- Tuberculosis
-
Tuberculosis Classification and external resources 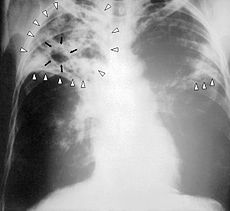
Chest X-ray of a person with advanced tuberculosisICD-10 A15–A19 ICD-9 010–018 OMIM 607948 DiseasesDB 8515 MedlinePlus 000077 000624 eMedicine med/2324 emerg/618 radio/411 MeSH D014376 Tuberculosis, MTB, or TB (short for tubercle bacillus) is a common and in many cases lethal infectious disease caused by various strains of mycobacteria, usually Mycobacterium tuberculosis.[1] Tuberculosis usually attacks the lungs but can also affect other parts of the body. It is spread through the air when people who have an active MTB infection cough, sneeze, or otherwise transmit their saliva through the air.[2] Most infections in humans result in an asymptomatic, latent infection, and about one in ten latent infections eventually progresses to active disease, which, if left untreated, kills more than 50% of those infected.
The classic symptoms are a chronic cough with blood-tinged sputum, fever, night sweats, and weight loss (the last giving rise to the formerly prevalent colloquial term "consumption"). Infection of other organs causes a wide range of symptoms. Diagnosis relies on radiology (commonly chest X-rays), a tuberculin skin test, blood tests, as well as microscopic examination and microbiological culture of bodily fluids. Treatment is difficult and requires long courses of multiple antibiotics. Social contacts are also screened and treated if necessary. Antibiotic resistance is a growing problem in (extensively) multi-drug-resistant tuberculosis. Prevention relies on screening programs and vaccination, usually with Bacillus Calmette-Guérin vaccine.
One third of the world's population is thought to be infected with M. tuberculosis,[3][4] and new infections occur at a rate of about one per second.[3] The proportion of people who become sick with tuberculosis each year is stable or falling worldwide but, because of population growth, the absolute number of new cases is still increasing.[3] In 2007 there were an estimated 13.7 million chronic active cases, 9.3 million new cases, and 1.8 million deaths, mostly in developing countries.[5] In addition, more people in the developed world contract tuberculosis because their immune systems are more likely to be compromised due to higher exposure to immunosuppressive drugs, substance abuse, or AIDS. The distribution of tuberculosis is not uniform across the globe; about 80% of the population in many Asian and African countries test positive in tuberculin tests, while only 5–10% of the US population test positive.[1]
Contents
Signs and symptoms
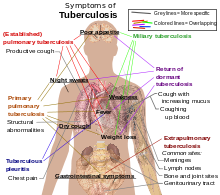 Main symptoms of variants and stages of tuberculosis,[6] with many symptoms overlapping with other variants, while others are more (but not entirely) specific for certain variants. Multiple variants may be present simultaneously.
Main symptoms of variants and stages of tuberculosis,[6] with many symptoms overlapping with other variants, while others are more (but not entirely) specific for certain variants. Multiple variants may be present simultaneously.
When tuberculosis becomes active, 75% of cases involve infection in the lungs (pulmonary TB). Symptoms include chest pain, coughing up blood, and a productive, prolonged cough for more than three weeks. Systemic symptoms include fever, chills, night sweats, appetite loss, weight loss, pallor, and fatigue.[7]
In the other 25% of active cases, the infection moves from the lungs, causing other kinds of TB, collectively denoted extrapulmonary tuberculosis.[8] This occurs more commonly in immunosuppressed persons and young children. Extrapulmonary infection sites include the pleura in tuberculous pleurisy, the central nervous system in meningitis, the lymphatic system in scrofula of the neck, the genitourinary system in urogenital tuberculosis, and the bones and joints in Pott's disease of the spine. When spread to the bones it is also known as "osseous tuberculosis",[9] a form of Osteomyelitis (as a complication of tuberculosis[1]). An especially serious form is disseminated TB, more commonly known as miliary tuberculosis. Extrapulmonary TB may co-exist with pulmonary TB.[10]
The radiological findings of tuberculosis are well described and chest x-ray plays an important role in its diagnosis.
Causes
Main article: Mycobacterium tuberculosis Scanning electron micrograph of Mycobacterium tuberculosis
Scanning electron micrograph of Mycobacterium tuberculosis
The main cause of TB, Mycobacterium tuberculosis (MTB), is a small aerobic non-motile bacillus. High lipid content of this pathogen accounts for many of its unique clinical characteristics.[11] It divides every 16 to 20 hours, an extremely slow rate compared with other bacteria, which usually divide in less than an hour.[12] Since MTB has a cell wall but lacks a phospholipid outer membrane, it is classified as a Gram-positive bacterium. However, if a Gram stain is performed, MTB either stains very weakly Gram-positive or does not retain dye as a result of the high lipid and mycolic acid content of its cell wall.[13] MTB can withstand weak disinfectants and survive in a dry state for weeks. In nature, the bacterium can grow only within the cells of a host organism, but M. tuberculosis can be cultured in vitro.[14]
Using histological stains on expectorate samples from phlegm (also called sputum), scientists can identify MTB under a regular microscope. Since MTB retains certain stains after being treated with acidic solution, it is classified as an acid-fast bacillus (AFB).[1][13] The most common acid-fast staining technique, the Ziehl-Neelsen stain, dyes AFBs a bright red that stands out clearly against a blue background. Other ways to visualize AFBs include an auramine-rhodamine stain and fluorescent microscopy.
The M. tuberculosis complex includes four other TB-causing mycobacteria: M. bovis, M. africanum, M. canetti, and M. microti.[15] M. africanum is not widespread, but in parts of Africa it is a significant cause of tuberculosis.[16][17] M. bovis was once a common cause of tuberculosis, but the introduction of pasteurized milk has largely eliminated this as a public health problem in developed countries.[1][18] M. canetti is rare and seems to be limited to Africa, although a few cases have been seen in African emigrants.[19] M. microti is mostly seen in immunodeficient people, although it is possible that the prevalence of this pathogen has been underestimated.[20]
Other known pathogenic mycobacteria include Mycobacterium leprae, Mycobacterium avium, and M. kansasii. The latter two are part of the nontuberculous mycobacteria (NTM) group. Nontuberculous mycobacteria cause neither TB nor leprosy, but they do cause pulmonary diseases resembling TB.[21]
Risk factors
People with silicosis have an approximately 30-fold greater risk for developing TB.[22] Silica particles irritate the respiratory system, causing immunogenic responses such as phagocytosis, which results in high lymphatic vessel deposits.[23] It is probably this interference and blockage of macrophage function that increases the risk of tuberculosis.[24] Persons with chronic renal failure and also on hemodialysis have an increased risk.[25] Persons with diabetes mellitus have a risk for developing active TB that is two to four times greater than persons without diabetes mellitus, and this risk is likely to be greater in persons with insulin-dependent or poorly controlled diabetes.[26] Other clinical conditions that have been associated with active TB include gastrectomy with attendant weight loss and malabsorption, jejunoileal bypass, renal and cardiac transplantation, carcinoma of the head or neck, and other neoplasms (e.g., lung cancer, lymphoma, and leukemia).[22]
Given that silicosis greatly increases the risk of tuberculosis, more research about the effect of various indoor or outdoor air pollutants on the disease would be necessary. Some possible indoor sources of silica include paint, concrete, and Portland cement. Crystalline silica is found in concrete, masonry, sandstone, rock, paint, and other abrasives. The cutting, breaking, crushing, drilling, grinding, or abrasive blasting of these materials may produce fine silica dust. It can also be in soil, mortar, plaster, and shingles.[27]
Low body weight is associated with risk of tuberculosis as well. A body mass index (BMI) below 18.5 increases the risk by 2 to 3 times. An increase in body weight lowers the risk.[28] People with diabetes mellitus are at increased risk of contracting tuberculosis,[29] and they have a poorer response to treatment, possibly due to poorer drug absorption.[30]
Diabetes increases the risk of TB three-fold.[31] The correlation between diabetes mellitus and TB is concerning for public health because it shows a distinct connection between a contagious disease and a chronic disease. TB is a highly contagious air-born bacteria. Therefore, contracting tuberculosis depends on whether or not a person comes into contact with the bacteria. Diabetics do not have an increased risk of contracting latent tuberculosis but studies have shown that people with diabetes mellitus are more likely to move from a latent form of TB to an active form of TB. This is where the public concern comes from, because when TB is active it is contagious and potentially fatal.[32]
Other conditions that increase risk include the sharing of needles among IV drug users, recent TB infection or a history of inadequately treated TB, chest X-ray suggestive of previous TB, showing fibrotic lesions and nodules, prolonged corticosteroid therapy and other immunosuppressive therapy, compromised immune system (30–40% of people with AIDS worldwide also have TB), hematologic and reticuloendothelial diseases, such as leukemia and Hodgkin's disease, end-stage kidney disease, intestinal bypass, chronic malabsorption syndromes, vitamin D deficiency,[33] and low body weight.[1][10]
Twin studies in the 1940s showed that susceptibility to TB was heritable. If one of a pair of twins got TB, then the other was more likely to get TB if he was identical than if he was not.[34] These findings were more recently confirmed by a series of studies in South Africa.[35][36][37] Specific gene polymorphisms in IL12B have been linked to tuberculosis susceptibility.[38]
Some drugs, including rheumatoid arthritis drugs that work by blocking tumor necrosis factor-alpha (an inflammation-causing cytokine), raise the risk of activating a latent infection due to the importance of this cytokine in the immune defense against TB.[39]
Mechanism
Transmission
When people suffering from active pulmonary TB cough, sneeze, speak, sing, or spit, they expel infectious aerosol droplets 0.5 to 5 µm in diameter. A single sneeze can release up to 40,000 droplets.[40] Each one of these droplets may transmit the disease, since the infectious dose of tuberculosis is very low and inhaling fewer than ten bacteria may cause an infection.[41][42]
