)
Somatomedin (Insulin-like growth factor 1)
ErbB/Epidermal growth factor
VEGF (1, 2, 3)Tumor necrosis factor receptor Ig superfamily Other/ungrouped Categories:- Human proteins
- Tyrosine kinase receptors
- Oncogenes
Wikimedia Foundation. 2010.