- Lidocaine
-
Lidocaine 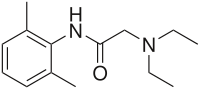
Systematic (IUPAC) name 2-(diethylamino)-
N-(2,6-dimethylphenyl)acetamideClinical data Trade names Xylocaine AHFS/Drugs.com Micromedex Detailed Consumer Information Pregnancy cat. A(AU) B(US) Legal status Prescription Only (S4) (AU) ? (US) Routes IV, subcutaneous, topical Pharmacokinetic data Bioavailability 35% (oral)
3% (topical)Metabolism Hepatic, 90% CYP1A2-mediated Half-life 1.5–2 hours Excretion renal Identifiers CAS number 137-58-6 
73-78-9 (hydrochloride)ATC code N01BB02 C01BB01 D04AB01 S02DA01 C05AD01 PubChem CID 3676 DrugBank DB00281 ChemSpider 3548 
UNII 98PI200987 
KEGG D00358 
ChEBI CHEBI:6456 
ChEMBL CHEMBL79 
Synonyms N-(2,6-dimethylphenyl)-N2,N2-diethylglycinamide Chemical data Formula C14H22N2O Mol. mass 234.34 g/mol SMILES eMolecules & PubChem Physical data Melt. point 68 °C (154 °F)  (what is this?) (verify)
(what is this?) (verify)Lidocaine (INN) (
 /ˈlaɪdɵkeɪn/), Xylocaine, or lignocaine (former BAN) (
/ˈlaɪdɵkeɪn/), Xylocaine, or lignocaine (former BAN) ( /ˈlɪɡnɵkeɪn/) is a common local anesthetic and antiarrhythmic drug. Lidocaine is used topically to relieve itching, burning and pain from skin inflammations, injected as a dental anesthetic or as a local anesthetic for minor surgery.
/ˈlɪɡnɵkeɪn/) is a common local anesthetic and antiarrhythmic drug. Lidocaine is used topically to relieve itching, burning and pain from skin inflammations, injected as a dental anesthetic or as a local anesthetic for minor surgery.Contents
History
Lidocaine, the first amino amide-type local anesthetic, was first synthesized under the name Xylocaine by Swedish chemist Nils Löfgren in 1943.[1] His colleague Bengt Lundqvist performed the first injection anesthesia experiments on himself.[1] It was first marketed in 1949. Etymology: from one of its many chemical names - [alpha-Diethylamino-2,6-dimethylacetani- ] - lide + ~ocaine.
Indications
The efficacy profile of lidocaine as a local anesthetic is characterized by a rapid onset of action and intermediate duration of efficacy. Therefore, lidocaine is suitable for infiltration, block and surface anesthesia. Longer-acting substances such as bupivacaine are sometimes given preference for spinal and peridural anesthesias; lidocaine, on the other hand, has the advantage of a rapid onset of action. Adrenaline vasoconstricts arteries and hence delays the resorption of Lidocaine, almost doubling the duration of anaesthesia. For surface anesthesia several formulations are available that can be used e.g. for endoscopies, before intubations etc. Buffering the pH of lidocaine makes local freezing less painful.[2]
Topical lidocaine has been shown to relieve postherpetic neuralgia (arising, for example, from shingles) in some patients, though there is not enough study evidence to recommend it as a first-line treatment.[3] It also has uses as a temporary fix for tinnitus. Although not completely curing the illness, it has been shown to reduce the effects by around two thirds.[4]
Lidocaine is also the most important class 1B antiarrhythmic drug: it is used intravenously for the treatment of ventricular arrhythmias (for acute myocardial infarction, digitalis poisoning, cardioversion or cardiac catheterization). However, a routine prophylactic administration is no longer recommended for acute cardiac infarction; the overall benefit of this measure is not convincing.
Lidocaine has also been efficient in refractory cases of status epilepticus.