People with prolonged, frequent, or intense contact are at particularly high risk of becoming infected, with an estimated 22% infection rate. A person with active but untreated tuberculosis can infect 10–15 other people per year.[3] Others at risk include people in areas where TB is common, people who inject illicit drugs, residents and employees of high-risk congregate settings, medically under-served and low-income populations, high-risk racial or ethnic minority populations, children exposed to adults in high-risk categories, those who are immunocompromised by conditions such as HIV/AIDS, people who take immunosuppressant drugs, and health care workers serving these high-risk clients.[43]
Transmission can only occur from people with active—not latent—TB.[1] The probability of transmission from one person to another depends upon the number of infectious droplets expelled by a carrier, the effectiveness of ventilation, the duration of exposure, and the virulence of the M. tuberculosis strain.[10] The chain of transmission can be broken by isolating people with active disease and starting effective anti-tuberculous therapy. After two weeks of such treatment, people with non-resistant active TB generally cease to be contagious. If someone does become infected, then it will take three to four weeks before the newly infected person can transmit the disease to others.[44]
Pathogenesis
About 90% of those infected with Mycobacterium tuberculosis have asymptomatic, latent TB infection (sometimes called LTBI), with only a 10% lifetime chance that a latent infection will progress to TB disease.[1] However, if untreated, the death rate for these active TB cases is more than 50%.[3]
TB infection begins when the mycobacteria reach the pulmonary alveoli, where they invade and replicate within the endosomes of alveolar macrophages.[1][45] The primary site of infection in the lungs is called the Ghon focus, and is generally located in either the upper part of the lower lobe, or the lower part of the upper lobe.[1] Simon foci may also be present. Bacteria are picked up by dendritic cells, which do not allow replication, although these cells can transport the bacilli to local (mediastinal) lymph nodes. Further spread is through the bloodstream to other tissues and organs where secondary TB lesions can develop in other parts of the lung (particularly the apex of the upper lobes), peripheral lymph nodes, kidneys, brain, and bone.[1][46] All parts of the body can be affected by the disease, though it rarely affects the heart, skeletal muscles, pancreas and thyroid.[47]
Tuberculosis is classified as one of the granulomatous inflammatory conditions. Macrophages, T lymphocytes, B lymphocytes, and fibroblasts are among the cells that aggregate to form granulomas, with lymphocytes surrounding the infected macrophages. The granuloma prevents dissemination of the mycobacteria and provides a local environment for interaction of cells of the immune system. Bacteria inside the granuloma can become dormant, resulting in a latent infection. Another feature of the granulomas of human tuberculosis is the development of abnormal cell death (necrosis) in the center of tubercles. To the naked eye this has the texture of soft white cheese and is termed caseous necrosis.[48]
If TB bacteria gain entry to the bloodstream from an area of damaged tissue they spread through the body and set up many foci of infection, all appearing as tiny white tubercles in the tissues. This severe form of TB disease is most common in infants and the elderly and is called miliary tuberculosis. People with this disseminated TB have a fatality rate near 100% if untreated. However, if treated early, the fatality rate is reduced to about 10%.[49]
In many people the infection waxes and wanes. Tissue destruction and necrosis are balanced by healing and fibrosis.[48] Affected tissue is replaced by scarring and cavities filled with cheese-like white necrotic material. During active disease, some of these cavities are joined to the air passages bronchi and this material can be coughed up. It contains living bacteria and can therefore spread the infection. Treatment with appropriate antibiotics kills bacteria and allows healing to take place. Upon cure, affected areas are eventually replaced by scar tissue.[48]
If untreated, infection with Mycobacterium tuberculosis can cause lobar pneumonia.[50]
Diagnosis
Main article: Tuberculosis diagnosisSee also: Tuberculosis classification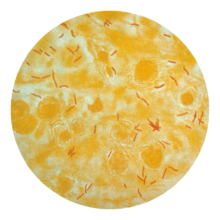 Mycobacterium tuberculosis (stained red) in sputum
Mycobacterium tuberculosis (stained red) in sputum
Tuberculosis is diagnosed definitively by identifying the causative organism (Mycobacterium tuberculosis) in a clinical sample (for example, sputum or pus). When this is not possible, a probable—although sometimes inconclusive[2]—diagnosis may be made using imaging (X-rays or scans), a tuberculin skin test (Mantoux test),[2] or a, Interferon Gamma Release Assay (IGRA).
The main problem with tuberculosis diagnosis is the difficulty in culturing this slow-growing organism in the laboratory (it may take 4 to 12 weeks for blood or sputum culture). A complete medical evaluation for TB must include a medical history, a physical examination, a chest X-ray, microbiological smears, and cultures. It may also include a tuberculin skin test, a serological test. The interpretation of the tuberculin skin test depends upon the person's risk factors for infection and progression to TB disease, such as exposure to other cases of TB or immunosuppression.[10]
Currently, latent infection is diagnosed in a non-immunized person by a tuberculin skin test, which yields a delayed hypersensitivity type response to an extract made from M. tuberculosis.[1] Those immunized for TB or with past-cleared infection will respond with delayed hypersensitivity parallel to those currently in a state of infection, so the test must be used with caution, particularly with regard to persons from countries where TB immunization is common.[51] Tuberculin tests have the disadvantage of producing false negatives, especially when the person is co-morbid with sarcoidosis, Hodgkins lymphoma, malnutrition, or most notably active tuberculosis disease.[1] The newer interferon release assays (IGRAs) such as T-SPOT.TB and QuantiFERON-TB Gold In Tube overcome many of these problems. IGRAs are in vitro blood tests that are more specific than the skin test. IGRAs detect the release of interferon gamma in response to mycobacterial proteins such as ESAT-6.[52] These are not affected by immunization or environmental mycobacteria, so generate fewer false positive results.[53] There is also evidence that IGRAs are more sensitive than the skin test.[54]
New TB tests have been developed that are fast and accurate. These include polymerase chain reaction assays for the detection of bacterial DNA.[55] One such molecular diagnostics test gives results in 100 minutes and is currently being offered to 116 low- and middle-income countries at a discount with support from WHO and the Bill and Melinda Gates foundation.[56]
Another such test, which was approved by the FDA in 1996, is the amplified mycobacterium tuberculosis direct test (MTD, Gen-Probe). This test yields results in 2.5 to 3.5 hours, and it is highly sensitive and specific when used to test smears positive for acid-fast bacilli (AFB).[57]
Screening
Mantoux tuberculin skin tests are often used for routine screening of high risk individuals.[58]
Prevention
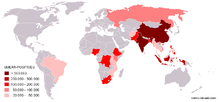 Map showing the 22 high-burden countries (HBC) that according to WHO account for 80% of all new TB cases arising each year. The Global Plan is especially aimed at these countries.
Map showing the 22 high-burden countries (HBC) that according to WHO account for 80% of all new TB cases arising each year. The Global Plan is especially aimed at these countries.
TB prevention and control takes two parallel approaches. In the first, people with TB and their contacts are identified and then treated. Identification of infections often involves testing high-risk groups for TB. In the second approach, children are vaccinated to protect them from TB. No vaccine is available that provides reliable protection for adults. However, in tropical areas where the levels of other species of mycobacteria are high, exposure to nontuberculous mycobacteria gives some protection against TB.[59]
The World Health Organization (WHO) declared TB a global health emergency in 1993, and the Stop TB Partnership developed a Global Plan to Stop Tuberculosis that aims to save 14 million lives between 2006 and 2015.[60] Since humans are the only host of Mycobacterium tuberculosis, eradication would be possible. This goal would be helped greatly by an effective vaccine.[61]
Vaccines
Many countries use the Bacillus Calmette-Guérin (BCG) vaccine as part of their TB control programmes, especially for infants. The BCG vaccine is one of the most widely used of all current vaccines, reaching >80% of neonates and infants in countries with a national vaccination schedule.[62] In the US, where TB is uncommon, BCG is not widely administered.[63] BCG was the first vaccine for TB. From 1905, Albert Calmette and Camille Guérin worked at the Institut Pasteur de Lille and the Pasteur Institute in France developing BCG, administering the first human trials in 1921.[64] However, deaths due to flawed manufacturing processes created public resistance to BCG, delaying mass vaccinations until after World War II.[65] The protective efficacy of BCG for preventing serious forms of TB (e.g. meningitis) in children is greater than 80%. Its protective efficacy for preventing pulmonary TB in adolescents and adults varies by country (as low as 0% in South India); in the United Kingdom, its effectiveness exceeds 75%.[66]
In South Africa, the country with the highest prevalence of TB, BCG is given to all children under age three.[67] However, BCG is less effective in areas where mycobacteria are less prevalent; therefore BCG is not given to the entire population in such countries. In the USA, for example, BCG vaccine is not recommended except for people who meet specific criteria:[10]
- Infants or children with negative skin test results who are continually exposed to untreated or ineffectively treated people or will be continually exposed to multi-drug-resistant tuberculosis (MDR-TB).
- Healthcare workers considered on an individual basis in settings in which a high percentage of MDR-TB has been found, transmission of MDR-TB is likely, and TB control precautions have been implemented and were not successful.
