- Erythromycin
-
Erythromycin 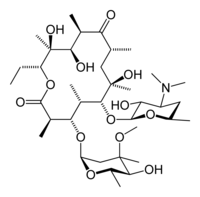

Systematic (IUPAC) name (3R,4S,5S,6R,7R,9R,11R,12R,13S,14R)-6-
{[(2S,3R,4S,6R)-4-(dimethylamino)-3-hydroxy-6-methyloxan-2-yl]oxy}-
14-ethyl-7,12,13-trihydroxy-4-{[(2R,4R,5S,6S)-
5-hydroxy-4-methoxy-4,6-dimethyloxan-2-yl]oxy}-
3,5,7,9,11,13-hexamethyl-1-oxacyclotetradecane-2,10-dioneClinical data Trade names E-mycin, Erythrocin AHFS/Drugs.com monograph MedlinePlus a682381 Pregnancy cat. A(AU) B(US) Legal status Prescription Only (S4) (AU) POM (UK) ℞-only (US) Routes oral, iv, im, topical, ophthalmic eye drops Pharmacokinetic data Bioavailability Depends on the ester type between 30% - 65% Protein binding 90% Metabolism liver (under 5% excreted unchanged) Half-life 1.5 hours Excretion bile Identifiers CAS number 114-07-8 
ATC code D10AF02 J01FA01 S01AA17 QJ51FA01 PubChem CID 3255 DrugBank DB00199 ChemSpider 12041 
UNII 63937KV33D 
KEGG D00140 
ChEBI CHEBI:42355 
ChEMBL CHEMBL532 
Chemical data Formula C37H67NO13 Mol. mass 733.93 g/mol SMILES eMolecules & PubChem - InChI=1S/C37H67NO13/c1-14-25-37(10,45)30(41)20(4)27(39)18(2)16-35(8,44)32(51-34-28(40)24(38(11)12)15-19(3)47-34)21(5)29(22(6)33(43)49-25)50-26-17-36(9,46-13)31(42)23(7)48-26/h18-26,28-32,34,40-42,44-45H,14-17H2,1-13H3/t18-,19-,20+,21+,22-,23+,24+,25-,26+,28-,29+,30-,31+,32-,34+,35-,36-,37-/m1/s1

Key:ULGZDMOVFRHVEP-RWJQBGPGSA-N
 (what is this?) (verify)
(what is this?) (verify)Erythromycin is a macrolide antibiotic that has an antimicrobial spectrum similar to or slightly wider than that of penicillin, and is often used for people who have an allergy to penicillins. For respiratory tract infections, it has better coverage of atypical organisms, including mycoplasma and Legionellosis. It was first marketed by Eli Lilly and Company, and it is today commonly known as EES (erythromycin ethylsuccinate, an ester prodrug that is commonly administered).
In structure, this macrocyclic compound contains a 14-membered lactone ring with ten asymmetric centers and two sugars (L-cladinose and D-desosamine), making it a compound very difficult to produce via synthetic methods.
Erythromycin is produced from a strain of the actinomycete Saccharopolyspora erythraea.
Contents
History
Abelardo Aguilar, a Filipino scientist, sent some soil samples to his employer Eli Lilly in 1949. Eli Lilly’s research team, led by J. M. McGuire, managed to isolate Erythromycin from the metabolic products of a strain of Streptomyces erythreus (designation changed to "Saccharopolyspora erythraea") found in the samples.
Lilly filed for patent protection of the compound and U.S. patent 2,653,899 was granted in 1953. The product was launched commercially in 1952 under the brand name Ilosone (after the Philippine region of Iloilo where it was originally collected from). Erythromycin was formerly also called Ilotycin.
In 1981, Nobel laureate (1965 in chemistry) and Professor of Chemistry at Harvard University (Cambridge, MA) Robert B. Woodward, along with a large number of members from his research group, posthumously reported the first stereocontrolled asymmetric chemical synthesis of Erythromycin A.
The antibiotic clarithromycin was invented by scientists at the Japanese drug company Taisho Pharmaceutical in the 1970s as a result of their efforts to overcome the acid instability of erythromycin.
