- Tretinoin
-
Tretinoin 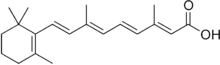

Systematic (IUPAC) name retinoic acid Clinical data Trade names Avita, Renova, Retin-a AHFS/Drugs.com monograph MedlinePlus a608032 Licence data US FDA:link Pregnancy cat. C(topical), D(oral)(US), X(oral)(Aus) Legal status Prescription Only (S4) (AU) POM (UK) ℞-only (US) Routes Topical, oral Pharmacokinetic data Protein binding > 95% Half-life 0.5-2 hours Identifiers CAS number 302-79-4 
ATC code D10AD01 L01XX14 PubChem CID 444795 DrugBank APRD00362 ChemSpider 392618 
UNII 5688UTC01R 
KEGG D00094 
ChEBI CHEBI:15367 
ChEMBL CHEMBL38 
Chemical data Formula C20H28O2 Mol. mass 300.4412 g/mol SMILES eMolecules & PubChem Physical data Melt. point 180 °C (356 °F)  (what is this?) (verify)
(what is this?) (verify)Tretinoin is the acid form of vitamin A and is also known as all-trans retinoic acid or ATRA. It is a drug commonly used to treat acne vulgaris and keratosis pilaris. It is available as a cream or gel (brand names Aberela, Airol, Renova, Atralin, Retin-A, Avita, Refissa, or Stieva-A). It is also used to treat acute promyelocytic leukemia (APL), and is sold for this indication by Roche under the brand name Vesanoid. It is also available as a generic.
Contents
Uses
Dermatology
Tretinoin is most commonly used as a form of acne treatment.[1] It was the first retinoid developed for this type of topical use. Tretinoin is the best studied retinoid in the treatment of photoaging.[2] It is used by some as a hair loss treatment and also a component of many commercial products that are advertised as being able to slow skin aging or remove wrinkles. Topical tretinoin is also used to treat and reduce the appearance of stretch marks by increasing collagen production in the dermis.[3]
Leukemia
In addition, tretinoin (marketed as Vesanoid) is also used to treat at least one form of cancer (acute promyelocytic leukemia (APL), also called acute myeloid leukemia subtype M3), usually together with other drugs, by causing the immature blood cells to differentiate (i.e. mature).[4][5] The pathology of the leukemia is due to the highly proliferative immature cells; retinoic acid drives these cells to develop into functional cells, which helps to alleviate the disease. It is usually prescribed for 15 days every 3 months at about 8-10 10 mg capsules per day.
Clinical pharmacology
Its success in treating acute promyelocytic leukemia was a major breakthrough in the treatment of this type of leukemia.[6] It works in APL because the majority of cases involve a chromosomal translocation of chromosomes 15 and 17, which causes genetic fusion of the retinoic acid receptor (RAR) gene to the promyelocytic leukemia (PML) gene. This fusion PML-RAR protein is responsible for preventing immature myeloid cells from differentiating into more mature cells. This block in differentiation is thought to cause leukemia. ATRA acts on PML-RAR to lift this block, causing the immature promyelocytes to differentiate to normal mature blood cells thus decreasing promyelocytes.
Side effects
In dermatological use
When used, dryness or increased sensitivity of the affected skin may occur. More sensitive of patients may also experience redness, scaling, itching, and burning. A gradual increase in the frequency and amount of tretinoin application is best, as this allows one's skin to adequately adjust to the drug. Patients should be careful to follow their physician's recommendations when beginning a round of treatment.
This product increases the risk of extreme sunburn; care should be taken (shade, sunscreen, etc.) to protect treated skin from overexposure to ultraviolet light.
Because usage of tretinoin may cause thinning of the skin, it is strongly recommended that patients who are using the drug abstain from hair removal waxing. The wax will, when removed, pull off the top level of epithelium (skin) with it, leaving a red, inflamed, sore mark for several days. Tweezing or threading (epilation) is a viable option for hair removal. The recommended timeframe to wait for a waxing treatment after using tretinoin varies from source to source; anywhere from 5 days to 3 months have been reported. Patients should consult with their aesthetician and dermatologist to discuss the best hair removal options during or after tretinoin use.