BCG provides some protection against severe forms of pediatric TB, but has been shown to be unreliable against adult pulmonary TB, which accounts for most of the disease burden worldwide. Currently, there are more cases of TB on the planet than at any other time in history and most agree there is an urgent need for a newer, more effective vaccine that would prevent all forms of TB—including drug resistant strains—in all age groups and among people with HIV.[68]
Several new vaccines to prevent TB infection are being developed, among others by Aeras and TBVI. The first recombinant tuberculosis vaccine, Mtb72F, entered clinical trials in the United States in 2004, sponsored by the National Institute of Allergy and Infectious Diseases (NIAID).[69][70] A 2005 study showed that a DNA TB vaccine given with conventional chemotherapy can accelerate the disappearance of bacteria as well as protect against re-infection in mice; it may take four to five years to be available in humans.[71] Another TB vaccine, MVA85A, is currently in phase II trials in South Africa,[72] and is based on a genetically modified vaccinia virus. Many other strategies are also being used to develop novel vaccines,[73] including both subunit vaccines (fusion molecules composed of two recombinant proteins delivered in an adjuvant) such as Hybrid-1, HyVac4, or M72, and recombinant adenoviruses such as Ad35.[74][75][76][77] Some of these vaccines can be effectively administered without needles, making them preferable for areas where HIV is common.[78] All of these vaccines have been successfully tested in humans and are now in extended testing in TB-endemic regions. To encourage further discovery, researchers and policymakers are promoting new economic models of vaccine development including prizes, tax incentives, and advance market commitments.[79][80]
An experimental vaccine, with positive results in mouse models, may be effective in not only preventing infection, but also in eradicating the infection once established.[81] A tuberculosis vaccine aimed at sterile Mtb eradication should be able to target latent Mtb as well as Mtb that causes early-stage tuberculosis.[82] The vaccine is a combination of antigens Ag85B and ESAT-6 as well as the protein Rv2660c. Ag85B and ESAT-6 together form the vaccine Hybrid-1, while Rv2660c is a protein that is expressed even in late-stage infections, when protein transcription is generally reduced. The novel combination of Ag85B, ESAT-6, and Rv2660c allows for both short- and long-term protection as a result of the continued expression of target proteins. The new vaccine, currently referred to as H56, works by promoting a polyfunctional CD4+ T cell response against tuberculosis protein components. Phase I clinical trials are scheduled to begin in Cape Town, South Africa, in March 2011.[81][dated info]
Treatment
Main article: Tuberculosis treatmentTreatment for TB uses antibiotics to kill the bacteria. Effective TB treatment is difficult, due to the unusual structure and chemical composition of the mycobacterial cell wall, which makes many antibiotics ineffective and hinders the entry of drugs.[83][84][85][86] The two antibiotics most commonly used are isoniazid and rifampicin. However, instead of the short course of antibiotics typically used to cure other bacterial infections, TB requires much longer periods of treatment (around 6 to 24 months) to entirely eliminate mycobacteria from the body.[10] Latent TB treatment usually uses a single antibiotic, while active TB disease is best treated with combinations of several antibiotics, to reduce the risk of the bacteria developing antibiotic resistance.[87] People with latent infections are treated to prevent them from progressing to active TB disease later in life.
Drug-resistant tuberculosis is transmitted in the same way as regular TB. Primary resistance occurs in persons infected with a resistant strain of TB. A person with fully susceptible TB develops secondary resistance (acquired resistance) during TB therapy because of inadequate treatment, not taking the prescribed regimen appropriately, or using low-quality medication.[87] Drug-resistant TB is a public health issue in many developing countries, as treatment is longer and requires more expensive drugs. Multi-drug-resistant tuberculosis (MDR-TB) is defined as resistance to the two most effective first-line TB drugs: rifampicin and isoniazid. Extensively drug-resistant TB (XDR-TB) is also resistant to three or more of the six classes of second-line drugs.[88]
The DOTS (Directly Observed Treatment Short-course) strategy of tuberculosis treatment recommended by WHO was based on clinical trials done in the 1970s by the Tuberculosis Research Centre in Chennai, India. The country in which a person with TB lives can determine what treatment they receive. This is because multi-drug-resistant tuberculosis is resistant to most first-line medications, so the use of second-line antituberculosis medications is necessary to cure the person. However, the price of these medications is high; thus, poor people in the developing world have no or limited access to these treatments.[89]
In the early 1900s to 1950s doctors would try to collapse the infected lung by breaking several ribs or inflating that half of the chest with air.[90]
Prognosis
Progression from TB infection to TB disease occurs when the TB bacilli overcome the immune system defenses and begin to multiply. In primary TB disease—1–5% of cases—this occurs soon after infection.[1] However, in the majority of cases, a latent infection occurs that has no obvious symptoms.[1] These dormant bacilli can produce tuberculosis in 2–23% of these latent cases, often many years after infection.[91]
The risk of reactivation increases with immunosuppression, such as that caused by infection with HIV. In people co-infected with M. tuberculosis and HIV, the risk of reactivation increases to 10% per year.[1] Studies utilizing DNA fingerprinting of M. tuberculosis strains have shown that reinfection contributes more substantially to recurrent TB than previously thought,[92] with between 12% and 77% of cases attributable to reinfection (instead of reactivation).[93]
Epidemiology
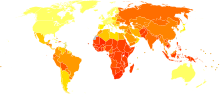 Age-standardized death from tuberculosis per 100,000 inhabitants in 2004.[94]
Age-standardized death from tuberculosis per 100,000 inhabitants in 2004.[94] no data≤ 1010–2525–5050–7575–100100–250250–500500–750750–10001000–20002000–3000≥ 3000
no data≤ 1010–2525–5050–7575–100100–250250–500500–750750–10001000–20002000–3000≥ 3000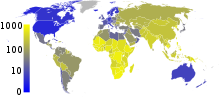 In 2007, the prevalence of TB per 100,000 people was highest in sub-Saharan Africa, and was also relatively high in Asia.[95]
In 2007, the prevalence of TB per 100,000 people was highest in sub-Saharan Africa, and was also relatively high in Asia.[95]
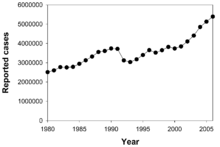 Annual number of new reported TB cases. Data from WHO.[96]
Annual number of new reported TB cases. Data from WHO.[96]
Roughly a third of the world's population has been infected with M. tuberculosis, and new infections occur at a rate of one per second.[3] However, not all infections with M. tuberculosis cause TB disease and many infections are asymptomatic.[97] In 2007, an estimated 13.7 million people had active TB disease, with 9.3 million new cases and 1.8 million deaths; the annual incidence rate varied from 363 per 100,000 in Africa to 32 per 100,000 in the Americas.[5] Tuberculosis is the world's greatest infectious killer of women of reproductive age and the leading cause of death among people with HIV/AIDS.[98]
The rise in HIV infections and the neglect of TB control programs have enabled a resurgence of tuberculosis.[99] The emergence of drug-resistant strains has also contributed to this new epidemic with, from 2000 to 2004, 20% of TB cases being resistant to standard treatments and 2% resistant to second-line drugs.[88] The rate at which new TB cases occur varies widely, even in neighbouring countries, apparently because of differences in health care systems.[100]
In 2007, the country with the highest estimated incidence rate of TB was Swaziland, with 1200 cases per 100,000 people. India had the largest total incidence, with an estimated 2.0 million new cases.[5] In developed countries, tuberculosis is less common and is mainly an urban disease. In the United Kingdom, the national average was 15 per 100,000 in 2007, and the highest incidence rates in Western Europe were 30 per 100,000 in Portugal and Spain. These rates compared with 98 per 100,000 in China and 48 per 100,000 in Brazil. In the United States, the overall tuberculosis case rate was 4 per 100,000 persons in 2007.[95] In Canada, tuberculosis is still endemic in some rural areas.[101]
The incidence of TB varies with age. In Africa, TB primarily affects adolescents and young adults.[102] However, in countries where TB has gone from high to low incidence, such as the United States, TB is mainly a disease of older people, or of the immunocompromised.[1][103]
There are a number of known factors that make people more susceptible to TB infection; worldwide the most important of these is HIV. Co-infection with HIV is a particular problem in Sub-Saharan Africa, due to the high incidence of HIV in these countries.[104][105] Smoking more than 20 cigarettes a day increases the risk of TB by two to four times.[106][107] Diabetes mellitus is also an important risk factor that is growing in importance in developing countries.[108] Other disease states that increase the risk of developing tuberculosis are Hodgkin lymphoma, end-stage renal disease, chronic lung disease, malnutrition, and alcoholism.[1]
Diet may also modulate risk. For example, among immigrants in London from the Indian subcontinent, vegetarian Hindu Asians were found to have an 8.5 fold increased risk of tuberculosis, compared to Muslims who ate meat and fish daily.[109] Although a causal link is not proved by this data,[110] this increased risk could be caused by micronutrient deficiencies: possibly iron, vitamin B12 or vitamin D.[109] Further studies have provided more evidence of a link between vitamin D deficiency and an increased risk of contracting tuberculosis.[111][112] Globally, the severe malnutrition common in parts of the developing world causes a large increase in the risk of developing active tuberculosis, due to its damaging effects on the immune system.[113][114] Along with overcrowding, poor nutrition may contribute to the strong link observed between tuberculosis and poverty.[115][116]
Prisoners are particularly vulnerable to infectious diseases such as HIV/AIDS and TB. Imprisonment facilities provide conditions that allow TB to spread rapidly due to overcrowding, poor nutrition, and a lack of health services. TB outbreaks have been reported in prisons and jails throughout the world, and is particularly concerning in the United States,[117] which incarcerates a larger proportion of its population than any other nation. The prevalence of TB in prisons is much higher than among the general population—in some countries as much as 40 times higher.[118][119]
