- Benzalkonium chloride
-
Benzalkonium chloride 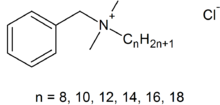 benzyl-dimethyl-tridecyl-azanium chlorideOther namesN-decyl-N-benzyl-N,N-dimethylammonium chloride (with n=10 for alkyl side chain)
benzyl-dimethyl-tridecyl-azanium chlorideOther namesN-decyl-N-benzyl-N,N-dimethylammonium chloride (with n=10 for alkyl side chain)Identifiers CAS number 8001-54-5 
ChemSpider 8423  =
=UNII F5UM2KM3W7 
EC number 264-151-6 KEGG D00857 
ATC code D08, D09AA11, R02AA16 Properties Molecular formula variable Molar mass variable Appearance white or yellow powder; gelatinous lumps; colorless solution Density 0.98 g/cm3 Solubility in water very soluble Hazards EU classification C, N [1] R-phrases R21/22, R34, R50 [1] S-phrases S36/37/39, S45, S61 [1] NFPA 704 Flash point 250 °C  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Benzalkonium chloride, also known as alkyldimethylbenzylammonium chloride and ADBAC, is a mixture of alkylbenzyldimethylammonium chlorides of various even-numbered[2] alkyl chain lengths. This product is a nitrogenous cationic surface-acting agent belonging to the quaternary ammonium group. It has three main categories of use; as a biocide, a cationic surfactant and phase transfer agent in the chemical industry.
Contents
Properties
Benzalkonium chloride is readily soluble in ethanol and acetone. Although dissolution in water is slow, aqueous solutions are easier to handle and are preferred. Solutions should be neutral to slightly alkaline, with colour ranging from colourless to a pale yellow. Solutions foam profusely when shaken, have a bitter taste and a faint almond-like odour which is only detectable in concentrated solutions.
Availability
Standard concentrates are manufactured as 50% and 80% w/w solutions, and sold under trade names such as BC50, BC80, BAC50, BAC80, etc. The 50% solution is purely aqueous, while more concentrated solutions require incorporation of rheology modifiers (alcohols, polyethylene glycols, etc.) to prevent increases in viscosity or gel formation under low temperature conditions.
Applications
The applications are extremely wide ranging, from disinfectant formulations, such as being an active ingredient in Dettol and Lysol brand products, to microbial corrosion inhibition in the oilfield sector, and a multi-surface mould, algae and moss remover.[3] It is used in:
- Pharmaceuticals such as leave-on skin antiseptics
- Antiseptic in Bactine to safely treat childhood scrapes and cuts.
- Advanced, next-generation hand sanitizers
- Hygienic towelettes and wet wipes
- Cosmetics such as eye and nasal drops, as a preservative
- Cleaners for floor and hard surfaces as a disinfectant
- High-level surgical instrument sterilizing and disinfection solutions
- Air and surface sprayable disinfectants
- Over-the-counter herpes cold sore and fever blister single-application treatments such as RELEEV and Viroxyn
- Algaecide
Benzalkonium chloride and other alcohol-free solutions are often used in preparations used for skin disinfection prior to use of syringes.[citation needed]
New generation, plant-derived versions are used in hand sanitizers that are more effective and safer than alcohol gels. As an antiseptic, it has the advantage of not burning when put on a wound, which is not the case with ethanol-based antiseptics or hydrogen peroxide. As the antiseptic in Bactine, benzalkonium chloride has been used safely for decades on childhood scrapes and cuts.
Biological activity
The greatest biocidal activity is associated with the C12-C14 alkyl derivatives. The mechanism of bactericidal/microbicidal action is thought to be due to disruption of intermolecular interactions. This can cause dissociation of cellular membrane lipid bilayers, which compromises cellular permeability controls and induces leakage of cellular contents. Other biomolecular complexes within the bacterial cell can also undergo dissociation. Enzymes, which finely control a wide range of respiratory and metabolic cellular activities, are particularly susceptible to deactivation. Critical intermolecular interactions and tertiary structures in such highly specific biochemical systems can be readily disrupted by cationic surfactants.
