- Anandamide
-
Anandamide 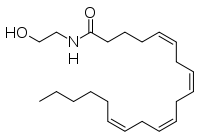 (5Z,8Z,11Z,14Z)-N-(2-hydroxyethyl)icosa-5,8,11,14-tetraenamideOther namesN-arachidonoylethanolamine
(5Z,8Z,11Z,14Z)-N-(2-hydroxyethyl)icosa-5,8,11,14-tetraenamideOther namesN-arachidonoylethanolamine
arachidonoylethanolamideIdentifiers CAS number 94421-68-8 
PubChem 5281969 ChemSpider 4445241 
UNII UR5G69TJKH 
MeSH Anandamide ChEBI CHEBI:2700 
ChEMBL CHEMBL15848 
IUPHAR ligand 737 Jmol-3D images Image 1
Image 2- O=C(NCCO)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC
CCCCC/C=C\C/C=C\C/C=C\C/C=C\CCCC(=O)NCCO
- InChI=1S/C22H37NO2/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-22(25)23-20-21-24/h6-7,9-10,12-13,15-16,24H,2-5,8,11,14,17-21H2,1H3,(H,23,25)/b7-6-,10-9-,13-12-,16-15-

Key: LGEQQWMQCRIYKG-DOFZRALJSA-N
InChI=1/C22H37NO2/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-22(25)23-20-21-24/h6-7,9-10,12-13,15-16,24H,2-5,8,11,14,17-21H2,1H3,(H,23,25)/b7-6-,10-9-,13-12-,16-15-
Key: LGEQQWMQCRIYKG-DOFZRALJBA
Properties Molecular formula C22H37NO2 Molar mass 347.53 g/mol  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Anandamide, also known as N-arachidonoylethanolamide or AEA, is an endogenous cannabinoid neurotransmitter. The name is taken from the Sanskrit word ananda, which means "bliss, delight", and amide.[1][2] It is synthesized from N-arachidonoyl phosphatidylethanolamine by multiple pathways.[3] It is degraded primarily by the fatty acid amide hydrolase (FAAH) enzyme, which converts anandamide into ethanolamine and arachidonic acid. As such, inhibitors of FAAH lead to elevated anandamide levels and are being pursued for therapeutic use.[4][5]
Contents
History
It was isolated and its structure was deciphered by a research group led by professor Raphael Mechoulam of the Hebrew University in Jerusalem, which included a post doctoral fellow, Dr. William Devane, a visiting Czech analytical chemist Dr. Lumír Hanuš and Dr. Aviva Breuer in 1992. Additional collaborators were Professor Roger Pertwee from Edinburgh and Professor Asher Mandelbaum of the Technion in Haifa.
Physiological functions
Anandamide's effects can be either central, in the brain, or peripheral, in other parts of the body. These distinct effects are mediated primarily by CB1 cannabinoid receptors in the central nervous system, and CB2 cannabinoid receptors in the periphery.[6] The latter are mainly involved in functions of the immune system. Cannabinoid receptors were originally discovered as being sensitive to Δ9-tetrahydrocannabinol (Δ9-THC, commonly called THC), which is the primary psychoactive cannabinoid found in cannabis. The discovery of anandamide came from research into CB1 and CB2, as it was inevitable that a naturally occurring (endogenous) chemical would be found to affect these receptors.
Moreover, anandamide is thought to be an endogenous ligand for vanilloid receptors (which are involved in the transduction of acute and inflammatory pain signals), activating the receptor in a PKC-dependent (protein kinase C-dependent) manner.[citation needed]
Anandamide has shown to impair working memory in rats.[7] Studies are under way to explore what role anandamide plays in human behavior, such as eating and sleep patterns, and pain relief.