A new rapid test to identify tuberculosis, combined with aggressive treatment strategies in developing world countries, has resulted in a recent drop in the number of TB cases and deaths.[120] As of 2010, the number of global cases had dropped to 8.8 million, after peaking at 9 million in 2005. The number of deaths had also fallen to 1.4 million in 2010, down from a peak of 1.8 million in 2003.[121]
A 2011 World Health Organisation report showed that if trends continue, all regions except Africa are on track to achieve a 50 percent decline in TB mortality by 2015. African mortality also declined, but at a slower rate due to the complicating factor of widespread HIV. China has achieved particularly dramatic progress, with an 80 percent decline in its TB mortality rate.[120]
History
Main article: History of tuberculosisTuberculosis has been present in humans since antiquity. The earliest unambiguous detection of Mycobacterium tuberculosis is in the remains of bison dated 17,000 years before the present.[122] However, whether tuberculosis originated in cattle and then transferred to humans, or diverged from a common ancestor, is currently unclear.[123] Skeletal remains show prehistoric humans (4000 BCE) had TB, and tubercular decay has been found in the spines of Egyptian mummies from 3000-2400 BCE.[124] Phthisis is a Greek term for consumption; around 460 BCE, Hippocrates identified phthisis as the most widespread disease of the times involving coughing up blood and fever, which was almost always fatal.[125] Genetic studies suggest that TB was present in The Americas from about the year 100 CE.[126]
Before the Industrial Revolution, tuberculosis may sometimes have been regarded as vampirism. When one member of a family died from it, the other members that were infected would lose their health slowly. People believed that this was caused by the original victim draining the life from the other family members. Furthermore, people who had TB exhibited symptoms similar to what people considered to be vampire traits. People with TB often have symptoms such as red, swollen eyes (which also creates a sensitivity to bright light), pale skin and coughing blood, suggesting the idea that the only way for the afflicted to replenish this loss of blood was by sucking blood.[127]
Although it was established that the pulmonary form was associated with 'tubercles' by Dr Richard Morton in 1689,[128][129] due to the variety of its symptoms, TB was not identified as a single disease until the 1820s and was not named 'tuberculosis' until 1839 by J. L. Schönlein.[130] During the years 1838–1845, Dr. John Croghan, the owner of Mammoth Cave, brought a number of tuberculosis sufferers into the cave in the hope of curing the disease with the constant temperature and purity of the cave air: they died within a year.[131] The first TB sanatorium opened in 1859 in Sokołowsko, Poland by Hermann Brehmer.[132]
The bacillus causing tuberculosis, Mycobacterium tuberculosis, was identified and described on March 24, 1882 by Robert Koch. He received the Nobel Prize in physiology or medicine in 1905 for this discovery.[133] Koch did not believe that bovine (cattle) and human tuberculosis were similar, which delayed the recognition of infected milk as a source of infection. Later, this source was eliminated by the pasteurization process. Koch announced a glycerine extract of the tubercle bacilli as a "remedy" for tuberculosis in 1890, calling it 'tuberculin'. It was not effective, but was later adapted as a test for pre-symptomatic tuberculosis.[134]
The first genuine success in immunizing against tuberculosis was developed from attenuated bovine-strain tuberculosis by Albert Calmette and Camille Guerin in 1906. It was called 'BCG' (Bacillus of Calmette and Guerin). The BCG vaccine was first used on humans in 1921 in France,[64] but it wasn't until after World War II that BCG received widespread acceptance in the USA, Great Britain, and Germany.[65]
Tuberculosis caused the most widespread public concern in the 19th and early 20th centuries as an endemic disease of the urban poor. In 1815, one in four deaths in England was of consumption; by 1918 one in six deaths in France were still caused by TB. After the establishment in the 1880s that the disease was contagious, TB was made a notifiable disease in Britain; there were campaigns to stop spitting in public places, and the infected poor were "encouraged" to enter sanatoria that resembled prisons; the sanatoria for the middle and upper classes offered excellent care and constant medical attention.[132] Whatever the purported benefits of the fresh air and labor in the sanatoria, even under the best conditions, 50% of those who entered were dead within five years (1916).[132]
The promotion of Christmas Seals began in Denmark during 1904 as a way to raise money for tuberculosis programs. It expanded to the United States and Canada in 1907–08 to help the National Tuberculosis Association (later called the American Lung Association).
In the United States, concern about the spread of tuberculosis played a role in the movement to prohibit public spitting except into spittoons.
In Europe, deaths from TB fell from 500 out of 100,000 in 1850 to 50 out of 100,000 by 1950. Improvements in public health were reducing tuberculosis even before the arrival of antibiotics, although the disease remained a significant threat to public health, such that when the Medical Research Council was formed in Britain in 1913 its initial focus was tuberculosis research.[135]
It was not until 1946 with the development of the antibiotic streptomycin that effective treatment and cure became possible. Prior to the introduction of this drug, the only treatment besides sanatoria were surgical interventions, including the pneumothorax technique—collapsing an infected lung to "rest" it and allow lesions to heal—a technique that was of little benefit and was largely discontinued by the 1950s.[136] The emergence of multidrug-resistant TB has again introduced surgery as part of the treatment for these infections. Here, surgical removal of chest cavities will reduce the number of bacteria in the lungs, as well as increasing the exposure of the remaining bacteria to drugs in the bloodstream, and is therefore thought increase the effectiveness of the chemotherapy.[137]
Hope that the disease could be completely eliminated have been dashed since the rise of drug-resistant strains in the 1980s. For example, tuberculosis cases in Britain, numbering around 50,000 in 1955, had fallen to around 5,500 in 1987, but in 2000 there were over 7,000 confirmed cases.[citation needed] Due to the elimination of public health facilities in New York and the emergence of HIV, there was a resurgence in the late 1980s.[138] The number of those failing to complete their course of drugs is high. NY had to cope with more than 20,000 "unnecessary" TB-patients with multidrug-resistant strains (resistant to, at least, both Rifampin and Isoniazid). The resurgence of tuberculosis resulted in the declaration of a global health emergency by the World Health Organization in 1993.[139]
Animal infection
Tuberculosis can be carried by mammals; domesticated species, such as cats and dogs, are generally free of tuberculosis, but wild animals may be carriers. In some places, regulations aiming to prevent the spread of TB restrict the ownership of novelty pets; for example, the U.S. state of California forbids the ownership of pet gerbils.[140]
Mycobacterium bovis causes TB in cattle. An effort to eradicate bovine tuberculosis from the cattle and deer herds of New Zealand is under way. It has been found that herd infection is more likely in areas where infected vector species such as Australian brush-tailed possums come into contact with domestic livestock at farm/bush borders.[141] Controlling the vectors through possum eradication and monitoring the level of disease in livestock herds through regular surveillance are seen as a "two-pronged" approach to ridding New Zealand of the disease.
In both the Republic of Ireland and Northern Ireland, badgers have been identified as a vector species for the transmission of bovine tuberculosis. As a result, the government in both regions has mounted an active campaign of eradication of the species in an effort to reduce the incidence of the disease. Badgers have been culled primarily by snaring and gassing. It remains a contentious issue, with proponents and opponents of the scheme citing their own studies to support their position.[142][143][144]
See also
- Mycobacterium Tuberculosis Structural Genomics Consortium
- TBVI The Tuberculosis Vaccine Initiative
References
- ^ a b c d e f g h i j k l m n o p q r Kumar V, Abbas AK, Fausto N, Mitchell RN (2007). Robbins Basic Pathology (8th ed.). Saunders Elsevier. pp. 516–522. ISBN 978-1-4160-2973-1.
- ^ a b c Konstantinos A (2010). "Testing for tuberculosis". Australian Prescriber 33 (1): 12–18. http://www.australianprescriber.com/magazine/33/1/12/18/.
- ^ a b c d e f "Tuberculosis Fact sheet N°104". World Health Organization. November 2010. http://www.who.int/mediacentre/factsheets/fs104/en/index.html. Retrieved 26 July 2011.
- ^ Jasmer RM, Nahid P, Hopewell PC (2002). "Clinical practice. Latent tuberculosis infection". N. Engl. J. Med. 347 (23): 1860–6. doi:10.1056/NEJMcp021045. PMID 12466511. http://jasoncartermd.com/resources/pdf/Latent%20TB%20Infection.pdf., which cites Dolin PJ, Raviglione MC, Kochi A (1994). "Global tuberculosis incidence and mortality during 1990-2000". Bull World Health Organ 72 (2): 213–20. PMC 2486541. PMID 8205640. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2486541.
- ^ a b c World Health Organization (2009). "Epidemiology". Global tuberculosis control: epidemiology, strategy, financing. pp. 6–33. ISBN 9789241563802. http://who.int/entity/tb/publications/global_report/2009/pdf/chapter1.pdf. Retrieved 12 November 2009.
- ^ Schiffman G (15 January 2009). "Tuberculosis Symptoms". eMedicineHealth. http://www.emedicinehealth.com/tuberculosis/page3_em.htm.
- ^ "Tuberculosis (TB) Symptoms". NHS Choices Tuberculosis. National Health Service (NHS) UK. http://www.nhs.uk/Conditions/Tuberculosis/Pages/Symptoms.aspx. Retrieved 17 April 2011.
- ^ Golden MP, Vikram HR (2005). "Extrapulmonary tuberculosis: an overview". American family physician 72 (9): 1761–8. PMID 16300038.
- ^ Mile WA (September 1974). "Osseous tuberculosis". J Natl Med Assoc. 66 (5): 400–3. PMC 2609260. PMID 4414957. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2609260.
- ^ a b c d e f "Core Curriculum on Tuberculosis: What the Clinician Should Know". Centers for Disease Control and Prevention (CDC), Division of Tuberculosis Elimination. 2000, updated August 2003. http://www.cdc.gov/tb/?404;http://www.cdc.gov:80/tb/pubs/corecurr/default.htm.
- ^ Southwick F (10 December 2007). "Chapter 4: Pulmonary Infections". Infectious Diseases: A Clinical Short Course, 2nd ed.. McGraw-Hill Medical Publishing Division. p. 104. ISBN 0071477225. http://pharma-books.blogspot.com/2009/01/infectious-disease-clinical-short.html.
- ^ Cox R (2004). "Quantitative relationships for specific growth rates and macromolecular compositions of Mycobacterium tuberculosis, Streptomyces coelicolor A3(2) and Escherichia coli B/r: an integrative theoretical approach". Microbiology 150 (Pt 5): 1413–26. doi:10.1099/mic.0.26560-0. PMID 15133103. http://mic.sgmjournals.org/cgi/content/full/150/5/1413?view=long&pmid=15133103#R35.
