- Gemcitabine
-
Gemcitabine 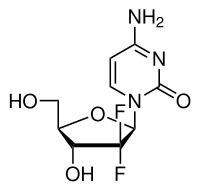
Systematic (IUPAC) name 4-amino-1-(2-deoxy-2,2-difluoro-β-D-erythro-pentofuranosyl)pyrimidin-2(1H)-on Clinical data Trade names Gemzar AHFS/Drugs.com monograph Pregnancy cat. D(US) Legal status ? Routes Oral, intravenous Pharmacokinetic data Protein binding <10% Half-life Short infusions 32-94 minutes
for long infusions 245-638 minutesIdentifiers CAS number 95058-81-4 ATC code L01BC05 PubChem CID 60750 DrugBank APRD00201 ChemSpider 54753 
UNII B76N6SBZ8R 
KEGG D02368 
ChEBI CHEBI:175901 
ChEMBL CHEMBL888 
Chemical data Formula C9H11F2N3O4 Mol. mass 263.198 g/mol SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Gemcitabine (pronunciation: jem-SITE-a-been) is a nucleoside analog used as chemotherapy. It is marketed as Gemzar by Eli Lilly and Company.
Contents
Pharmacology
Chemically gemcitabine is a nucleoside analog in which the hydrogen atoms on the 2' carbon of deoxycytidine are replaced by fluorine atoms.
As with fluorouracil and other analogues of pyrimidines, the triphosphate analogue of gemcitabine replaces one of the building blocks of nucleic acids, in this case cytidine, during DNA replication. The process arrests tumor growth, as only one additional nucleoside can be attached to the "faulty" nucleoside, resulting in apoptosis.
Another target of gemcitabine is the enzyme ribonucleotide reductase (RNR). The diphosphate analogue binds to RNR active site and inactivates the enzyme irreversibly. Once RNR is inhibited, the cell cannot produce the deoxyribonucleotides required for DNA replication and repair, and cell apoptosis is induced.[1]
Administration
Gemcitabine is administered by the intravenous route, since it is extensively metabolized by the gastrointestinal tract. Dose ranges from 1-1.2 g/m2 of body surface area according to type of cancer treated.[2]
Indications
Gemcitabine is used in various carcinomas: non-small cell lung cancer, pancreatic cancer, bladder cancer and breast cancer. It is being investigated for use in esophageal cancer, and is used experimentally in lymphomas and various other tumor types. Gemcitabine represents an advance in pancreatic cancer care[citation needed]. It is also not as debilitating as some other forms of chemotherapy.
A study reported in the Journal of the American Medical Association in 2007 suggested that gemcitabine showed benefit in patients with pancreatic cancer who were felt to have successful tumor resections.[3]
Gemcitabine became first line treatment for bladder cancer Stage 4 with metastases in combination with cisplatin after a study in 2000 with 405 patients showed similar efficacy but less toxicity compared to the former MVAC regimen.[4] This new CG-regimen involves taking cisplatin on day 2 and taking gemcitabine on days 1, 8, and 15. In July 2006 the FDA approved gemcitabine for use with carboplatin in the treatment of advanced ovarian cancer that has relapsed at least 6 months after completion of platinum-based (e.g., carboplatin or cisplatin) therapy. Neutropenia was the most commonly reported adverse effect (90% of patients). Other serious adverse effects were mostly hematologic. Gemcitabine was recently also investigated for advanced cancer of the biliary tract and gallbladder and was found to have a modest effect on the tumor when combined with cisplatin (NEJM 2010).