Scientists at Chugai Pharmaceuticals discovered an erythromycin-derived motilin agonist called Mitemcinal that is believed to have strong prokinetic properties (similar to erythromycin,) but lacking antibiotic properties. At the present time, erythromycin is commonly used off-label for gastric motility indications such as gastroparesis. If Mitemcinal can be shown to be as effective a prokinetic agent, it would represent a significant advance in the GI field as treatment with this drug would not carry the risk of unintentional selection for antibiotic-resistant bacteria.
Synthesis
Over the years since the discovery of erythromycin A and its activity as an antimicrobial, many attempts have been made to synthesize it in the laboratory. However, the presence of ten stereospecific carbons and several points of distinct substitution has made the total synthesis of erythromycin A a formidable task.[1] Complete syntheses of erythromycins’ related structures and precursors such as 6- deoxyerythronolide B have been accomplished, giving way to possible syntheses of different erythromycins and other macrolide antimicrobials.[2] However, Woodward did successfully complete the synthesis of erythromycin A.[3][4][5] This total synthesis begins with (7) and (8). After being coupled, the resulting structure is subjected to a series of reactions, including hydrolysis and stereospecific aldolization. The resulting pure enone is then converted to the desired dithiadecalin product (9) through a series of reduction and oxidation reactions. (9) is then converted to both a ketone (10) and an aldehyde (11).

Figure 1 a)
NaH, THF, Me2SO; b)AcOH, H2O; c)MsCL, Py; d)alumina, EtOAc; e)NaBH4, MeOH; f)MeOCH2I, KH, THF; g)OsO4, ether; NaHSO3, Py(aq); h)Me2C(OMe)2, TsOH, CH2Cl2; i)CF3COOH, CH2Cl2; j)(CF3CO)2O, Me2SO, CH2Cl2; (i-Pr)2NEt; k)Ra(Ni)-(W-2), EtOH, reflux; l)o-NO2C6H4SeCN, P(n-Bu)3, THF; m)O3, MeOH, CH2Cl2; Me2S, NaHCO3.
With these two species, each of which resembling key segments of the erythronolide A seco acid, an aldol condensation is carried out to yield (12). (12) is put through several reactions, including the addition of benzyl thiol, the coupling of enolates, and stereospecific reduction to yield (13), which contains the carbon skeleton and stereocenters of the erythronolide A seco acid.

Figure 2 a)
Mesityllithium, THF; b)(CF3CO)2O, Me2SO, CH2Cl2; (i-Pr)2NEt; c), KH, HMPA, THF; AcCl; d)NaBH4, MeOH, CH2Cl2; e)MsCl, Py; DMAP, Py, MeOH; f)PhCH2SH, n-BuLi, THF; g)LAH, ether; h)Ac2O, DMAP, CH2Cl2; i)Ra(Ni)-(W-2), EtOH, DMF, reflux; j)o-NO2C6H4SeCN, P(n- Bu)3, THF; 30% H2O2, THF; k)O3, MeOH, CH2Cl2; Me2S, NaHCO3; l)EtCOSCMe3, LDA, THF; m)t-BuLi, (CH2NMe2)2, THF; AcOH.
(13) is then subjected to a series of reactions including successive deprotections and acetylization to yield (14), a compound that is more likely to yield a lactone. Acetalization of (14) yields a precursor to the desired thioester (15), which was achieved by replacing the terminal methoxy group with the required thio group. (15) is then lactonized in 70% yield to give (16).

Figure 3 a)
Na2CO3, MeOH; b)(PhOCH2CO)2O, Py, DMAP, CH2Cl2; c)MsCl, Py; d)LiOH, 30% H2O2, THF; e)LiN3, HMPA(aq); f)H2(1 atm), PtO2, THF; g)ClCOOC6H4-p-NO2, CH2Cl2, NaHCO3(aq); h)NH2OH⋅HCl, KH2PO4, MeOH(aq), reflux; i)Et3N, CH2Cl2; j)mesitaldehyde dimethyl acetal, CF3COOH, CH2Cl2; k)EtSLi, HMPA; l)ClCOS-2-Py, Et3N, CH2Cl2.
To complete the synthesis of erythromycin A, (16) is acylated with p-phenylbenzoyl chloride, undergoes hydrolysis, and is deprotected at its hydroxyl groups at carbons 3 and 5 to yield (17). After a series of glycosidation reactions, (17) is converted to (18). (18) is then subjected to a number of reactions, eventually converting carbon 9 from an amine to the ketone, yielding the final erythromycin A product (1).