- ^ a b Madison B (2001). "Application of stains in clinical microbiology". Biotech Histochem 76 (3): 119–25. doi:10.1080/714028138. PMID 11475314.
- ^ Parish T, Stoker N (1999). "Mycobacteria: bugs and bugbears (two steps forward and one step back)". Mol Biotechnol 13 (3): 191–200. doi:10.1385/MB:13:3:191. PMID 10934532.
- ^ van Soolingen D et al. (1997). "A novel pathogenic taxon of the Mycobacterium tuberculosis complex, Canetti: characterization of an exceptional isolate from Africa". Int. J. Syst. Bacteriol. 47 (4): 1236–45. doi:10.1099/00207713-47-4-1236. PMID 9336935.
- ^ Niemann S et al. (2002). "Mycobacterium africanum Subtype II Is Associated with Two Distinct Genotypes and Is a Major Cause of Human Tuberculosis in Kampala, Uganda". J. Clin. Microbiol. 40 (9): 3398–405. doi:10.1128/JCM.40.9.3398-3405.2002. PMC 130701. PMID 12202584. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=130701.
- ^ Niobe-Eyangoh SN et al. (2003). "Genetic Biodiversity of Mycobacterium tuberculosis Complex Strains from Patients with Pulmonary Tuberculosis in Cameroon". J. Clin. Microbiol. 41 (6): 2547–53. doi:10.1128/JCM.41.6.2547-2553.2003. PMC 156567. PMID 12791879. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=156567.
- ^ Thoen C, Lobue P, de Kantor I (2006). "The importance of Mycobacterium bovis as a zoonosis". Vet. Microbiol. 112 (2–4): 339–45. doi:10.1016/j.vetmic.2005.11.047. PMID 16387455.
- ^ Pfyffer GE, Auckenthaler R, van Embden JD, van Soolingen D (1998). "Mycobacterium canettii, the smooth variant of M. tuberculosis, isolated from a Swiss patient exposed in Africa". Emerging Infect. Dis. 4 (4): 631–4. doi:10.3201/eid0404.980414. PMC 2640258. PMID 9866740. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2640258.
- ^ Niemann S, Richter E, Dalügge-Tamm H, Schlesinger H, Graupner D, Königstein B, Gurath G, Greinert U, Rüsch-Gerdes S (2000). "Two cases of Mycobacterium microti derived tuberculosis in HIV-negative immunocompetent patients". Emerg Infect Dis 6 (5): 539–42. doi:10.3201/eid0605.000516. PMC 2627952. PMID 10998387. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2627952.
- ^ American Thoracic Society (1997). "Diagnosis and treatment of disease caused by nontuberculous mycobacteria. This official statement of the American Thoracic Society was approved by the Board of Directors, March 1997. Medical Section of the American Lung Association". Am J Respir Crit Care Med 156 (2 Pt 2): S1–25. PMID 9279284.
- ^ a b ATS/CDC Statement Committee on Latent Tuberculosis Infection (June 2000). "Targeted tuberculin testing and treatment of latent tuberculosis infection. American Thoracic Society". MMWR Recomm Rep 49 (RR–6): 1–51. PMID 10881762. http://www.cdc.gov/mmwr/preview/mmwrhtml/rr4906a1.htm#tab3.
- ^ Lee JH (1948). "Tuberculosis and Silicosis". Can Med Assoc J. 58 (4): 349–353. PMC 1591092. PMID 18916106. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1591092.
- ^ Varkey B (26 January 2011). "Silicosis". WebMD. http://emedicine.medscape.com/article/302027-overview#a0199. Retrieved 26 July 2011.
- ^ Segall L, Covic A (June 2010). "Diagnosis of tuberculosis in dialysis patients: current strategy". Clin J Am Soc Nephrol 5 (6): 1114–22. doi:10.2215/CJN.09231209. PMID 20413440. http://cjasn.asnjournals.org/cgi/pmidlookup?view=long&pmid=20413440.
- ^ Jeon CY, Murray MB (July 2008). Williams, Brian. ed. "Diabetes Mellitus Increases the Risk of Active Tuberculosis: A Systematic Review of 13 Observational Studies". PLoS Medicine 5 (7): e152. doi:10.1371/journal.pmed.0050152. PMC 2459204. PMID 18630984. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2459204.
- ^ "NIOSH – Silicosis: Learn the Facts!". cdc.gov. http://www.cdc.gov/niosh/docs/2004-108/. Retrieved 13 April 2010.
- ^ Leung CC, Lam TH, Chan WM et al. (June 2007). "Lower risk of tuberculosis in obesity". Arch. Intern. Med. 167 (12): 1297–304. doi:10.1001/archinte.167.12.1297. PMID 17592104. http://archinte.ama-assn.org/cgi/pmidlookup?view=long&pmid=17592104. "Figures OI70054T5, OI70054F1"
- ^ Restrepo, BI (2007). "Convergence of the tuberculosis and diabetes epidemics: renewal of old acquaintances". Clin Infect Dis 45 (4): 436–8. doi:10.1086/519939. PMC 2900315. PMID 17638190. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2900315.
- ^ Nijland HMJ et al. (2006). "Exposure to rifampicin is strongly reduced in patients with tuberculosis and type 2 diabetes". Clin Infect Dis 43 (7): 848–854. doi:10.1086/507543. PMID 16941365.
- ^ Stevenson CR, Forouhi NG, Roglic G et al. (2007). "Diabetes and tuberculosis: the impact of the diabetes epidemic on tuberculosis incidence". BMC Public Health 7: 234. doi:10.1186/1471-2458-7-234. PMC 2001194. PMID 17822539. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2001194.
- ^ Dooley KE, Chaisson RE (2009). "Tuberculosis and diabetes mellitus: convergence of two epidemics". Lancet Infect Dis 9 (12): 737–46. doi:10.1016/S1473-3099(09)70282-8. PMC 2945809. PMID 19926034. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2945809.
- ^ Nnoaham KE, Clarke A (2008). "Low serum vitamin D levels and tuberculosis: a systematic review and meta-analysis". Int J Epidemiol 37 (1): 113–19. doi:10.1093/ije/dym247. PMID 18245055.
- ^ Kallmann FJ, Reisner D (1942). "Twin studies on the significance of genetic factors in tuberculosis". Am Rev Tuberc 16: 593–617.
- ^ Jepson A et al. (2001). "Genetic Regulation of Acquired Immune Responses to Antigens of Mycobacterium tuberculosis: a Study of Twins in West Africa". Infect Immun 69 (6): 3989–94. doi:10.1128/IAI.69.6.3989-3994.2001. PMC 98461. PMID 11349068. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=98461.
- ^ Sepulveda RL, Heiba IM, Navarrete C, Elston RC, Gonzalez B, Sorensen RU (1994). "Tuberculin reactivity after newborn BCG immunization in mono‐ and dizygotic twins". Tuber Lung Dis 75 (2): 138–43. doi:10.1016/0962-8479(94)90043-4. PMID 8032047.
- ^ Cobat A et al. (2010). "High heritability of antimycobacterial immunity in an area of hyperendemicity for tuberculosis disease". J Infect Dis 201 (1): 15–19. doi:10.1086/648611. PMID 19938975.
- ^ Tso HW, Lau YL, Tam CM, Wong HS, Chiang KS (2004). "Associations between IL12B polymorphisms and tuberculosis in the Hong Kong Chinese population". J Infect Dis 190 (5): 913–9. doi:10.1086/422693. PMID 15295696.
- ^ Mutlu G, Mutlu E, Bellmeyer A, Rubinstein I (2006). "Pulmonary adverse events of anti-tumor necrosis factor-alpha antibody therapy". Am J Med 119 (8): 639–46. doi:10.1016/j.amjmed.2006.01.015. PMID 16887405.
- ^ Cole E, Cook C (1998). "Characterization of infectious aerosols in health care facilities: an aid to effective engineering controls and preventive strategies". Am J Infect Control 26 (4): 453–64. doi:10.1016/S0196-6553(98)70046-X. PMID 9721404.
- ^ Nicas M, Nazaroff WW, Hubbard A (2005). "Toward understanding the risk of secondary airborne infection: emission of respirable pathogens". J Occup Environ Hyg 2 (3): 143–54. doi:10.1080/15459620590918466. PMID 15764538.
- ^ Behr MA et al. (1999). "Transmission of Mycobacterium tuberculosis from patients smear-negative for acid-fast bacilli". Lancet 353 (9151): 444–9. doi:10.1016/S0140-6736(98)03406-0. PMID 9989714.
- ^ Griffith D, Kerr C (1996). "Tuberculosis: disease of the past, disease of the present". J Perianesth Nurs 11 (4): 240–5. doi:10.1016/S1089-9472(96)80023-2. PMID 8964016.
- ^ "Causes of Tuberculosis". Mayo Clinic. 21 December 2006. http://www.mayoclinic.com/health/tuberculosis/DS00372/DSECTION=3. Retrieved 19 October 2007.
- ^ Houben E, Nguyen L, Pieters J (2006). "Interaction of pathogenic mycobacteria with the host immune system". Curr Opin Microbiol 9 (1): 76–85. doi:10.1016/j.mib.2005.12.014. PMID 16406837.
- ^ Herrmann J, Lagrange P (2005). "Dendritic cells and Mycobacterium tuberculosis: which is the Trojan horse?". Pathol Biol (Paris) 53 (1): 35–40. doi:10.1016/j.patbio.2004.01.004. PMID 15620608.
- ^ Agarwal R, Malhotra P, Awasthi A, Kakkar N, Gupta D (2005). "Tuberculous dilated cardiomyopathy: an under-recognized entity?". BMC Infect Dis 5 (1): 29. doi:10.1186/1471-2334-5-29. PMC 1090580. PMID 15857515. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1090580.