Inhaled lidocaine can be used as an antitussive (cough suppressor) acting peripherally below the larynx.[5] [6]
Lidocaine has also proved effective in treating jellyfish stings, both numbing the affected area and preventing further nematocyst discharge [7]
Contraindications
Contraindications for the use of lidocaine include:
- Heart block, second or third degree (without pacemaker)
- Severe sinoatrial block (without pacemaker)
- Serious adverse drug reaction to lidocaine or amide local anaesthetics
- Concurrent treatment with quinidine, flecainide, disopyramide, procainamide (Class I antiarrhythmic agents)
- Prior use of Amiodarone hydrochloride
- Hypotension not due to Arrhythmia
- Bradycardia
- Accelerated idioventricular rhythm
- Pacemaker
- Porphyria, especially acute porphyria (AIP); lidocaine is known to be porphyrogenic although similar drugs (e.g. bupivacaine, tetracaine) are known to be safe.[8][9]
Adverse effects
Adverse drug reactions (ADRs) are rare when lidocaine is used as a local anesthetic and is administered correctly. Most ADRs associated with lidocaine for anesthesia relate to administration technique (resulting in systemic exposure) or pharmacological effects of anesthesia, but allergic reactions only rarely occur.[10]
Systemic exposure to excessive quantities of lidocaine mainly result in central nervous system (CNS) and cardiovascular effects – CNS effects usually occur at lower blood plasma concentrations and additional cardiovascular effects present at higher concentrations, though cardiovascular collapse may also occur with low concentrations. CNS effects may include CNS excitation (nervousness, tingling around the mouth (also known as circumoral paraesthesia), tinnitus, tremor, dizziness, blurred vision, seizures) followed by depression, and with increasingly heavier exposure: drowsiness, loss of consciousness, respiratory depression and apnoea). Cardiovascular effects include hypotension, bradycardia, arrhythmias, and/or cardiac arrest – some of which may be due to hypoxemia secondary to respiratory depression.[11]
ADRs associated with the use of intravenous lidocaine are similar to toxic effects from systemic exposure above. These are dose-related and more frequent at high infusion rates (≥3 mg/minute). Common ADRs include: headache, dizziness, drowsiness, confusion, visual disturbances, tinnitus, tremor, and/or paraesthesia. Infrequent ADRs associated with the use of lidocaine include: hypotension, bradycardia, arrhythmias, cardiac arrest, muscle twitching, seizures, coma, and/or respiratory depression.[11]
Overdosage
Overdosage with lidocaine can be a result of excessive administration via topical or parenteral routes, accidental oral ingestion of topical preparations by children, accidental intravenous (rather than subcutaneous, intrathecal or paracervical) injection or prolonged use of subcutaneous infiltration anesthesia during cosmetic surgical procedures. These occurrences have often led to severe toxicity or death in both children and adults. Lidocaine and its two major metabolites may be quantified in blood, plasma or serum to confirm the diagnosis in potential poisoning victims or to assist in the forensic investigation in a case of fatal overdosage. It is important in the interpretation of analytical results to recognize that lidocaine is often routinely administered intravenously as an antiarrhthymic agent in critical cardiac care situations.[12] Treatment with intravenous lipid emulsions (used for parental feeding) to reverse the effects of local anaesthetic toxicity is becoming more common place. [13]
Insensitivity to lidocaine
Relative insensitivity to lidocaine is genetic. In hypokalemic sensory overstimulation, relative insensitivity to lidocaine has been described in people who also have attention deficit hyperactivity disorder. In dental anesthesia, a relative insensitivity to lidocaine can occur for anatomical reasons due to unexpected positions of nerves. Some people with Ehlers-Danlos syndrome are insensitive to lidocaine.[14]
Dosage forms
Lidocaine, usually in the form of lidocaine hydrochloride, is available in various forms including:
- Injected local anesthetic (sometimes combined with epinephrine to reduce bleeding)
- Dermal patch (sometimes combined with prilocaine)
- Intravenous injection (sometimes combined with epinephrine to reduce bleeding)
- Intravenous infusion
- Nasal instillation/spray (combined with phenylephrine)
- Oral gel (often referred to as "viscous lidocaine" or abbreviated "lidocaine visc" or "lidocaine hcl visc" in pharmacology; used as teething gel)
- Oral liquid
- Topical gel (as with Aloe vera gels that include lidocaine)[15]
- Topical liquid
- Topical patch (lidocaine 5% patch is marketed as "Lidoderm" in the US (since 1999) and "Versatis" in the UK (since 2007 by Grünenthal))
- Topical aerosol spray
- Inhaled via a nebulizer
Adulterant in cocaine
Lidocaine is often added to cocaine as a diluent.[16] Cocaine numbs the gums when applied, and since lidocaine causes stronger numbness,[17] a user gets the impression of high-quality cocaine when in actuality, the user is receiving a diluted product.[18]
Preparation
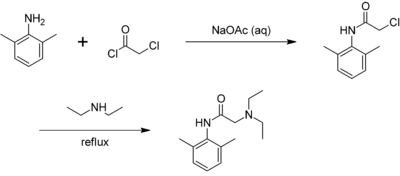
Lidocaine may be prepared in two steps by the reaction of 2,6-xylidine with chloroacetyl chloride, followed by the reaction with diethylamine:[19][20]
Pharmacokinetics
Lidocaine is approximately 95% metabolized (dealkylated) in the liver by CYP3A4 to the pharmacologically-active metabolites monoethylglycinexylidide (MEGX) and then subsequently to the inactive glycine xylidide. MEGX has a longer half life than lidocaine but also is a less potent sodium channel blocker.[21]
The elimination half-life of lidocaine is approximately 90–120 minutes in most patients. This may be prolonged in patients with hepatic impairment (average 343 minutes) or congestive heart failure (average 136 minutes).[22]
Pharmacodynamics
Anesthesia
Lidocaine alters signal conduction in neurons by blocking the fast voltage gated sodium (Na+) channels in the neuronal cell membrane that are responsible for signal propagation.[23] With sufficient blockage the membrane of the postsynaptic neuron will not depolarize and will thus fail to transmit an action potential. This creates the anaesthetic effect by not merely preventing pain signals from propagating to the brain but by stopping them before they begin. Careful titration allows for a high degree of selectivity in the blockage of sensory neurons, whereas higher concentrations will also affect other modalities of neuron signaling.
