- Hydrogen economy
-
The hydrogen economy is a proposed system of delivering energy using hydrogen. The term hydrogen economy was coined by John Bockris during a talk he gave in 1970 at General Motors (GM) Technical Center.[1]
Hydrogen advocates promote hydrogen as potential fuel for motive power (including cars and boats), the energy needs of buildings and portable electronics. Free hydrogen does not occur naturally in quantity, and thus it must be generated from some other energy source by steam reformation of natural gas or another method. Hydrogen is therefore an energy carrier (like electricity), not a primary energy source (like coal). The utility of a hydrogen economy depends on issues of energy sourcing, including fossil fuel use, climate change, and sustainable energy generation.
Rationale
A hydrogen economy is proposed to solve some of the negative effects of using hydrocarbon fuels where the carbon is released to the atmosphere. Modern interest in the hydrogen economy can generally be traced to a 1970 technical report by Lawrence W. Jones of the University of Michigan.[2]
In the current hydrocarbon economy, transportation is fueled primarily by petroleum. Burning of hydrocarbon fuels emits carbon dioxide and other pollutants. The supply of economically usable hydrocarbon resources in the world is limited, and the demand for hydrocarbon fuels is increasing, particularly in China, India and other developing countries.
Proponents of a world-scale hydrogen economy argue that hydrogen can be an environmentally cleaner source of energy to end-users, particularly in transportation applications, without release of pollutants (such as particulate matter) or carbon dioxide at the point of end use. A 2004 analysis asserted that "most of the hydrogen supply chain pathways would release significantly less carbon dioxide into the atmosphere than would gasoline used in hybrid electric vehicles" and that significant reductions in carbon dioxide emissions would be possible if carbon capture or carbon sequestration methods were utilized at the site of energy or hydrogen production.[3]
Hydrogen has a high energy density by weight. An Otto cycle internal combustion engine running on hydrogen is said to have a maximum efficiency of about 38%, 8% higher than gasoline internal combustion engine.[4]
The combination of the fuel cell and electric motor is 2-3 times more efficient than an internal combustion engine.[5] However, the high capital costs of fuel cells, about $5,500/kW in 2002,[6] are one of the major obstacles of its development, meaning that the fuel cell is only technically, but not economically, more efficient than an internal combustion engine.[7]
Other technical obstacles include hydrogen storage issues and the purity requirement of hydrogen used in fuel cells – with current technology, an operating fuel cell requires the purity of hydrogen to be as high as 99.999%. On the other hand, hydrogen engine conversion technology is more economical than fuel cells.[8]
Perspective: current hydrogen market (current hydrogen economy)
Hydrogen production is a large and growing industry. Globally, some 50 million metric tons of hydrogen,[9] equal to about 170 million tons of oil equivalent, were produced in 2004. The growth rate is around 10% per year. Within the United States, 2004 production was about 11 million metric tons (MMT), an average power flow of 48 gigawatts. (For comparison, the average electric production in 2003 was some 442 gigawatts.) As of 2005, the economic value of all hydrogen produced worldwide is about $135 billion per year.[10]
There are two primary uses for hydrogen today. About half is used to produce ammonia (NH3) via the Haber process, which is then used directly or indirectly as fertilizer. Because both the world population and the intensive agriculture used to support it are growing, ammonia demand is growing. The other half of current hydrogen production is used to convert heavy petroleum sources into lighter fractions suitable for use as fuels. This latter process is known as hydrocracking. Hydrocracking represents an even larger growth area, since rising oil prices encourage oil companies to extract poorer source material, such as tar sands and oil shale. The scale economies inherent in large scale oil refining and fertilizer manufacture make possible on-site production and "captive" use. Smaller quantities of "merchant" hydrogen are manufactured and delivered to end users as well.
If energy for hydrogen production were available (from wind, solar, fission or fusion nuclear power etc.), use of the substance for hydrocarbon synfuel production could expand captive use of hydrogen by a factor of 5 to 10. Present U.S. use of hydrogen for hydrocracking is roughly 4 million metric tons per year (4 MMT/yr). It is estimated that 37.7 MMT/yr of hydrogen would be sufficient to convert enough domestic coal to liquid fuels to end U.S. dependence on foreign oil importation,[11] and less than half this figure to end dependence on Middle East oil. Coal liquefaction would present significantly worse emissions of carbon dioxide than does the current system of burning fossil petroleum, but it would eliminate the political and economic vulnerabilities inherent in oil importation.