- ^ a b c Grosset J (2003). "Mycobacterium tuberculosis in the Extracellular Compartment: an Underestimated Adversary". Antimicrob Agents Chemother 47 (3): 833–6. doi:10.1128/AAC.47.3.833-836.2003. PMC 149338. PMID 12604509. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=149338.
- ^ Kim J, Park Y, Kim Y, Kang S, Shin J, Park I, Choi B (2003). "Miliary tuberculosis and acute respiratory distress syndrome". Int J Tuberc Lung Dis 7 (4): 359–64. PMID 12733492.
- ^ Sambandamurth V, Wang X, Chen B, Russell R, Derrick S, Collins F, Morris S, Jacobs W (2002). "A pantothenate auxotroph of Mycobacterium tuberculosis is highly attenuated and protects mice against tuberculosis". Nat Med 8 (10): 1171–74. doi:10.1038/nm765. PMID 12219086.
- ^ Rothel J, Andersen P (2005). "Diagnosis of latent Mycobacterium tuberculosis infection: is the demise of the Mantoux test imminent?". Expert Rev Anti Infect Ther 3 (6): 981–93. doi:10.1586/14787210.3.6.981. PMID 16307510.
- ^ Nahid P, Pai M, Hopewell P (2006). "Advances in the Diagnosis and Treatment of Tuberculosis". Proc Am Thorac Soc 3 (1): 103–10. doi:10.1513/pats.200511-119JH. PMC 2658675. PMID 16493157. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2658675.
- ^ Pai M, Zwerling A, Menzies D (2008). "Systematic Review: T-Cell–based Assays for the Diagnosis of Latent Tuberculosis Infection: An Update". Ann. Intern. Med. 149 (3): 1–9. PMC 2951987. PMID 18593687. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2951987.
- ^ Lalvani A, Richeldi L, Kunst H (2005). "Interferon gamma assays for tuberculosis". Lancet Infect Dis 5 (6): 322–4; author reply 325–7. doi:10.1016/S1473-3099(05)70118-3. PMID 15919613.
- ^ Reddy JR, Kwang J, Lechtenberg KF, Khan NC, Prasad RB, Chengappa MM (2002). "An immunochromatographic serological assay for the diagnosis of Mycobacterium tuberculosis". Comp. Immunol. Microbiol. Infect. Dis. 25 (1): 21–7. doi:10.1016/S0147-9571(01)00016-9. PMID 11831744.
- ^ "WHO says Cepheid rapid test will transform TB care". Reuters. 8 December 2010. http://www.reuters.com/article/idUSTRE6B71RF20101208.
- ^ Guerra RL et al. (2007). "Use of the amplified mycobacterium tuberculosis direct test in a public health laboratory: test performance and impact on clinical care". Chest 132 (3): 946–51. doi:10.1378/chest.06-2959. PMID 17573496.
- ^ Bloch, Alan B.; Advisory Council for the Elimination of Tuberculosis (September 1995). "Screening for tuberculosis and tuberculosis infection in high-risk populations. Recommendations of the Advisory Council for the Elimination of Tuberculosis". MMWR Recomm Rep 44 (RR–11): 19–34. PMID 7565540. http://www.cdc.gov/mmwr/preview/mmwrhtml/00038873.htm.
- ^ Fine P, Floyd S, Stanford J, Nkhosa P, Kasunga A, Chaguluka S, Warndorff D, Jenkins P, Yates M, Ponnighaus J (2001). "Environmental mycobacteria in northern Malawi: implications for the epidemiology of tuberculosis and leprosy". Epidemiol Infect 126 (3): 379–87. doi:10.1017/S0950268801005532. PMC 2869706. PMID 11467795. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2869706.
- ^ "The Global Plan to Stop TB". World Health Organization. 2011. http://www.stoptb.org/global/plan/. Retrieved 13 June 2011.
- ^ Martin C (2006). "Tuberculosis vaccines: past, present and future". Curr Opin Pulm Med 12 (3): 186–91. doi:10.1097/01.mcp.0000219267.27439.1b. PMID 16582673.
- ^ World Health, Organization (23 January 2004). "BCG Vaccine". Weekly Epidemiological Record 79 (4): 27–38. PMID 14768305. http://www.who.int/immunization/wer7904BCG_Jan04_position_paper.pdf. Retrieved 26 July 2011.
- ^ "Vaccine and Immunizations: TB Vaccine (BCG)". Centers for Disease Control and Prevention. 2011. http://www.cdc.gov/tb/topic/vaccines/. Retrieved 26 July 2011.
- ^ a b Bonah C (2005). "The 'experimental stable' of the BCG vaccine: safety, efficacy, proof, and standards, 1921–1933". Stud Hist Philos Biol Biomed Sci 36 (4): 696–721. doi:10.1016/j.shpsc.2005.09.003. PMID 16337557.
- ^ a b Comstock G (1994). "The International Tuberculosis Campaign: a pioneering venture in mass vaccination and research". Clin Infect Dis 19 (3): 528–40. doi:10.1093/clinids/19.3.528. PMID 7811874.
- ^ Bannon M, Finn A (1999). "BCG and tuberculosis". Arch Dis Child 80 (1): 80–3. doi:10.1136/adc.80.1.80. PMC 1717792. PMID 10325767. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1717792.
- ^ World Health Organization (August 2006). "WHO/UNICEF Review of National Immunization Coverage 1980–2005: South Africa" (PDF). Archived from the original on 30 June 2007. http://web.archive.org/web/20070630073246/http://www.who.int/immunization_monitoring/data/south_africa.pdf. Retrieved 8 June 2007.
- ^ Skeiky YA, Sadoff JC (2006). "Advances in tuberculosis vaccine strategies". Nature reviews. Microbiology 4 (6): 469–76. doi:10.1038/nrmicro1419. PMID 16710326.
- ^ "First U.S. Tuberculosis Vaccine Trial in 60 Years Begins" (Press release). National Institute of Allergy and Infectious Diseases. 26 January 2004. http://www.nih.gov/news/pr/jan2004/niaid-26.htm. Retrieved 27 September 2009.
- ^ Skeiky YA, Alderson MR, Ovendale PJ, Guderian JA, Brandt L, Dillon DC, Campos-Neto A, Lobet Y, Dalemans W (2004). "Differential immune responses and protective efficacy induced by components of a tuberculosis polyprotein vaccine, Mtb72F, delivered as naked DNA or recombinant protein". J Immunol 172 (12): 7618–28. PMID 15187142. http://www.jimmunol.org/cgi/pmidlookup?view=long&pmid=15187142.
- ^ Ha S, Jeon B, Youn J, Kim S, Cho S, Sung Y (2005). "Protective effect of DNA vaccine during chemotherapy on reactivation and reinfection of Mycobacterium tuberculosis". Gene Ther 12 (7): 634–8. doi:10.1038/sj.gt.3302465. PMID 15690060.
- ^ Ibanga H, Brookes R, Hill P, Owiafe P, Fletcher H, Lienhardt C, Hill A, Adegbola R, McShane H (2006). "Early clinical trials with a new tuberculosis vaccine, MVA85A, in tuberculosis-endemic countries: issues in study design". Lancet Infect Dis 6 (8): 522–8. doi:10.1016/S1473-3099(06)70552-7. PMID 16870530.
- ^ Doherty, TM; Andersen, P (2005). "Vaccines for Tuberculosis: Novel Concepts and Recent Progress". Clinical Microbiology Reviews 18 (4): 687–702. doi:10.1128/CMR.18.4.687-702.2005. PMC 1265910. PMID 16223953. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1265910.
- ^ "Vaccine Research – Tuberculosis". Statens Serum Institut. Archived from the original on 17 July 2007. http://web.archive.org/web/20070717164308/http://www.ssi.dk/sw13853.asp. Retrieved 1 March 2009.
- ^ "Statens Serum Institut (SSI), Intercell (ICLL), and Aeras Global Tuberculosis Vaccine Foundation (Aeras) announce the initiation of a clinical trial for a novel vaccine candidate". Aeras. 4 December 2007. http://www.aeras.org/newscenter/news-detail.php?id=705. Retrieved 1 March 2009.
- ^ "Vaccine Discovery — Overview". Aeras. http://www.aeras.org/our-approach/vaccine-development.php?discovery-overview. Retrieved 1 March 2009.
- ^ "Tuberculosis Vaccine". Crucell. http://www.crucell.com/R_and_D-Clinical_Development-Tuberculosis_Vaccine. Retrieved 1 March 2009.
- ^ Dietrich J, Andersen C, Rappuoli R, Doherty TM, Jensen CG, Andersen P (2006). "Mucosal Administration of Ag85B-ESAT-6 Protects against Infection with Mycobacterium tuberculosis and Boosts Prior Bacillus Calmette-Guérin Immunity" (PDF). Journal of Immunology 177 (9): 6353–60. http://www.jimmunol.org/cgi/reprint/177/9/6353.pdf. Retrieved 1 March 2009.
- ^ Webber D, Kremer M (2001). "Stimulating Industrial R&D for Neglected Infectious Diseases: Economic Perspectives". Bulletin of the World Health Organization 79 (8): 693–801. http://www.who.int/bulletin/archives/79(8)735.pdf.
- ^ Barder O, Kremer M, Williams H (2006). "Advance Market Commitments: A Policy to Stimulate Investment in Vaccines for Neglected Diseases". The Economists' Voice 3 (3). doi:10.2202/1553-3832.1144. http://www.bepress.com/ev/vol3/iss3/art1.
- ^ a b Aagaard C, Hoang T, Dietrich J, Cardona PJ, Izzo A, Dolganov G, Schoolnik GK, Cassidy JP, Billeskov R, Andersen P (2011). "A Multistage Tuberculosis Vaccine that Confers Efficient Protection Before and After Exposure". Nature Medicine 17 (2): 189–94. doi:10.1038/nm.2285. PMID 21258338.