Illegal uses
Lidocaine is not currently listed by the World Anti-Doping Agency as an illegal substance.[24] Lidocaine is used as an adjuvant, adulterant, mimic and diluent to illegal street drugs (e.g. cocaine). (See U.S. DEA web site).
Compendial status
- Japanese Pharmacopoeia 15
- United States Pharmacopeia 31[25]
See also
Notes and references
- ^ a b Nils Löfgren (1948). Xylocaine: a new synthetic drug.
- ^ Cepeda MS, Tzortzopoulou A, Thackrey M, Hudcova J, Arora Gandhi P, Schumann R (2010). Tzortzopoulou, Aikaterini. ed. "Adjusting the pH of lidocaine for reducing pain on injection". Cochrane Database Syst Rev 12 (12): CD006581. doi:10.1002/14651858.CD006581.pub2. PMID 21154371.
- ^ Khaliq W, Alam S, Puri N (2007). Khaliq, Waqas. ed. "Topical lidocaine for the treatment of postherpetic neuralgia". Cochrane Database Syst Rev (2): CD004846. doi:10.1002/14651858.CD004846.pub2. PMID 17443559.
- ^ http://newsvote.bbc.co.uk/1/hi/health/7175306.stm
- ^ RSD931, a novel antitussive agent acting on airway sensory nerves
- ^ Inhaled lidocaine is used to suppress cough during bronchoscopy. Animal studies and a few human studies suggest that lidocaine has an antitussive effect...
- ^ 17. Birsa LM, Verity PG, Lee RF, (2010). "Evaluation of the effects of various chemicals on discharge of and pain caused by jellyfish nematocysts". Comparative Biochemistry and Physiology, Part C 151 (4): 426–430. doi:10.1016/j.cbpc.2010.01.007. PMID 20116454.
- ^ http://db.porphyriafoundation.com/forms/drug-list.lasso
- ^ www.merck.com/media/mmpe/pdf/Table_155-4.pdf
- ^ D Jackson, AH Chen, and CR Bennett (1994). "Identifying true lidocaine allergy". J Am Dent Assoc 125 (10): 1362–1366. PMID 7844301. http://www.ncbi.nlm.nih.gov/pubmed/7844301.
- ^ a b Rossi S, editor. Australian Medicines Handbook 2006. Adelaide: Australian Medicines Handbook; 2006. ISBN 0-9757919-2-3
- ^ R. Baselt, Disposition of Toxic Drugs and Chemicals in Man, 8th edition, Biomedical Publications, Foster City, CA, 2008, pp. 840-844.
- ^ Pickard J Guidelines and the adoption of 'lipid rescue' therapy for local anaesthetic toxicity. Anaesthesia 2009;64:122-5
- ^ Grahame, Rodney; Grahame, R; Norris, P; Hopper, C (2005). "Local anaesthetic failure in joint hypermobility syndrome". Journal of the Royal Society of Medicine 98 (2): 84–85. doi:10.1258/jrsm.98.2.84. PMC 1079398. PMID 15684369. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1079398.
- ^ Solarcaine topical gel, July 27, 2009
- ^ Naissa Prévide Bernardo, Maria Elisa Pereira Bastos Siqueira, Maria José Nunes de Paiva, Patrícia Penido Maia (2003). "Caffeine and other adulterants in seizures of street cocaine in Brazil". International Journal of Drug Policy 14 (4): 331–334. doi:10.1016/S0955-3959(03)00083-5. http://www.sciencedirect.com/science/article/B6VJX-497HGRX-5/2/3b5f81654f1e907ceb3095b4b5207362.