Figure 4 a)
BPCOCl, Et3N, DMAP, CH2Cl2; b)NaOH(aq), THF, i-PrOH; c)SiO2, CF3COOH(aq), CH2Cl2; d)Na- Hg/MeOH; e)N-chlorosuccinimide, Py; f)AgF, HMPA; g)- H2O.
Total yield for Woodward’s complete synthesis of erythromycin A was approximately 0.02%, leaving future scientists with a clear goal for improvement.
Available forms
Erythromycin is available in enteric-coated tablets, slow-release capsules, oral suspensions, ophthalmic solutions, ointments, gels, and injections.
The following erythromycin combinations are available for oral dosage:[6]
- erythromycin base (capsules, tablets)
- erythromycin estolate (capsules, oral suspension, tablets). This form is contraindicated during pregnancy.[7]
- erythromycin ethylsuccinate (oral suspension, tablets)
- erythromycin stearate (oral suspension, tablets)
For injection the available combinations are:[6]
- erythromycin gluceptate
- erythromycin lactobionate.
Brand names include Robimycin, E-Mycin, E.E.S. Granules, E.E.S.-200, E.E.S.-400, E.E.S.-400 Filmtab, Erymax, Ery-Tab, Eryc, Ranbaxy, Erypar, EryPed, Eryped 200, Eryped 400, Erythrocin Stearate Filmtab, Erythrocot, E-Base, Erythroped, Ilosone, MY-E, Pediamycin, Zineryt, Abboticin, Abboticin-ES, Erycin, PCE Dispertab, Stiemycine, Acnasol and Tiloryth.
Adverse effects
Gastrointestinal disturbances, such as diarrhea, nausea, abdominal pain, and vomiting, are very common because erythromycin is a motilin agonist.[8] Because of this, erythromycin tends not to be prescribed as a first-line drug. However, erythromycin may be useful in treating gastroparesis due to this pro-motility effect. Intravenous erythromycin may also be used in endoscopy as an adjunct to clear gastric contents.
More serious side-effects include arrhythmia with prolonged QTc intervals including Torsades-de-Pointe and reversible deafness. Allergic reactions range from urticaria to anaphylaxis. Cholestasis, Stevens-Johnson syndrome, and toxic epidermal necrolysis are some other rare side-effects that may occur.
Exposure to erythromycin (especially long courses at antimicrobial doses, and also through breastfeeding) has been linked to an increased probability of pyloric stenosis in young infants.[9] Erythromycin used for feeding intolerance in young infants has not been associated with hypertrophic pyloric stenosis.[9]
Erythromycin estolate has been associated with reversible hepatotoxicity in pregnant women in the form of elevated serum glutamic-oxaloacetic transaminase and is not recommended during pregnancy. Some evidence suggests similar hepatotoxicity in other populations.[10]
It can also affect the central nervous system, causing psychotic reactions, nightmares and night sweats.[11]
It may also alter the effectiveness of combined oral contraceptive pills because of its effect on the gut flora. Erythromycin is an inhibitor of the cytochrome P450 system, which means that it can have a rapid effect on levels of other drugs metabolised by this system, e.g., warfarin.
Mechanism of action
Erythromycin displays bacteriocidal activity, especially at higher concentrations,[12] but the mechanism is not fully understood. By binding to the 50s subunit of the bacterial 70s rRNA complex, protein synthesis and subsequent structure and function processes critical for life or replication are inhibited.[12] Erythromycin interferes with aminoacyl translocation, preventing the transfer of the tRNA bound at the A site of the rRNA complex to the P site of the rRNA complex. Without this translocation, the A site remains occupied and, thus, the addition of an incoming tRNA and its attached amino acid to the nascent polypeptide chain is inhibited. This interferes with the production of functionally useful proteins, which is the basis of this antimicrobial action.
Pharmacokinetics
Erythromycin is easily inactivated by gastric acid; therefore, all orally-administered formulations are given as either enteric-coated or more-stable salts or esters, such as erythromycin ethylsuccinate. Erythromycin is very rapidly absorbed, and diffuses into most tissues and phagocytes. Due to the high concentration in phagocytes, erythromycin is actively transported to the site of infection, where, during active phagocytosis, large concentrations of erythromycin are released.