Benzalkonium chloride solutions are fast-acting biocidal agents with a moderately long duration of action. They are active against bacteria and some viruses, fungi, and protozoa. Bacterial spores are considered to be resistant. Solutions are bacteriostatic or bactericidal according to their concentration. Gram-positive bacteria are generally more susceptible than Gram-negative. Activity is not greatly affected by pH, but increases substantially at higher temperatures and prolonged exposure times. In a 1998 study utilizing the FDA protocol, a non-alcohol sanitizer utilizing the active ingredient benzalkonium chloride met the FDA performance standards, while Purell, a popular alcohol-based sanitizer, did not. The study found that a benzalkonium chloride-based sanitizer was the most favorable non-alcohol-based hand sanitizer.[4] Advancements in the quality and efficacy of benzalkonium chloride in current non-alcohol hand sanitizers has addressed the CDC concerns regarding gram negative bacteria, with the leading products being equal if not more effective against gram negative, particularly NDM1 and other antibiotic resistant bacteria.
Newer formulations using benzalkonium blended with various quaternary ammonium derivatives can be used to extend the biocidal spectrum and enhance the efficacy of benzalkonium based disinfection products. This technique has been used to improve virucidal activity of quaternary ammonium-based formulations to healthcare infection hazards such as hepatitis, HIV, etc. Quaternary ammonium formulations are now the disinfectants of choice for hospitals. This is on account of user and patient safety even on contact with treated surfaces and the absence of harmful fumes. Benzalkonium solutions for hospital use tend to be neutral to alkaline, non-corrosive on metal surfaces, non-staining, and safe to use on all washable surfaces.
The use of appropriate supporting excipients can also greatly improve efficacy and detergency, and prevent deactivation under use conditions. Formulation requires great care, as benzalkonium solutions can readily be inactivated in the presence of organic and inorganic contamination. Solutions are incompatible with soaps, and must not be mixed with anionic surfactants. Hard water salts can also reduce biocidal activity. As with any disinfectant, it is recommended that surfaces are free from visible dirt and interfering materials for maximal disinfection performance by quaternary ammonium products.
Although hazardous levels are not likely to be reached under normal use conditions, benzalkonium and other detergents can pose a hazard to marine organisms. Quaternary ammonium disinfectants are effective at very low ppm levels, so excess use should be avoided.
Safety
ADBAC is highly toxic to fish (LC50 = 280 μg ai/L), very highly toxic to aquatic invertebrates (LC50 = 5.9 μg ai/L), moderately toxic to birds (LD50 = 136 mg/kg-bw), and slightly toxic to mammals (LD50 = 430 mg/kg-bw).[5] Benzalkonium chloride solutions of 10% or more are toxic to humans, causing irritation to the skin and mucosa, and death if taken internally.[6]
Several studies have identified allergic reactions to benzalkonium chloride by some individuals[7][8][9][10][11][12][13] but several studies have cast doubt on its reputation for other negative health effects.[14][15] It is still widely used in eyewashes, nasal sprays, hand and face washes, mouthwashes, spermicidal creams, and in various other cleaners, sanitizers, and disinfectants. Manufacturers of over-the-counter artificial tears and eye washes became concerned about chemical sensitivity from long-term daily use and have in some products substituted EDTA as a preservative. Some have packaged eye drops in single-use vials with no preservative[16] for sensitive eyes. There has also been concern that long-term use of benzalkonium as a preservative in nasal sprays may cause swelling of mucosa and lead to rhinitis medicamentosa. Although some studies have found no correlation between use of benzalkonium chloride in nasal sprays and rhinitis medicamentosa,[17] others have found benzalkonium chloride in oxymetazoline nasal spray to worsen rhinitis medicamentosa in healthy volunteers after both long-term use[18][19] and short-term use.[20]