Anandamide is also important for implantation of the early stage embryo in its blastocyst form into the uterus. Therefore cannabinoids such as Δ9-THC might influence processes during the earliest stages of human pregnancy.[8] Peak plasma anandamide occurs at ovulation and positively correlates with peak estradiol and gonadotrophin levels, suggesting that these may be involved in the regulation of AEA levels.[9]
Anandamide is also important in the regulation of feeding behavior, and the neural generation of motivation and pleasure. In addition, anandamide injected directly into the forebrain reward-related brain structure nucleus accumbens enhances the pleasurable responses of rats to a rewarding sucrose taste, and enhances food intake as well.[6][10]
A study published in 1998 shows that anandamide inhibits human breast cancer cell proliferation.[11]
In an attempt to find anandamide or anandamide type endocannabinoids, researchers in 1996 found N-oleoylethanolamine and N-linoleoylethanolamine which may act as cannabinoid mimics.[12] Later studies confirmed that anandamide is not found in chocolate and the two compounds reported earlier did not interact with cannabinoid receptors. The neurological basis of chocolate's appeal still remains elusive.[13]
 Lumír Ondřej Hanuš (left), discoverer of endogenous ligand, anandamide, from brain (1992) and Raphael Mechoulam (right), discoverer of psychoactive compound, (-)-trans-delta-9-tetrahydrocannabinol, from Cannabis sativa L. (1964). Both compounds bind to the cannabinoid receptors in the brain.
Lumír Ondřej Hanuš (left), discoverer of endogenous ligand, anandamide, from brain (1992) and Raphael Mechoulam (right), discoverer of psychoactive compound, (-)-trans-delta-9-tetrahydrocannabinol, from Cannabis sativa L. (1964). Both compounds bind to the cannabinoid receptors in the brain.
Synthesis and degradation
The human body synthesizes anandamide from N-arachidonoyl phosphatidylethanolamine (NAPE), which is itself made by transferring arachidonic acid from lecithin to the free amine of cephalin through an N-acyltransferase enzyme.[14][15] Anandamide synthesis from NAPE occurs via multiple pathways and includes enzymes such as phospholipase A2, phospholipase C and NAPE-PLD.[3]
Endogenous anandamide is present at very low levels and has a very short half-life due to the action of the enzyme fatty acid amide hydrolase (FAAH), which breaks it down into free arachidonic acid and ethanolamine. Studies of piglets show that dietary levels of arachidonic acid and other essential fatty acids affect the levels of anandamide and other endocannabinoids in the brain.[16] High fat diet feeding in mice increases levels of anandamide in the liver and increases lipogenesis.[17] This suggests that anandamide may play a role in the development of obesity, at least in rodents.
Paracetamol (or acetaminophen in the U.S.A.) is metabolically combined with arachidonic acid by FAAH to form AM404.[18] This metabolite of paracetamol is a potent agonist at the TRPV1 vanilloid receptor, a weak agonist at both CB1 and CB2 receptors, and an inhibitor of anandamide reuptake. As a result, anandamide levels in the body and brain are elevated. In this fashion, paracetamol acts as a pro-drug for a cannabimimetic metabolite. This action may be partially or fully responsible for the analgesic effects of paracetamol.[19][20]
See also
- Cannabinoids
- Virodhamine
- Tetrahydrocannabinol (THC)
- 2-Arachidonoylglycerol
- Fatty acid amide hydrolase
References
- ^ Devane WA, Hanus L, Breuer A, Pertwee RG, Stevenson LA, Griffin G, Gibson D, Mandelbaum A, Etinger A, Mechoulam R (December 1992). "Isolation and structure of a brain constituent that binds to the cannabinoid receptor". Science 258 (5090): 1946–9. doi:10.1126/science.1470919. PMID 1470919.
- ^ Mechoulam R, Fride E (1995). "The unpaved road to the endogenous brain cannabinoid ligands, the anandamides". In Pertwee RG. Cannabinoid receptors. Boston: Academic Press. pp. 233–258. ISBN 0-12-551460-3.