In leukemia use
There is a unique complication of retinoic acid syndrome in patients with acute promyelocytic leukemia. This is associated with the development of dyspnea, fever, weight gain, peripheral edema and is treated with dexamethasone. The etiology of retinoic acid syndrome has been attributed to capillary leak syndrome from cytokine release from the differentiating promyelocytes.
Teratogenicity
It is a teratogen, and therefore can cause birth defects and tests have shown increases in fetal skull abnormalities in rats. Women who are or may be pregnant, or who are seeking to become pregnant, are therefore warned against using it.[7] This teratogenic effect is caused by the interference of the exogenous retinoic acid with endogenous retinoic acid signaling, which plays a role in patterning the developing embryo. However the risks of topical tretinoin to the fetus seems to be limited.[8]
Research uses
A study published by the European Respiratory Journal in 2002 suggested that tretinoin can reverse the effects of emphysema in mice by returning elasticity (and regenerating lung tissue through gene mediation) to the alveoli.[9] Studies suggested that this might form a promising treatment in human emphysema patients.[10] However, a newer follow-up study done in 2006 found inconclusive results ("no definitive clinical benefits") using Vitamin A (retinoic acid) in treatment of emphysema in humans and stated that further research is needed to reach conclusions on this treatment.[11]
See also
- Baldness treatments
- Hypervitaminosis A syndrome
- Talarozole, an experimental drug potentiating the effects of tretinoin
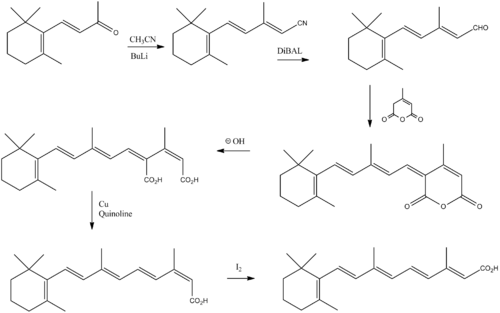
Manufacture
Lewin, Anita H.; Whaley, M. Glenn; Parker, Steven R.; Carroll, F. Ivy; Moreland, Charles G. (1982). "12-Carboxyretinoic acids. Synthesis and structure". The Journal of Organic Chemistry 47 (10): 1799. doi:10.1021/jo00349a001.
Footnotes
- ^ MedlinePlus Drug Information: Tretinoin Topical
- ^ Stefanaki C, Stratigos A, Katsambas A (June 2005). "Topical retinoids in the treatment of photoaging". J Cosmet Dermatol 4 (2): 130–4. doi:10.1111/j.1473-2165.2005.40215.x. PMID 17166212.
- ^ Arthur W. Perry (2007). Straight talk about cosmetic surgery. Yale University Press. pp. 63. ISBN 978-0300121049.
- ^ Huang M, Ye Y, Chen S, Chai J, Lu J, Zhoa L, Gu L, Wang Z (1988). "Use of all-trans retinoic acid in the treatment of acute promyelocytic leukemia" (PDF). Blood 72 (2): 567–72. PMID 3165295. http://www.bloodjournal.org/cgi/reprint/72/2/567.pdf. - Abstract
- ^ Castaigne S, Chomienne C, Daniel M, Ballerini P, Berger R, Fenaux P, Degos L (1990). "All-trans retinoic acid as a differentiation therapy for acute promyelocytic leukemia. I. Clinical results" (PDF). Blood 76 (9): 1704–9. PMID 2224119. http://www.bloodjournal.org/cgi/reprint/76/9/1704.pdf.
- ^ Sanz M (2006). "Treatment of acute promyelocytic leukemia". Hematology Am Soc Hematol Educ Program 2006 (1): 147–55. doi:10.1182/asheducation-2006.1.147. PMID 17124054. http://www.asheducationbook.org/cgi/content/full/2006/1/147.