Currently, global hydrogen production is 48% from natural gas, 30% from oil, and 18% from coal; water electrolysis accounts for only 4%.[12] The distribution of production reflects the effects of thermodynamic constraints on economic choices: of the four methods for obtaining hydrogen, partial combustion of natural gas in a NGCC (natural gas combined cycle) power plant offers the most efficient chemical pathway and the greatest off-take of usable heat energy.
The large market and sharply rising prices in fossil fuels have also stimulated great interest in alternate, cheaper means of hydrogen production.[13] As of 2002, most hydrogen is produced on site and the cost is approximately $0.32/lb and, if not produced on site, the cost of liquid hydrogen is about $1.00/lb to $1.40/lb.[14]
Production, storage, infrastructure
Main article: Hydrogen technologiesFurther information: Timeline of hydrogen technologiesToday hydrogen is mainly produced (90%) from fossil sources.[15] Linking its centralized production to a fleet of light-duty fuel cell vehicles would require the siting and construction of a distribution infrastructure with large investment of capital.[citation needed] Further, the technological challenge of providing safe, energy-dense storage of hydrogen on-board the vehicle must be overcome to provide sufficient range between fillups.[citation needed]
Methods of production
Main article: Hydrogen productionMolecular hydrogen is not available on Earth in convenient natural reservoirs. Most hydrogen on Earth is bonded to oxygen in water. Manufacturing elemental hydrogen does require the consumption of a hydrogen carrier such as a fossil fuel or water. The former consumes the fossil resource and produces carbon dioxide, but often requires no further energy input beyond the fossil fuel. Decomposing water requires electrical or heat input, generated from some primary energy source (fossil fuel, nuclear power or a renewable energy). Note that the energy provided by the energy source provides all of the energy that is available in the hydrogen fuel.
Current production methods
Hydrogen is industrially produced from steam reforming, which uses fossil fuels such as natural gas, oil, or coal.[16] The energy content of the produced hydrogen is less than the energy content of the original fuel, some of it being lost as excessive heat during production. Steam reforming leads to carbon dioxide emissions, in the same way as a car engine would do.
A small part (4% in 2006) is produced by electrolysis using electricity and water, consuming approximately 50 kilowatt-hours of electricity per kilogram of hydrogen produced.
Kværner-process
The Kværner-process or Kvaerner carbon black & hydrogen process (CB&H)[15] is a method, developed in the 1980s by a Norwegian company of the same name, for the production of hydrogen from hydrocarbons (CnHm), such as methane, natural gas and biogas. Of the available energy of the feed, approximately 48% is contained in the Hydrogen, 40% is contained in activated carbon and 10% in superheated steam.[17]
Biological production
Main article: Biological hydrogen production (Algae)Fermentative hydrogen production is the fermentative conversion of organic substrate to biohydrogen manifested by a diverse group bacteria using multi enzyme systems involving three steps similar to anaerobic conversion. Dark fermentation reactions do not require light energy, so they are capable of constantly producing hydrogen from organic compounds throughout the day and night. Photofermentation differs from dark fermentation because it only proceeds in the presence of light. For example photo-fermentation with Rhodobacter sphaeroides SH2C can be employed to convert small molecular fatty acids into hydrogen.[18] Electrohydrogenesis is used in microbial fuel cells where hydrogen is produced from organic matter (e.g. from sewage, or solid matter [19]) while 0.2 - 0.8 V is applied.
Biological hydrogen can be produced in an algae bioreactor. In the late 1990s it was discovered that if the algae is deprived of sulfur it will switch from the production of oxygen, i.e. normal photosynthesis, to the production of hydrogen.[citation needed]
Biological hydrogen can be produced in bioreactors that use feedstocks other than algae, the most common feedstock being waste streams. The process involves bacteria feeding on hydrocarbons and excreting hydrogen and CO2. The CO2 can be sequestered successfully by several methods, leaving hydrogen gas. A prototype hydrogen bioreactor using waste as a feedstock is in operation at Welch's grape juice factory in North East, Pennsylvania.