- ^ Kaufmann SH (2010). "Future Vaccinations Strategies against Tuberculosis Thinking outside the box". Immunity 33 (4): 567–77. doi:10.1016/j.immuni.2010.09.015. PMID 21029966.
- ^ Acharya NPV, Senn M, Lederer E (1967). "Sur la presence et structure de mycolate d'arabinose dans les lipides lies de deux souches de Mycobacteries". Compte Rendu Acad Sci Hebd Acad Sci D. 264: 2173–6.
- ^ Migliore D, Acharya NPV, Jolles P (1966). "Characterization of large quantities of glutamic acid in the walls of human virulent strains of mycobacteria". Compte Rendu Acad Sci Hebd Acad Sci D. 263 (11): 846–8. PMID 4958543.
- ^ Acharya PV, Goldman DS (1970). "Chemical Composition of the Cell Wall of the H37Ra Strain of Mycobacterium tuberculosis". J Bacteriol 102 (3): 733–9. PMC 247620. PMID 4988039. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=247620.
- ^ Brennan PJ, Nikaido H (1995). "The envelope of mycobacteria". Annu. Rev. Biochem. 64: 29–63. doi:10.1146/annurev.bi.64.070195.000333. PMID 7574484.
- ^ a b O'Brien R (1994). "Drug-resistant tuberculosis: etiology, management and prevention". Semin Respir Infect 9 (2): 104–12. PMID 7973169.
- ^ a b Centers for Disease Control and Prevention (CDC) (2006). "Emergence of Mycobacterium tuberculosis with extensive resistance to second-line drugs—worldwide, 2000–2004". MMWR Morb Mortal Wkly Rep 55 (11): 301–5. PMID 16557213. http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5511a2.htm.
- ^ Farmer P (2001). "The major infectious diseases in the world—to treat or not to treat?". N. Engl. J. Med. 345 (3): 208–10. doi:10.1056/NEJM200107193450310. PMID 11463018.
- ^ Pope AS (1938). "The Role of the Sanatorium in Tuberculosis Control". The Milbank Memorial Fund Quarterly 16 (4): 327–37. doi:10.2307/3347949. JSTOR 3347949.
- ^ Parrish N, Dick J, Bishai W (1998). "Mechanisms of latency in Mycobacterium tuberculosis". Trends Microbiol 6 (3): 107–12. doi:10.1016/S0966-842X(98)01216-5. PMID 9582936.
- ^ Lambert M et al. (2003). "Recurrence in tuberculosis: relapse or reinfection?". Lancet Infect Dis 3 (5): 282–7. doi:10.1016/S1473-3099(03)00607-8. PMID 12726976.
- ^ Verver S et al. (2005). "Rate of reinfection tuberculosis after successful treatment is higher than rate of new tuberculosis". Am J Respir Crit Care Med 171 (12): 1430–5. doi:10.1164/rccm.200409-1200OC. PMID 15831840. See also:
- Das S et al. (1993). "Application of DNA fingerprinting with IS986 to sequential mycobacterial isolates obtained from pulmonary tuberculosis in Hong Kong before, during and after short-course chemotherapy". Tuber Lung Dis 74 (1): 47–51. doi:10.1016/0962-8479(93)90068-9. PMID 8098637.
- Das S et al. (1995). "IS6110 restriction fragment length polymorphism typing of clinical isolates of Mycobacterium tuberculosis from people with pulmonary tuberculosis in Madras, south India". Tuber Lung Dis 76 (6): 550–4. doi:10.1016/0962-8479(95)90533-2. PMID 8593378.
- García De Viedma D (2002). "Tuberculosis recurrences: reinfection plays a role in a population whose clinical/epidemiological characteristics do not favor reinfection". Arch Intern Med 162 (16): 1873–9. doi:10.1001/archinte.162.16.1873. PMID 12196086.
- Van Rie A et al. (1999). "Exogenous reinfection as a cause of recurrent tuberculosis after curative treatment". N Engl J Med 341 (16): 1174–9. doi:10.1056/NEJM199910143411602. PMID 10519895.
- Shen G, Xue Z, Shen X et al. (November 2006). "The study recurrent tuberculosis and exogenous reinfection, Shanghai, China". Emerging Infect. Dis. 12 (11): 1776–8. PMID 17283636. http://www.cdc.gov/ncidod/EID/vol12no11/05-1207.htm.
- ^ "WHO Disease and injury country estimates". World Health Organization. 2004. http://www.who.int/healthinfo/global_burden_disease/estimates_country/en/index.html. Retrieved 11 November 2009.
- ^ a b World Health Organization (2009). "The Stop TB Strategy, case reports, treatment outcomes and estimates of TB burden". Global tuberculosis control: epidemiology, strategy, financing. pp. 187–300. ISBN 9789241563802. http://who.int/tb/publications/global_report/2009/annex_3/en/index.html. Retrieved 14 November 2009.
- ^ World Health Organization. "WHO report 2008: Global tuberculosis control". http://www.who.int/tb/publications/global_report/2008/annex_3/en/index.html. Retrieved 13 April 2009.
- ^ "Fact Sheets: The Difference Between Latent TB Infection and Active TB Disease". Centers for Disease Control. 20 June 2011. http://www.cdc.gov/tb/publications/factsheets/general/LTBIandActiveTB.htm. Retrieved 26 July 2011.
- ^ Stop TB Partnership (4 December 2002). "London tuberculosis rates now at Third World proportions". PR Newswire Europe. http://www.prnewswire.co.uk/cgi/news/release?id=95088. Retrieved 3 October 2006.
- ^ Iademarco MF, Castro KG (2003). "Epidemiology of tuberculosis". Semin Respir Infect 18 (4): 225–40. doi:10.1053/S0882-0546(03)00074-4. PMID 14679472.
- ^ Sobero R, Peabody J (2006). "Tuberculosis control in Bolivia, Chile, Colombia and Peru: why does incidence vary so much between neighbors?". Int J Tuberc Lung Dis 10 (11): 1292–5. PMID 17131791.
- ^ Al-Azem A, Kaushal Sharma M, Turenne C, Hoban D, Hershfield E, MacMorran J, Kabani A (1998). "Rural outbreaks of Mycobacterium tuberculosis in a Canadian province". Abstr Intersci Conf Antimicrob Agents Chemother Intersci Conf Antimicrob Agents Chemother 38: 555. abstract no. L-27. http://gateway.nlm.nih.gov/MeetingAbstracts/ma?f=102188560.html.
- ^ World Health Organization. "Global Tuberculosis Control Report, 2006 – Annex 1 Profiles of high-burden countries" (PDF). http://www.who.int/tb/publications/global_report/2006/pdf/full_report_correctedversion.pdf. Retrieved 13 October 2006.
- ^ Centers for Disease Control and Prevention (12 September 2006). "2005 Surveillance Slide Set". http://www.cdc.gov/nchstp/tb/pubs/slidesets/surv/surv2005/default.htm. Retrieved 13 October 2006.
- ^ World Health Organization. "Global tuberculosis control – surveillance, planning, financing WHO Report 2006". http://www.who.int/tb/publications/global_report/en/index.html. Retrieved 13 October 2006.
- ^ Chaisson RE, Martinson NA (2008). "Tuberculosis in Africa—combating an HIV-driven crisis". N Engl J Med 358 (11): 1089–92. doi:10.1056/NEJMp0800809. PMID 18337598.
- ^ Davies PDO, Yew WW, Ganguly D et al. (2006). "Smoking and tuberculosis: the epidemiological association and pathogenesis". Trans R Soc Trop Med Hyg 100 (4): 291–8. doi:10.1016/j.trstmh.2005.06.034. PMID 16325875.
- ^ Jha P, Jacob B, Gajalakshmi V et al. (2008). "A nationally representative case–control study of smoking and death in India". N Engl J Med 358 (11): 1137–47. doi:10.1056/NEJMsa0707719. PMID 18272886.
- ^ Restrepo BI (2007). "Convergence of the tuberculosis and diabetes epidemics: renewal of old acquaintances". Clin. Infect. Dis. 45 (4): 436–8. doi:10.1086/519939. PMC 2900315. PMID 17638190. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2900315.
- ^ a b Strachan DP, Powell KJ, Thaker A, Millard FJ, Maxwell JD (1995-02). "Vegetarian diet as a risk factor for tuberculosis in immigrant south London Asians". Thorax 50 (2): 175–80. doi:10.1136/thx.50.2.175. PMC 473919. PMID 7701458. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=473919.
- ^ Davis L (1995). "Vegetarian diet and tuberculosis in immigrant Asians". Thorax 50 (8): 915–6. doi:10.1136/thx.50.8.915-c. PMC 474924. PMID 7570453. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=474924.
- ^ Ustianowski A, Shaffer R, Collin S, Wilkinson RJ, Davidson RN (2005). "Prevalence and associations of vitamin D deficiency in foreign-born persons with tuberculosis in London". J Infect 50 (5): 432–7. doi:10.1016/j.jinf.2004.07.006. PMID 15907552. http://linkinghub.elsevier.com/retrieve/pii/S0163-4453(04)00161-6.
- ^ Nnoaham KE, Clarke A (2008). "Low serum vitamin D levels and tuberculosis: a systematic review and meta-analysis". Int J Epidemiol 37 (1): 113–9. doi:10.1093/ije/dym247. PMID 18245055. http://ije.oxfordjournals.org/cgi/pmidlookup?view=long&pmid=18245055.
- ^ Schaible UE, Kaufmann SH (2007). "Malnutrition and Infection: Complex Mechanisms and Global Impacts". PLoS medicine 4 (5): e115. doi:10.1371/journal.pmed.0040115. PMC 1858706. PMID 17472433. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1858706.