GemCarbo chemotherapy for lung cancer
GemCarbo chemotherapy, consisting of a combination of gemcitabine and carboplatin, is used to treat several different types of cancer, but is most commonly used to treat lung cancer.[5] GemCarbo chemotherapy is usually given as a day patient treatment, involving a blood test the day before, and the drugs are given by an infusion. The GemCarbo regimen is given as a 21-day cycle and on the first day of treatment the patient is given both the gemcitabine and carboplatin. On the same day of the following week (day eight) there is a drip of gemcitabine only. There then follows a rest period of two weeks which completes one cycle of chemotherapy. The next cycle of treatment is given after a rest period, which will be three weeks after the first injection. Usually 4–6 cycles of treatment are given over a period of 3–4 months and this makes up a course of treatment.
Side effects
The following side effects are common (occurring in more than 30%) for patients taking Gemcitabine:
- Flu-like symptoms such as muscle pain, fever, headache, chills, and fatigue
- Fever (within 6–12 hours of first dose)
- Fatigue
- Nausea (mild)
- Vomiting
- Poor appetite
- Skin rash
Low blood counts. White and red blood cells and platelets may temporarily decrease, increasing risk of infection, anemia, and bleeding.
Nadir (low point), the time between chemotherapy cycles at which blood counts are at their lowest.
Onset: none noted Nadir: 10–14 days Recovery: day 21
Temporary increases in liver enzymes. Blood or protein in the urine.
These are less common side effects (occurring in 10-29%) for patients receiving Gemcitabine:
- Diarrhea
- Weakness
- Hair loss
- Mouth sores
- Difficulty sleeping
- Shortness of breath (see lung problems)
History
Gemcitabine was first synthesized in Larry Hertel's lab at Eli Lilly during the early 1980s.[6] It was intended as an antiviral drug, but preclinical testing showed that it killed leukemia cells in vitro.[6]
References
- ^ N. M. F. S. A. Cerqueira, P. A. Fernandes, M. J. Ramos (2007). "Understanding ribonucleotide reductase inactivation by gemcitabine". Chemistry, a European Journal 13 (30): 8507–15. doi:10.1002/chem.200700260. PMID 17636467.
- ^ Chu E., DeVita V. T., "Physicians' Cancer Chemotherapy Drug Manual, 2007", Jones & Bartlett, 2007.
- ^ Oettle H, Post S, Neuhaus P, et al. (January 2007). "Adjuvant chemotherapy with gemcitabine vs observation in patients undergoing curative-intent resection of pancreatic cancer: a randomized controlled trial". JAMA 297 (3): 267–77. doi:10.1001/jama.297.3.267. PMID 17227978. http://jama.ama-assn.org/cgi/pmidlookup?view=long&pmid=17227978.
- ^ Von Der Maase, H; Hansen, SW; Roberts, JT; Dogliotti, L; Oliver, T; Moore, MJ; Bodrogi, I; Albers, P et al. (2000). "Gemcitabine and cisplatin versus methotrexate, vinblastine, doxorubicin, and cisplatin in advanced or metastatic bladder cancer: results of a large, randomized, multinational, multicenter, phase III study.". J Clin Oncol 18 (17): 3068. PMID 11001674.
- ^ Macmillan GemCarbo chemotherapy
- ^ a b Sneader, Walter (2005). Drug discovery: a history. New York: Wiley. pp. 259. ISBN 0-471-89979-8.
External links
Eli Lilly and Company Corporate directors John C. Lechleiter · Ralph Alvarez · Sir Winfried Bischoff · Michael L. Eskew · Martin S. Feldstein · J. Erik Fyrwald · Alfred G. Gilman · Karen N. Horn · Ellen R. Marram · Douglas R. Oberhelman · Franklyn G. Prendergast · Kathi P. SeifertProducts Annual revenue: $20.4 billion USD ( -10.17% FY 2008) · Employees: 40,250 · Stock symbol: NYSE: LLY · Website: www.lilly.comCategories:
-10.17% FY 2008) · Employees: 40,250 · Stock symbol: NYSE: LLY · Website: www.lilly.comCategories:- Eli Lilly and Company
- Antineoplastic antimetabolites
- Organofluorides
- Pyrimidones
Wikimedia Foundation. 2010.