Contact lens solutions typically contain 0.002% to 0.01% benzalkonium chloride.[21] K. C. Swan[22] found that repeated use of benzalkonium chloride at concentrations of 1:5000 (0.02%) or stronger can denature corneal protein and cause irreversible damage to the eye. Swan also found that 0.04% to 0.05% solutions of benzalkonium chloride can cause punctures of the corneal epithelium. The disadvantages of using benzalkonium chloride solutions with contact lenses are also discussed in the literature.[23] [24]
A disinfectant containing benzalkonium chloride and the related compound didecyldimethylammonium chloride (DDAC) has been identified as the most probable cause of birth defects and fertility problems in caged mice.[25]
A 2009 study has found that benzalkonium chloride, when used in less than lethal concentrations, resulted in an increased resistance to the disinfectant solution, and a 256-fold increase in resistance of the bacteria Pseudomonas aeruginosa to the ciprofloxacin antibiotic, even though the bacterial colonies had not been previously exposed to the antibiotic.[26]
See also
- Stearalkonium chloride
- Polyaminopropyl biguanide, an alternative preservative for contact lens solutions
References
- ^ a b c Record of Quaternary ammonium compounds, benzyl-C8–18-alkyldimethyl, chlorides in the European chemical Substances Information System ESIS
- ^ U.S. Environmental Protection Agency: Reregistration Eligibility Decision for Alkyl Dimethyl Benzyl Ammonium Chloride (ADBAC)
- ^ Sika mould remover as an example
- ^ David Dyer Kenneth Gerenraich Peter Whams (1998). Testing a New Alcohol-Free Hand Sanitizer to Combat Infection. AORN Journal Vol 68 Issue 2. pp. 239–251. http://www.aornjournal.org/article/S0001-2092%2806%2962517-9/abstract.
- ^ Frank T. Sanders, ed (August 2006). Reregistration Eligibility Decision for Alkyl Dimethyl Benzyl Ammonium Chloride (ADBAC) (Report). U.S. Environmental Protection Agency Office of Prevention, Pesticides, and Toxic Substances. pp. 114. http://www.epa.gov/oppsrrd1/REDs/adbac_red.pdf. Retrieved 2009-03-31.
- ^ Seymour Stanton Block (2001). Disinfection, sterilization, and preservation (5, illustrated ed.). Lippincott Williams & Wilkins. p. 311. ISBN 0683307401. http://books.google.com/?id=3f-kPJ17_TYC&pg=PA303&lpg=PA303.
- ^ Park HJ, Kang HA, Lee JY, Kim HO (2000). "Allergic contact dermatitis from benzalkonium chloride in an antifungal solution". Contact Derm. 42 (5): 306–7. PMID 10789868.
- ^ Liu H, Routley I, Teichmann KD (2001). "Toxic endothelial cell destruction from intraocular benzalkonium chloride". J Cataract Refract Surg 27 (11): 1746–50. doi:10.1016/S0886-3350(01)01067-7. PMID 11709246.
- ^ Chiambaretta F, Pouliquen P, Rigal D (1997). "[Allergy and preservatives. Apropos of 3 cases of allergy to benzalkonium chloride]" (in French). J Fr Ophtalmol 20 (1): 8–16. PMID 9099278.
- ^ Wong DA, Watson AB (2001). "Allergic contact dermatitis due to benzalkonium chloride in plaster of Paris". Australasian J. Dermatology 42 (1): 33–5. doi:10.1046/j.1440-0960.2001.00469.x. PMID 11233718.
- ^ Kanerva L, Jolanki R, Estlander T (2000). "Occupational allergic contact dermatitis from benzalkonium chloride". Contact Derm. 42 (6): 357–8. PMID 10871106.
- ^ Oiso N, Fukai K, Ishii M (2005). "Irritant contact dermatitis from benzalkonium chloride in shampoo". Contact Derm. 52 (1): 54. doi:10.1111/j.0105-1873.2005.0483j.x. PMID 15701139.
