- Morphine-N-oxide
-
Morphine-N-oxide 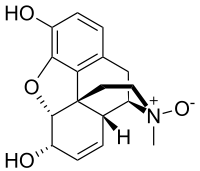 (4R,4aR,7S,7aR,12bS)-3-Methyl-2,3,4,4a,7,7a-hexahydro-1H-4,12-methano[1]benzofuro[3,2-e]isoquinoline-7,9-diol 3-oxide[citation needed]
(4R,4aR,7S,7aR,12bS)-3-Methyl-2,3,4,4a,7,7a-hexahydro-1H-4,12-methano[1]benzofuro[3,2-e]isoquinoline-7,9-diol 3-oxide[citation needed]Identifiers CAS number 639-46-3 
PubChem 5362459 ChemSpider 4515047 
EC number 211-355-8 KEGG C11786 
Jmol-3D images Image 1
Image 2- CN1(=O)CC[C@@]23[C@H]4Oc5c2c(C[C@@H]1[C@@H]3C=C[C@@H]4O)ccc5O
CN1(=O)CC[C@]23[C@H]4OC5=C(O)C=CC(C[C@@H]1[C@@H]2C=C[C@@H]4O)=C35
Properties Molecular formula C17H19NO4 Molar mass 301.34 g mol−1 Exact mass 301.131408101 g mol−1 Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox references Morphine-N-oxide (genomorphine) is an active opioid metabolite of morphine. Morphine itself, in trials with rats, acts 11–22 times more potent than morphine-N-oxide subcutaneously and 39–89 times more potent intraperitoneally. However pretreatment with amiphenazole or tacrine increases the potency of morphine-N-oxide in relation to morphine (intraperitoneally more so than in subcutaneous administration). A possible explanation is that morphine-N-oxide is rapidly inactivated in the liver and impairment of inactivation processes or enzymes increase functionality.[1]
See also
References
- ^ The analgesic action of morphine-N-oxide. M. R. Fennessy. Br J Pharmacol. 1968 October; 34(2): 337–344. PMCID: PMC1703337

This analgesic-related article is a stub. You can help Wikipedia by expanding it. - CN1(=O)CC[C@@]23[C@H]4Oc5c2c(C[C@@H]1[C@@H]3C=C[C@@H]4O)ccc5O