- ^ Tretinoin facts and comparisons at Drugs.com
- ^ Loureiro KD, Kao KK, Jones KL, Alvarado S, Chavez C, Dick L, Felix R, Johnson D, Chambers CD (July 2005). "Minor malformations characteristic of the retinoic acid embryopathy and other birth outcomes in children of women exposed to topical tretinoin during early pregnancy". Am J Med Genet A 136 (2): 117–21. doi:10.1002/ajmg.a.30744. PMID 15940677.
- ^ "Vitamin may cure smoking disease". BBC News. December 22, 2003. http://news.bbc.co.uk/2/hi/health/3329103.stm. Retrieved 2006-11-18.
- ^ Mao J, Goldin J, Dermand J, Ibrahim G, Brown M, Emerick A, McNitt-Gray M, Gjertson D, Estrada F, Tashkin D, Roth M (1 March 2002). "A pilot study of all-trans-retinoic acid for the treatment of human emphysema". Am J Respir Crit Care Med 165 (5): 718–23. PMID 11874821. http://ajrccm.atsjournals.org/cgi/content/full/165/5/718.
- ^ Roth M, Connett J, D'Armiento J, Foronjy R, Friedman P, Goldin J, Louis T, Mao J, Muindi J, O'Connor G, Ramsdell J, Ries A, Scharf S, Schluger N, Sciurba F, Skeans M, Walter R, Wendt C, Wise R (2006). "Feasibility of retinoids for the treatment of emphysema study". Chest 130 (5): 1334–45. doi:10.1378/chest.130.5.1334. PMID 17099008. http://www.chestjournal.org/cgi/content/full/130/5/1334.
External links
- BASF Technical Information
- "Prescription Medications for Treating Acne". American Academy of Dermatology. http://www.skincarephysicians.com/acnenet/prescriptmeds.html. Retrieved 2006-07-15.
Acne-treating agents (D10) Antibacterial Keratolytic Anti-inflammatory Antibiotics Hormonal Retinoids Combinations Adapalene/benzoyl peroxide • Benzoyl peroxide/clindamycin • Clindamycin/tretinoin • Erythromycin/isotretinoin • Sulfacetamide/sulfurVitamins (A11) Fat soluble D2 (Ergosterol, Ergocalciferol#) · D3 (7-Dehydrocholesterol, Previtamin D3, Cholecalciferol, 25-hydroxycholecalciferol, Calcitriol (1,25-dihydroxycholecalciferol), Calcitroic acid) · D4 (Dihydroergocalciferol) · D5 · D analogues (Dihydrotachysterol, Calcipotriol, Tacalcitol, Paricalcitol)Water soluble B1 (Thiamine#) · B2 (Riboflavin#) · B3 (Niacin, Nicotinamide#) · B5 (Pantothenic acid, Dexpanthenol, Pantethine) · B6 (Pyridoxine#, Pyridoxal phosphate, Pyridoxamine) · B7 (Biotin) · B9 (Folic acid, Dihydrofolic acid, Folinic acid) · B12 (Cyanocobalamin, Hydroxocobalamin, Methylcobalamin, Cobamamide) · CholineCombinations M: NUT
cof, enz, met
noco, nuvi, sysi/epon, met
drug(A8/11/12)
Carotenes (C40) α-Carotene · β-Carotene · γ-Carotene · δ-Carotene · ε-Carotene · ζ-Carotene · Lycopene · Neurosporene · Phytoene · PhytoflueneXanthophylls (C40) Antheraxanthin · Astaxanthin · Canthaxanthin · Citranaxanthin · Cryptoxanthin · Diadinoxanthin · Diatoxanthin · Dinoxanthin · Flavoxanthin · Fucoxanthin · Lutein · Neoxanthin · Rhodoxanthin · Rubixanthin · Violaxanthin · ZeaxanthinApocarotenoids (C<40) Vitamin A retinoids (C20) Retinoid drugs Acitretin · Alitretinoin · Bexarotene · Etretinate · Fenretinide · Isotretinoin · Tazarotene · TretinoinCategories:- Anti-acne preparations
- Antineoplastic drugs
- Carboxylic acids
- Carotenoids
- Retinoids
Wikimedia Foundation. 2010.