- ^ a b Wang, J.; Ueda, N. (2009). "Biology of endocannabinoid synthesis system". Prostaglandins & Other Lipid Mediators 89 (3–4): 112–119. doi:10.1016/j.prostaglandins.2008.12.002. PMID 19126434.
- ^ Gaetani, S.; Dipasquale, P.; Romano, A.; Righetti, L.; Cassano, T.; Piomelli, D.; Cuomo, V. (2009). Chapter 5 the Endocannabinoid System as a Target for Novel Anxiolytic and Antidepressant Drugs. 85. pp. 57. doi:10.1016/S0074-7742(09)85005-8.
- ^ Hwang, J.; Adamson, C.; Butler, D.; Janero, D. R.; Makriyannis, A.; Bahr, B. A. (2009). "Enhancement of endocannabinoid signaling by fatty acid amide hydrolase inhibition: A neuroprotective therapeutic modality". Life Sciences 86 (15-16): 615. doi:10.1016/j.lfs.2009.06.003. PMC 2848893. PMID 19527737. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2848893.
- ^ a b Pacher P, Batkai S, Kunos G (2006). "The Endocannabinoid System as an Emerging Target of Pharmacotherapy". Pharmacol Rev. 58 (3): 389–462. doi:10.1124/pr.58.3.2. PMC 2241751. PMID 16968947. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2241751.
- ^ allet PE, Beninger RJ (1996). "The endogenous cannabinoid receptor agonist anandamide impairs memory in rats". Behavioural Pharmacology 7 (3): 276–284. http://www.behaviouralpharm.com/pt/re/bpharm/abstract.00008877-199605000-00008.htm.
- ^ Piomelli D (January 2004). "THC: moderation during implantation". Nat. Med. 10 (1): 19–20. doi:10.1038/nm0104-19. PMID 14702623.
- ^ El-Talatini MR, Taylor AH, Konje JC (April 2010). "The relationship between plasma levels of the endocannabinoid, anandamide, sex steroids, and gonadotrophins during the menstrual cycle". Fertil. Steril. 93 (6): 1989–96. doi:10.1016/j.fertnstert.2008.12.033. PMID 19200965.
- ^ Mahler SV, Smith KS, Berridge KC (November 2007). "Endocannabinoid hedonic hotspot for sensory pleasure: anandamide in nucleus accumbens shell enhances 'liking' of a sweet reward". Neuropsychopharmacology 32 (11): 2267–78. doi:10.1038/sj.npp.1301376. PMID 17406653.
- ^ De Petrocellis L, Melck D, Palmisano A, Bisogno T, Laezza C, Bifulco M, Di Marzo V (July 1998). "The endogenous cannabinoid anandamide inhibits human breast cancer cell proliferation". Proc. Natl. Acad. Sci. U.S.A. 95 (14): 8375–80. doi:10.1073/pnas.95.14.8375. PMC 20983. PMID 9653194. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=20983.
- ^ di Tomaso E, Beltramo M, Piomelli D. (Aug 1996). "Brain cannabinoids in chocolate". Nature 382 (6593): 677–8. doi:10.1038/382677a0. PMID 8751435.
- ^ Mann, John (2009). Turn On and Tune In - Psychedelics, Narcotics and Euphorics. Cambridge, UK: Royal Society of Chemistry Publishing. p. 95. ISBN 978-1-84755-909-8.
- ^ Natarajan V, Reddy PV, Schmid PC, Schmid HH (August 1982). "N-Acylation of ethanolamine phospholipids in canine myocardium". Biochim. Biophys. Acta 712 (2): 342–55. PMID 7126608.
- ^ Cadas H, di Tomaso E, Piomelli D (February 1997). "Occurrence and biosynthesis of endogenous cannabinoid precursor, N-arachidonoyl phosphatidylethanolamine, in rat brain". J. Neurosci. 17 (4): 1226–42. PMID 9006968.