- ^ Howell, Kimberly. "Take a big-picture approach when dealing with corneal sensation". http://www.eyeplastics.org/topics/thyroid/thyroid_news/corneal_sensation.htm. Retrieved 2009-04-23. "Lidocaine is more potent, with rapid diffusion and penetration."[dead link]
- ^ 599 F.2d 635
- ^ T. J. Reilly (1999). "The Preparation of Lidocaine". J. Chem. Ed. 76 (11): 1557. doi:10.1021/ed076p1557. http://jchemed.chem.wisc.edu/Journal/Issues/1999/Nov/abs1557.html.
- ^ J.L. Bengt, M.L. Niles, U.S. Patent 2,441,498 (1948).
- ^ Flomenbaum NE, Howland MA, Goldfrank, LR, et al., ed (2006). Goldfrank's Toxicologic Emergencies, 8th ed.. McGraw-Hill.
- ^ Thomson PD, Melmon KL, Richardson JA, et al. (1973). "Lidocaine pharmacokinetics in advanced heart failure, liver disease, and renal failure in humans". Ann. Intern. Med. 78 (4): 499–508. PMID 4694036.
- ^ Catterall WA (2002). "Molecular mechanisms of gating and drug block of sodium channels". Novartis Found Symp. Novartis Foundation Symposia 241: 206–32. doi:10.1002/0470846682.ch14. ISBN 9780470846681. PMID 11771647.
- ^ http://www.wada-ama.org/Documents/World_Anti-Doping_Program/WADP-Prohibited-list/WADA_Prohibited_List_2010_EN.pdf
- ^ Revision Bulletin: Lidocaine and Prilocaine Cream–Revision to Related Compounds Test
External links
Antiarrhythmic agents (C01B) Channel blockers Amiodarone • Dronedarone • Bretylium • Bunaftine • Dofetilide • Ibutilide • Nifekalant • Sotalol • Tedisamil • Vernakalant • E-4031Receptor agonists
and antagonistsIon transporters Vasoprotectives (C05) Antihemorrhoidals for topical use corticosteroids (Hydrocortisone, Prednisolone, Betamethasone, Fluorometholone, Fluocortolone, Dexamethasone, Fluocinolone acetonide, Fluocinonide)
local anesthetics (Lidocaine, Tetracaine, Benzocaine, Cinchocaine, Procaine, Oxetacaine, Pramocaine)
other (Tribenoside)Antivaricose therapy heparins or heparinoids for topical use (Organo-heparinoid, Sodium apolate, Heparin, Pentosan polysulfate)
sclerosing agents for local injection (Monoethanolamine oleate, Polidocanol, Invert sugar, Sodium tetradecyl sulfate, Phenol)
other (Calcium dobesilate)Capillary stabilising agents bioflavonoids (Rutoside, Monoxerutin, Diosmin, Troxerutin, Hidrosmin) - other (Tribenoside, Etamsylate)Antipruritics (D04) Antihistamines for topical use Anesthetics for topical use Anesthetics: Local anesthetics - primarily sodium channel blockers (N01B) Esters Amylocaine • Benzocaine • Butacaine • Butamben • Chloroprocaine • Dimethocaine • Meprylcaine • Metabutoxycaine • Orthocaine • Propoxycaine • Procaine (Novocaine) • Proxymetacaine • Risocaine • TetracaineAmides Articaine • Bupivacaine # /Levobupivacaine/Ropivacaine • Carticaine • Cinchocaine • Etidocaine • Lidocaine # • Mepivacaine • Prilocaine • TrimecaineCombinations Throat preparations (R02) Antiseptics Acriflavinium chloride • Ambazone • Benzalkonium • Benzethonium • Cetrimonium (bromide/chloride) • Cetylpyridinium • Chlorhexidine • Chlorquinaldol • Dequalinium • Dichlorobenzyl alcohol • Hexamidine • Hexylresorcinol • Myristyl-benzalkonium • Oxyquinoline • Phenol • Povidone-iodineAntibiotics Local anesthetics Other Otologicals (S02) Anti-infectives Acetic acid • Aluminium acetotartrate • Boric acid • Chloramphenicol • Chlorhexidine • Ciprofloxacin • Clioquinol • Gentamicin • Hydrogen peroxide • Miconazole • Neomycin • Nitrofurazone • Ofloxacin • Polymyxin B • Rifamycin • TetracyclineCorticosteroids Analgesics and anesthetics M: EAR
anat(e/p)/phys/devp
noco/cong, epon
proc, drug(S2)
Categories:- Antiarrhythmic agents
- Local anesthetics
- Sodium channel blockers
- World Health Organization essential medicines
- Acetamides
Wikimedia Foundation. 2010.