Metabolism
Most of erythromycin is metabolised by demethylation in the liver. Its main elimination route is in the bile. There is little renal excretion. Erythromycin's elimination half-life is 1.5 hours.
Interactions
Erythromycin is metabolized by enzymes of the cytochrome P450 system, in particular, by isozymes of the CYP3A superfamily, CYP3A (Hunt et al., 1992). The activity of the CYP3A enzymes can be induced or inhibited by certain drugs (e.g. dexomethasone) which can cause it to affect the metabolism of many different drugs, e.g. erythromycin. If other CYP3A substrates — drugs that are broken down by CYP3A - such as simvastatin (Zocor), lovastatin (Mevacor), or atorvastatin (Lipitor)—are taken concomitantly with erythromycin, levels of the substrates will increase, often causing adverse effects. A noted drug interaction involves erythromycin and simvastatin, resulting in increased simvastatin levels and the potential for rhabdomyolysis. Another group of CYP3A4 substrates are drugs used for migraine such as ergotamine and dihydroergotamine; their adverse effects may be more pronounced if erythromycin is associated.[11] Earlier case reports on sudden death prompted a study on a large cohort that confirmed a link between erythromycin, ventricular tachycardia, and sudden cardiac death in patients also taking drugs that prolong the metabolism of erythromycin (like verapamil or diltiazem) by interfering with CYP3A4.[13] Hence, erythromycin should not be administered to people using these drugs, or drugs that also prolong the QT interval. Other examples include terfenadine (Seldane, Seldane-D), astemizole (Hismanal), cisapride (Propulsid, withdrawn in many countries for prolonging the QT time) and pimozide (Orap). Theophylline, which is used mostly in asthma, is also contraindicated.
Erythromycin is not recommended when using clindamycin-containing products, even topical products such as Duac or BenzaClin. In general, the simultaneous use two different erythromycin derivatives (such as clindamycin and Mitemcinal) should be avoided as drugs in this macrolide family possess a common mechanism of action.
Erythromycin-derived compounds
- Mitemcinal
- Azithromycin / Zithromax / Zitromax / Sumamed
- Clarithromycin / Biaxin
- Roxithromycin / Rulid / Surlid / Roxid
- Telithromycin
- Dirithromycin / Dynabac
- Cethromycin
- Spiramycin
- Ansamycin
- Oleandomycin
- Carbomycin
- Tylocine
Notes
- ^ Pal, S. A journey across the sequential development of macrolides and ketolides related to erythromycin. Tetrahedron. 2006. 62. 3171-3200.
- ^ Evans, D. A.; Kim, A. S. Synthesis of 6-Deoxyerythronolide B. Implementation of a General Strategy for the Synthesis of Macrolide Antibiotics. Tetrahedron Lett. 1997. 38. 53-56.
- ^ Woodward, R. B.; Logusch, E.; Nambiar, K. P.; Sakan, K.; Ward, D. E.; Au-Yeung, B. -W.; Balaram, P.; Browne, L. J.; Card, P. J.; Chen, C. H.; et al. Asymmetric Total Synthesis of Erythromycin. 1. Synthesis of an Erythronolide A Seco Acid Derivative via Asymmetric Induction. J. Am. Chem. Soc. 1981. 103. 3210-3213.
- ^ Woodward, R. B.; Logusch, E.; Nambiar, K. P.; Sakan, K.; Ward, D. E.; Au-Yeung, B. -W.; Balaram, P.; Browne, L. J.; Card, P. J.; Chen, C. H.; et al. Asymmetric Total Synthesis of Erythromycin. 2. Synthesis of an Erythronolide A Lactone System. J. Am. Chem. Soc. 1981. 103. 3213-3215.
- ^ Woodward, R. B.; Logusch, E.; Nambiar, K. P.; Sakan, K.; Ward, D. E.; Au-Yeung, B. -W.; Balaram, P.; Browne, L. J.; Card, P. J.; Chen, C. H.; et al. Asymmetric Total Synthesis of Erythromycin. 3. Total Synthesis of Erythromycin. J. Am. Chem. Soc. 1981. 103. 3215-3217.