- ^ Berger A, Crozier G, Bisogno T, Cavaliere P, Innis S, Di Marzo V (May 2001). "Anandamide and diet: Inclusion of dietary arachidonate and docosahexaenoate leads to increased brain levels of the corresponding N-acylethanolamines in piglets". Proc. Natl. Acad. Sci. U.S.A. 98 (11): 6402–6. doi:10.1073/pnas.101119098. PMC 33480. PMID 11353819. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=33480.
- ^ Osei-Hyiaman D, DePetrillo M, Pacher P, Liu J, Radaeva S, Bátkai S, Harvey-White J, Mackie K, Offertáler L, Wang L, Kunos G (May 2005). "Endocannabinoid activation at hepatic CB1 receptors stimulates fatty acid synthesis and contributes to diet-induced obesity". J. Clin. Invest. 115 (5): 1298–305. doi:10.1172/JCI23057. PMC 1087161. PMID 15864349. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1087161.
- ^ Högestätt, E. D.; Jönsson, B. A. G.; Ermund, A.; Andersson, D. A.; Björk, H.; Alexander, J. P.; Cravatt, B. F.; Basbaum, A. I. et al. (2005). "Conversion of Acetaminophen to the Bioactive N-Acylphenolamine AM404 via Fatty Acid Amide Hydrolase-dependent Arachidonic Acid Conjugation in the Nervous System". Journal of Biological Chemistry 280 (36): 31405–31412. doi:10.1074/jbc.M501489200. PMID 15987694.
- ^ Bertolini A, Ferrari A, Ottani A, Guerzoni S, Tacchi R, Leone S (2006). "Paracetamol: new vistas of an old drug". CNS Drug Rev 12 (3–4): 250–75. doi:10.1111/j.1527-3458.2006.00250.x. PMID 17227290.
- ^ Sinning C, Watzer B, Coste O, Nüsing RM, Ott I, Ligresti A, Di Marzo V, Imming P (December 2008). "New analgesics synthetically derived from the paracetamol metabolite N-(4-hydroxyphenyl)-(5Z,8Z,11Z,14Z)-icosatetra-5,8,11,14-enamide". J. Med. Chem. 51 (24): 7800–5. doi:10.1021/jm800807k. PMID 19053765.
External links
- Could anandamide be the missing link to "runner's high"? Accessed 2008-10-18
Cannabinoids Plant cannabinoids Cannabinoid metabolites 8,11-DiOH-THC · 11-COOH-THC · 11-OH-THC
Endogenous cannabinoids Arachidonoyl ethanolamide (Anandamide or AEA) · 2-Arachidonoylglycerol (2-AG) · 2-Arachidonyl glyceryl ether (noladin ether) · Virodhamine · Palmitoylethanolamide (PEA) · N-Arachidonoyl dopamine (NADA) · Oleamide · RVD-Hpα
Synthetic cannabinoid
receptor agonistsClassical cannabinoids
(Dibenzopyrans)A-40174 · A-41988 · A-42574 · Ajulemic acid · AM-087 · AM-411 · AM-855 · AM-905 · AM-906 · AM-919 · AM-926 · AM-938 · AM-4030 · AMG-1 · AMG-3 · AMG-36 · AMG-41 · Dexanabinol (HU-211) · DMHP · Dronabinol · HHC · HU-210 · JWH-051 · JWH-133 · JWH-139 · JWH-161 · JWH-229 · JWH-359 · KM-233 · L-759,633 · L-759,656 · Levonantradol (CP 50,5561) · Nabazenil · Nabidrox (Canbisol) · Nabilone · Nabitan · Naboctate · O-581 · O-774 · O-806 · O-823 · O-1057 · O-1125 · O-1238 · O-2365 · O-2372 · O-2373 · O-2383 · O-2426 · O-2484 · O-2545 · O-2694 · O-2715 · O-2716 · O-3223 · O-3226 · Parahexyl · Perrottetinene · Pirnabine · THC-O-acetate · THC-O-phosphate
