- Crosslinking of DNA
-
Crosslinks in DNA occur when various exogenous or endogenous agents react with two different positions in the DNA. This can either occur in the same strand (intrastrand crosslink) or in the opposite strands of the DNA (interstrand crosslink). Crosslinks also occur between DNA and protein. DNA replication is blocked by crosslinks, which causes replication arrest and cell death if the crosslink is not repaired.
The RAD51 family plays a role in repair.[1]
Contents
Agents that Crosslink DNA
I. Exogenous Cross Linking Agents
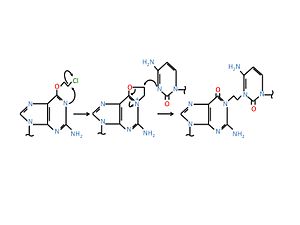
Alkylating agents such as 1, 3-bis(2-chloroethyl)-1-nitrosourea (BCNU, Carmustine)) and nitrogen mustard which are used in chemotherapy can cross link with DNA at N7 position of guanine on the opposite strands forming interstrand crosslink.[2]
Cisplatin (cis-diamminedichloroplatinum(II)) and its derivative forms DNA cross links as monoadduct, interstrand crosslink, intrastrand crosslink or DNA protein crosslink. Mostly it acts on the adjacent N-7 guanine forming 1, 2 intrastrand crosslink.[3][4]
II. Endogenous Cross Linking Agents
- Nitrous acid is formed in the stomach from dietary sources of nitrites. It induces formation of interstrand DNA crosslinks at the aminogroup of exocyclic N2 of guanine at CG sequences.
- Reactive chemicals such as malondialdehyde which are formed endogenously as the product of lipid peroxidation. They create etheno adducts formed by aldehyde which undergo rearrangements to form crosslinks on opposite strands.[5]
- Psoralens are natural compounds (furocoumarins) present in plants. These compounds get activated in the presence of UV - A. They form covalent adducts with pyrimidines. Covalent adducts are formed by linking 3, 4 (pyrone) or 4', 5’ (furan) edge of psoralen to 5, 6 double bond of thymine. Psoralens can form two types of monoadducts and one diadduct (an interstrand crosslink) reacting with thymine.[6] The crosslinking reaction by Psoralens targets TA sequences intercalating in DNA and linking one base of the DNA with the one below it. Psoralen adducts cause replication arrest and is used in the treatment of psoriasis and vitiligo.
- Aldehydes such as acrolein and crotonaldehyde found in tobacco smoke or automotive exhaust can form DNA interstrand crosslinks in DNA. Guanine adducts of DNA can also react with protein. A Schiff base formation between protein and aldehyde causes this DNA protein interstrand link
- Formaldehyde (HCHO) induces protein-DNA and protein-protein crosslinks, and is a common reagent of choice for molecular biology experiments.[7] These crosslinks may be reversed by incubation at 70°C.[8]
References
- ^ Gruver AM, Miller KA, Rajesh C, et al. (November 2005). "The ATPase motif in RAD51D is required for resistance to DNA interstrand crosslinking agents and interaction with RAD51C". Mutagenesis 20 (6): 433–40. doi:10.1093/mutage/gei059. PMID 16236763. http://mutage.oxfordjournals.org/cgi/pmidlookup?view=long&pmid=16236763.
- ^ Ali-Osman F, Rairkar A, Young P (January 1995). "Formation and repair of 1,3-bis-(2-chloroethyl)-1-nitrosourea and cisplatin induced total genomic DNA interstrand crosslinks in human glioma cells". Cancer Biochem. Biophys. 14 (4): 231–41. PMID 7767897.
- ^ Poklar N, Pilch DS, Lippard SJ, Redding EA, Dunham SU, Breslauer KJ (July 1996). "Influence of cisplatin intrastrand crosslinking on the conformation, thermal stability, and energetics of a 20-mer DNA duplex". Proc. Natl. Acad. Sci. U.S.A. 93 (15): 7606–11. doi:10.1073/pnas.93.15.7606. PMC 38793. PMID 8755522. http://www.pnas.org/cgi/pmidlookup?view=long&pmid=8755522.
- ^ Rudd GN, Hartley JA, Souhami RL (1995). "Persistence of cisplatin-induced DNA interstrand crosslinking in peripheral blood mononuclear cells from elderly and young individuals". Cancer Chemother. Pharmacol. 35 (4): 323–6. doi:10.1007/BF00689452. PMID 7828275.
- ^ Mathews & Vanholde, Biochemistry, 2nd Edition. Benjamin Cummings Publication
- ^ Qi Wu, Laura A Christensen, Randy J Legerski & Karen M Vasquez, Mismatch repair participates in error-free processing of DNA interstrand crosslinks in human cells,EMBO reports 6, 6, 551–557 (2005).
- ^ Formaldehyde Crosslinking Experiments [1]
- ^ Somashe Niranjanakumaria, Erika Lasdaa, Robert Brazasa, Mariano A. Garcia-Blanco. Reversible cross-linking combined with immunoprecipitation to study RNA–protein interactions in vivo. Methods. 2002 Feb;26(2):182-90. [2]
External links
Categories:
Wikimedia Foundation. 2010.