- ^ a b drugs.com > erythromycin (Oral route, Parenteral route) Retrieved on Jan 11, 2010
- ^ Sexually Transmitted Diseases Treatment Guidelines 2006 Centers for Disease Control and Prevention. MMWR 2006;55
- ^ Weber FH Jr, et. al (April 1993). "Erythromycin: a motilin agonist and gastrointestinal prokinetic agent". American Journal of Gastroenterology 88 (4): 485–90. PMID 8470625.
- ^ a b Maheshwai N (March 2007). "Are young infants treated with erythromycin at risk for developing hypertrophic pyloric stenosis?". Arch. Dis. Child. 92 (3): 271–3. doi:10.1136/adc.2006.110007. PMC 2083424. PMID 17337692. http://adc.bmj.com/cgi/pmidlookup?view=long&pmid=17337692.
- ^ McCormack WM, George H, Donner A, Kodgis LF, Albert S, Lowe EW, Kass EH. Hepatotoxicity of erythromycin estoate during pregnancy. Antimicrob Agents Chemother 1997;12:630-5.
- ^ a b Erythromycin. Belgian Center for Pharmacotherapeutical Information. Retrieved July 20, 2008.
- ^ a b Katzung PHARMACOLOGY, 9e Section VIII. Chemotherapeutic Drugs Chapter 44. Chloramphenicol, Tetracyclines, Macrolides, Clindamycin, & Streptogramins
- ^ Ray WA, Murray KT, Meredith S, Narasimhulu SS, Hall K, Stein CM. Oral Erythromycin and the Risk of Sudden Death from Cardiac Causes. N Engl J Med 2004;351:1089-96.
References
- British National Formulary "BNF 49" March 2005.
- Mims C, Dockrell HM, Goering RV, Roitt I, Wakelin D, Zuckerman M. Chapter 33: Attacking the Enemy: Antimicrobial Agents and Chemotherapy: Macrolides. In: Medical Microbiology (3rd Edition). London: Mosby Ltd; 2004. p 489
- Hunt,Ch.M., Watkins, P.B., Saenger, P., Stave, G.M., Barlascini, N., Watlington, Ch.O., Wright, J.T., Guzelian, P.S., (1992) Heterogeneity of CYP3A isoforms metabolizing erythromycin and cortisol. Clinical Pharmacology and Therapeutics 51: 18-23
External links
- U.S. Patent 2,653,899
- E.E.S. (Erythromycin Ethylsuccinate) Drug Information: Uses, Side Effects, Drug Interactions and Warnings at RxList
Acne-treating agents (D10) Antibacterial Keratolytic Anti-inflammatory Antibiotics Hormonal Retinoids Combinations Adapalene/benzoyl peroxide • Benzoyl peroxide/clindamycin • Clindamycin/tretinoin • Erythromycin/isotretinoin • Sulfacetamide/sulfurAntibacterials: protein synthesis inhibitors (J01A, J01B, J01F, J01G, QJ01XQ) 30S -mycin (Streptomyces)Neomycin# (Framycetin, Paromomycin, Ribostamycin)
Kanamycin# (Amikacin, Arbekacin, Bekanamycin, Dibekacin, Tobramycin)
Paromomycin-micin (Micromonospora)Tetracyclines50S Linezolid • Torezolid • Eperezolid • Posizolid • RadezolidPleuromutilinsRetapamulin • Tiamulin • ValnemulinErythromycin# • Azithromycin# • Spiramycin • Midecamycin • Oleandomycin • Roxithromycin • Josamycin • Troleandomycin • Clarithromycin • Miocamycin • Rokitamycin • Dirithromycin • Flurithromycin • Ketolide (Telithromycin, Cethromycin, Solithromycin)EF-G Steroid antibacterialsCategories:- Alcohols
- Amines
- Eli Lilly and Company
- Ethers
- Ketones
- Macrolide antibiotics
- World Health Organization essential medicines
- InChI=1S/C37H67NO13/c1-14-25-37(10,45)30(41)20(4)27(39)18(2)16-35(8,44)32(51-34-28(40)24(38(11)12)15-19(3)47-34)21(5)29(22(6)33(43)49-25)50-26-17-36(9,46-13)31(42)23(7)48-26/h18-26,28-32,34,40-42,44-45H,14-17H2,1-13H3/t18-,19-,20+,21+,22-,23+,24+,25-,26+,28-,29+,30-,31+,32-,34+,35-,36-,37-/m1/s1
Wikimedia Foundation. 2010.