- ^ Basketter DA, Marriott M, Gilmour NJ, White IR (2004). "Strong irritants masquerading as skin allergens: the case of benzalkonium chloride". Contact Derm. 50 (4): 213–7. doi:10.1111/j.0105-1873.2004.00331.x. PMID 15186375.
- ^ Graf P (2001). "Benzalkonium chloride as a preservative in nasal solutions: re-examining the data". Respir Med 95 (9): 728–33. doi:10.1053/rmed.2001.1127. PMID 11575893.
- ^ Marple B, Roland P, Benninger M (2004). "Safety review of benzalkonium chloride used as a preservative in intranasal solutions: an overview of conflicting data and opinions". Otolaryngol Head Neck Surg 130 (1): 131–41. doi:10.1016/j.otohns.2003.07.005. PMID 14726922.
- ^ Use of benzalkonium in ocular preparations and eye drops
- ^ Marple, B; Roland, P; Benninger, M (2004). "Safety review of benzalkonium chloride used as a preservative in intranasal solutions: an overview of conflicting data and opinions.". Otolaryngology--head and neck surgery : official journal of American Academy of Otolaryngology-Head and Neck Surgery 130 (1): 131–41. doi:10.1016/j.otohns.2003.07.005. PMID 14726922.
- ^ . doi:10.1111/j.1365-2222.1995.tb01069.x9.
- ^ Graf, P (2005). "Rhinitis medicamentosa: a review of causes and treatment.". Treatments in respiratory medicine 4 (1): 21–9. PMID 15725047.
- ^ Graf, P; Enerdal, J; Hall�n, H (1999). "Ten days' use of oxymetazoline nasal spray with or without benzalkonium chloride in patients with vasomotor rhinitis.". Archives of otolaryngology--head & neck surgery 125 (10): 1128–32. PMID 10522506.
- ^ U.S. Patent 5,725,887, column 2, line 8
- ^ Swan, K. C., "Reactivity of the Ocular Tissues to Wetting Agents", Am. J. Ophthalmol., 27, 118 (1944),
- ^ Otten, Mary; Szabocsik, John M. (1976). "Measurement of Preservative Binding with SOFLENS® (polymacon) Contact Lens". Clinical and Experimental Optometry 59 (8): 277. doi:10.1111/j.1444-0938.1976.tb01445.x.
- ^ M Akers, "Consideration in selecting antimicrobial preservative agents for parenteral product development", Pharmaceutical Technology, May, p. 36 (1984).
- ^ Hunt, P (Jun 2008). "Lab disinfectant harms mouse fertility. Patricia Hunt interviewed by Brendan Maher.". Nature 453 (7198): 964. doi:10.1038/453964a. PMID 18563110.
- ^ Mc Cay, P. H.; Ocampo-Sosa, A. A.; Fleming, G. T. A. (2009). "Effect of subinhibitory concentrations of benzalkonium chloride on the competitiveness of Pseudomonas aeruginosa grown in continuous culture". Microbiology 156 (Pt 1): 30–8. doi:10.1099/mic.0.029751-0. PMID 19815578.
Further reading
- Bernstein IL: Is the use of benzalkonium chloride as a preservative for nasal formulations a safety concern? J Allergy Clin Immunol 2000 Jan; 105(1 Pt 1): 39-44.
- Graf P: Adverse effects of benzalkonium chloride on the nasal mucosa: allergic rhinitis and rhinitis medicamentosa. Clin Ther 1999 Oct; 21(10): 1749-55.
- Graf P, Hallen H, Juto JE: Benzalkonium chloride in a decongestant nasal spray aggravates rhinitis medicamentosa in healthy volunteers. Clin Exp Allergy 1995 May; 25(5): 395-400.
External links
Categories:- Chlorides
- Quaternary ammonium compounds
- Cationic surfactants
- Antiseptics
- Disinfectants
- Corrosion inhibitors
Wikimedia Foundation. 2010.