Nonclassical cannabinoidsBenzoylindoles1-Butyl-3-(2-methoxybenzoyl)indole · 1-Butyl-3-(4-methoxybenzoyl)indole · 1-Pentyl-3-(2-methoxybenzoyl)indole · AM-630 · AM-679 · AM-694 · AM-1241 · AM-2233 · GW-405,833 (L-768,242) · Pravadoline · RCS-4 · WIN 54,461
NaphthoylindolesNaphthylmethylindolesJWH-175 · JWH-184 · JWH-185 · JWH-192 · JWH-194 · JWH-195 · JWH-196 · JWH-197 · JWH-199
PhenylacetylindolesCannabipiperidiethanone · JWH-167 · JWH-203 · JWH-249 · JWH-250 · JWH-251 · JWH-302 · RCS-8
NaphthoylpyrrolesEicosanoidsAM-883 · Arachidonyl-2'-chloroethylamide (ACEA) · Arachidonylcyclopropylamide (ACPA) · Methanandamide (AM-356) · O-585 · O-689 · O-1812 · O-1860 · O-1861
Others(1-Pentylindol-3-yl)-(2,2,3,3-tetramethylcyclopropyl)methanone · N-(S)-Fenchyl-1-(2-morpholinoethyl)-7-methoxyindole-3-carboxamide · A-796,260 · A-834,735 · A-836,339 · Abnormal cannabidiol · AB-001 · AM-1248 · AZ-11713908 · BAY 38-7271 · BAY 59-3074 · CB-13 · CB-86 · GW-842,166X · JWH-171 · JWH-176 · JTE 7-31 · Leelamine · MDA-19 · O-1918 · O-2220 · Org 28312 · Org 28611 · SER-601 · VSN-16 · WIN 56,098
Allosteric modulators of
cannabinoid receptorsOrg 27569 · Org 27759 · Org 29647
Endocannabinoid
activity enhancersAM-404 · CAY-10401 · CAY-10429 · JZL184 · JZL195 · N-arachidonoyl-serotonin · O-1624 · PF-04457845 · PF-622 · PF-750 · PF-3845 · PHOP · URB-447 · URB-597 · URB-602 · URB-754 · Genistein · Arvanil · Olvanil · Kaempferol · Biochanin A
Cannabinoid receptor
antagonists and
inverse agonistsAM-251 · AM-281 · AM-630 · BML-190 · CAY-10508 · CB-25 · CB-52 · CB-86 · Drinabant · Hemopressin · Ibipinabant (SLV319) · JTE-907 · LY-320,135 · Taranabant (MK-0364) · MK-9470 · NESS-0327 · O-1184 · O-1248 · O-2050 · O-2654 · Otenabant · Rimonabant (SR141716) · SR144528 · Surinabant (SR147778) · TM-38837 · VCHSR
Neurotransmitters Amino acids Alanine · Aspartate · Cycloserine · DMG · GABA · Glutamate · Glycine · Hypotaurine · Kynurenic acid (Transtorine) · NAAG (Spaglumic acid) · NMG (Sarcosine) · Serine · Taurine · TMG (Betaine)
Endocannabinoids 2-AG · 2-AGE (Noladin ether) · AEA (Anandamide) · NADA · OAE (Virodhamine) · Oleamide · PEA (Palmitoylethanolamide) · RVD-Hpα · Hp (Hemopressin)
Gasotransmitters Monoamines Purines Trace amines 3-ITA · 5-MeO-DMT · Bufotenin · DMT · NMT · Octopamine · Phenethylamine · Synephrine · Thyronamine · Tryptamine · Tyramine
Others 1,4-BD · Acetylcholine · GBL · GHB · Histamine
Categories:- Cannabinoids
- Neurotransmitters
- Amides
- Biomolecules
- Alcohols
- O=C(NCCO)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC
Wikimedia Foundation. 2010.