- ^ Lönnroth K, Raviglione M (2008). "Global epidemiology of tuberculosis: prospects for control". Seminars in respiratory and critical care medicine 29 (5): 481–91. doi:10.1055/s-0028-1085700. PMID 18810682.
- ^ Davies PD (2003). "The world-wide increase in tuberculosis: how demographic changes, HIV infection and increasing numbers in poverty are increasing tuberculosis". Annals of medicine 35 (4): 235–43. doi:10.1080/07853890310005713. PMID 12846265.
- ^ Spence DP, Hotchkiss J, Williams CS, Davies PD (1993). "Tuberculosis and poverty". BMJ (Clinical research ed.) 307 (6907): 759–61. doi:10.1136/bmj.307.6907.759. PMC 1696420. PMID 8219945. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1696420.
- ^ "Increased US Prison Population Has Profound Demographic Consequences, Disproportionately Affecting Black Males". Medicalnewstoday.com. http://www.medicalnewstoday.com/releases/117049.php. Retrieved 2011-10-24.
- ^ "Tuberculosis: stopping a killer that can't be kept behind bars". ICRC. http://www.icrc.org/web/eng/siteeng0.nsf/html/azerbaijan-interview-220310. Retrieved 26 July 2011.
- ^ Larouzé B, Sánchez A, Diuana V (2008). "Tuberculosis behind bars in developing countries: a hidden shame to public health". Trans. R. Soc. Trop. Med. Hyg. 102 (9): 841–2. doi:10.1016/j.trstmh.2008.04.020. PMID 18513772.
- ^ a b Global Tuberculosis Control, World Health Organization, 2011
- ^ Tuberculosis on the decline for first time ever – UN report, October 11, 2011, UN News Centre
- ^ Rothschild BM, Martin LD, Lev G et al. (August 2001). "Mycobacterium tuberculosis complex DNA from an extinct bison dated 17,000 years before the present". Clin. Infect. Dis. 33 (3): 305–11. doi:10.1086/321886. PMID 11438894. http://www.journals.uchicago.edu/cgi-bin/resolve?CID001531.
- ^ Pearce-Duvet J (2006). "The origin of human pathogens: evaluating the role of agriculture and domestic animals in the evolution of human disease". Biol Rev Camb Philos Soc 81 (3): 369–82. doi:10.1017/S1464793106007020. PMID 16672105.
- ^ Zink A, Sola C, Reischl U, Grabner W, Rastogi N, Wolf H, Nerlich A (2003). "Characterization of Mycobacterium tuberculosis Complex DNAs from Egyptian Mummies by Spoligotyping". J Clin Microbiol 41 (1): 359–67. doi:10.1128/JCM.41.1.359-367.2003. PMC 149558. PMID 12517873. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=149558.
- ^ Hippocrates. Aphorisms. Accessed 07 October 2006.
- ^ Konomi N, Lebwohl E, Mowbray K, Tattersall I, Zhang D (2002). "Detection of Mycobacterial DNA in Andean Mummies". J Clin Microbiol 40 (12): 4738–40. doi:10.1128/JCM.40.12.4738-4740.2002. PMC 154635. PMID 12454182. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=154635.
- ^ Sledzik P, Bellantoni N (1994). "Brief communication: bioarcheological and biocultural evidence for the New England vampire folk belief". Am J Phys Anthropol 94 (2): 269–74. doi:10.1002/ajpa.1330940210. PMID 8085617.
- ^ Léon Charles Albert Calmette at Who Named It?
- ^ Trail RR (April 1970). "Richard Morton (1637–1698)". Med Hist 14 (2): 166–74. PMC 1034037. PMID 4914685. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1034037.
- ^ Zur Pathogenie der Impetigines. Auszug aus einer brieflichen Mitteilung an den Herausgeber. [Müller’s] Archiv für Anatomie, Physiologie und wissenschaftliche Medicin. 1839, page 82.
- ^ Kentucky: Mammoth Cave long on history. CNN. 27 February 2004. Accessed 08 October 2006.
- ^ a b c McCarthy OR (August 2001). "The key to the sanatoria". J R Soc Med 94 (8): 413–7. PMC 1281640. PMID 11461990. http://www.jrsm.org/cgi/pmidlookup?view=long&pmid=11461990.
- ^ Nobel Foundation. The Nobel Prize in Physiology or Medicine 1905. Accessed 07 October 2006.
- ^ Waddington K (January 2004). "To stamp out "so terrible a malady": bovine tuberculosis and tuberculin testing in Britain, 1890-1939". Med Hist 48 (1): 29–48. PMC 546294. PMID 14968644. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=546294.
- ^ Medical Research Council. Origins of the MRC. Accessed 07 October 2006.
- ^ Wolfart W (1990). "[Surgical treatment of tuberculosis and its modifications—collapse therapy and resection treatment and their present-day sequelae]" (in German). Offentl Gesundheitswes 52 (8–9): 506–11. PMID 2146567.
- ^ Lalloo UG, Naidoo R, Ambaram A (May 2006). "Recent advances in the medical and surgical treatment of multi-drug resistant tuberculosis". Curr Opin Pulm Med 12 (3): 179–85. doi:10.1097/01.mcp.0000219266.27439.52. PMID 16582672. http://meta.wkhealth.com/pt/pt-core/template-journal/lwwgateway/media/landingpage.htm?issn=1070-5287&volume=12&issue=3&spage=179.
- ^ Paolo WF, Nosanchuk JD (May 2004). "Tuberculosis in New York city: recent lessons and a look ahead". Lancet Infect Dis 4 (5): 287–93. doi:10.1016/S1473-3099(04)01004-7. PMID 15120345. http://linkinghub.elsevier.com/retrieve/pii/S1473309904010047.
- ^ World Health Organization (WHO). Frequently asked questions about TB and HIV. Retrieved 6 October 2006.
- ^ 14 CA ADC § 671 Barclays official California code of regulations; Title 14. Natural resources; Division 1. Fish and game commission – Department of fish and game; Subdivision 3. General regulations; Chapter 3. Miscellaneous.
- ^ Tweddle N, Livingstone P (1994). "Bovine tuberculosis control and eradication programs in Australia and New Zealand". Vet Microbiol 40 (1–2): 23–39. doi:10.1016/0378-1135(94)90044-2. PMID 8073626.
- ^ The Department of Agriculture & Food (Ireland). Disease Eradication Schemes - Bovine Tuberculosis and Brucellosis. Retrieved on 8 May 2006.
- ^ Cassidy, Martin. Badgers targeted over bovine TB. BBC News 2 December, 2004. Retrieved on 8 May 2006.
- ^ National Federation of Badger Groups (Ireland). Cattle blamed for massive increase in bovine TB. Retrieved on 8 May 2006.
Further reading
- Blumberg HM, Leonard MK, Jasmer RM (2005). "Update on the treatment of tuberculosis and latent tuberculosis infection". JAMA 293 (22): 2776–84. doi:10.1001/jama.293.22.2776. PMID 15941808.
- Lawlor C (2007). Consumption and Literature. Basingstoke: Palgrave Macmillan. ISBN 0230020038.
- Nemery B, Yew WW, Albert R et al. (2005). "Tuberculosis, nontuberculous lung infection, pleural disorders, pulmonary function, respiratory muscles, occupational lung disease, pulmonary infections, and social issues in AJRCCM in 2004". Am. J. Respir. Crit. Care Med. 171 (6): 554–62. doi:10.1164/rccm.2412009. PMID 15753485.
External links
- Tuberculosis at the Open Directory Project
- "Stop TB Partnership". http://www.stoptb.org/. Retrieved 2011-04-06.
- "Tuberculosis (TB)". Centers for Disease Control. http://www.cdc.gov/tb/default.htm. Retrieved 2011-04-06.
- "Tuberculosis (TB)". UK Health Protection Agency. http://www.hpa.org.uk/infections/topics_az/tb/menu.htm. Retrieved 2011-04-06.
- "The Tuberculosis Coalition for Technical Assistance (TBCTA)". http://www.tbcta.org. Retrieved 2011-04-06.
Actinobacteria (high-G+C) Infectious diseases · Bacterial diseases: G+ (primarily A00–A79, 001–041, 080–109) Actinomycineae Actinomyces israelii (Actinomycosis, Cutaneous actinomycosis) · Tropheryma whipplei (Whipple's disease) · Arcanobacterium haemolyticum (Arcanobacterium haemolyticum infection)Corynebacterineae Tuberculosis: Ghon focus/Ghon's complex · Pott disease · brain (Meningitis, Rich focus) · Tuberculous lymphadenitis (Tuberculous cervical lymphadenitis) · cutaneous (Scrofuloderma, Erythema induratum, Lupus vulgaris, Prosector's wart, Tuberculosis cutis orificialis, Tuberculous cellulitis, Tuberculous gumma) · Lichen scrofulosorum · Tuberculid (Papulonecrotic tuberculid) · Primary inoculation tuberculosis · Miliary · Tuberculous pericarditis · Urogenital tuberculosis · Multi-drug-resistant tuberculosis · Extensively drug-resistant tuberculosisLeprosy: Tuberculoid leprosy · Borderline tuberculoid leprosy · Borderline leprosy · Borderline lepromatous leprosy · Lepromatous leprosy · Histoid leprosyR3: M. avium complex/Mycobacterium avium/Mycobacterium intracellulare/MAP (MAI infection) · M. ulcerans (Buruli ulcer) · M. haemophilumCorynebacterium diphtheriae (Diphtheria) · Corynebacterium minutissimum (Erythrasma) · Corynebacterium jeikeium (Group JK corynebacterium sepsis)Bifidobacteriaceae Gardnerella vaginalisDiseases of poverty Diseases of poverty Neglected diseases Miscellaneous Categories:- Tuberculosis
- Health in Africa
- Health in India
- Mycobacterium-related cutaneous conditions
Wikimedia Foundation. 2010.