Biocatalysed electrolysis
Besides regular electrolysis, electrolysis using microbes is another possibility. With biocatalysed electrolysis, hydrogen is generated after running through the microbial fuel cell and a variety of aquatic plants can be used. These include reed sweetgrass, cordgrass, rice, tomatoes, lupines, algae [20]
Electrolysis of water
 Electrolysis of water ship Hydrogen Challenger
Electrolysis of water ship Hydrogen Challenger
Hydrogen can be made via high pressure electrolysis or low pressure electrolysis of water. Current best processes have an efficiency of 50% to 80%,[21][22][23] so that 1 kg of hydrogen (which has a specific energy of 143 MJ/kg, about 40 kWh/kg) requires 50 to 79 kWh of electricity. At 8 cents/kWh, that's $4.00/kg, which is with traditional methods 3 to 10 times the price of hydrogen from steam reformation of natural gas.[14] The price difference is due to the efficiency of direct conversion of fossil fuels to produce hydrogen, rather than burning fuel to produce electricity. Hydrogen from natural gas, used to replace e.g. gasoline, emits more CO2 than the gasoline it would replace, and so is no help in reducing greenhouse gases.[24]
High-pressure electrolysis
High pressure electrolysis is the electrolysis of water by decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) by means of an electric current being passed through the water. The difference with a standard electrolyzer is the compressed hydrogen output around 120-200 Bar (1740-2900 psi).[25] By pressurising the hydrogen in the electrolyser the need for an external hydrogen compressor is eliminated, the average energy consumption for internal compression is around 3%.[26]
High-temperature electrolysis
Main article: High-temperature electrolysisHydrogen can be generated from energy supplied in the form of heat and electricity through high-temperature electrolysis (HTE). Because some of the energy in HTE is supplied in the form of heat, less of the energy must be converted twice (from heat to electricity, and then to chemical form), and so potentially far less energy is required per kilogram of hydrogen produced.
While nuclear-generated electricity could be used for electrolysis, nuclear heat can be directly applied to split hydrogen from water. High temperature (950–1000 °C) gas cooled nuclear reactors have the potential to split hydrogen from water by thermochemical means using nuclear heat. Research into high-temperature nuclear reactors may eventually lead to a hydrogen supply that is cost-competitive with natural gas steam reforming. General Atomics predicts that hydrogen produced in a High Temperature Gas Cooled Reactor (HTGR) would cost $1.53/kg. In 2003, steam reforming of natural gas yielded hydrogen at $1.40/kg. At 2005 natural gas prices, hydrogen costs $2.70/kg.
High-temperature electrolysis has been demonstrated in a laboratory, at 108 megajoules (thermal) per kilogram of hydrogen produced,[27] but not at a commercial scale. In addition, this is lower-quality "commercial" grade Hydrogen, unsuitable for use in fuel cells.[28]
Photoelectrochemical water splitting
Using electricity produced by photovoltaic systems offers the cleanest way to produce hydrogen. Water is broken into hydrogen and oxygen by electrolysis—a photoelectrochemical cell (PEC) process which is also named artificial photosynthesis. Research aimed toward developing higher-efficiency multi-junction cell technology is underway by the photovoltaic industry. If this process is assisted by photocatalysts suspended directly in water instead of using photovoltaic and an electrolytic system, the reaction is in just one step, which can improve efficiency.[29][30]
Concentrating solar thermal
Very high temperatures are required to dissociate water into hydrogen and oxygen. A catalyst is required to make the process operate at feasible temperatures. Heating the water can be achieved through the use of concentrating solar power. Hydrosol-2 is a 100-kilowatt pilot plant at the Plataforma Solar de Almería in Spain which uses sunlight to obtain the required 800 to 1,200 °C to heat water. Hydrosol II has been in operation since 2008. The design of this 100-kilowatt pilot plant is based on a modular concept. As a result, it may be possible that this technology could be readily scaled up to the megawatt range by multiplying the available reactor units and by connecting the plant to heliostat fields (fields of sun-tracking mirrors) of a suitable size.[31]
Photoelectrocatalytic production
A method studied by Thomas Nann and his team at the University of East Anglia consists of a gold electrode covered in layers of indium phosphide (InP) nanoparticles. They introduced an iron-sulfur complex into the layered arrangement, which when submerged in water and irradiated with light under a small electric current, produced hydrogen with an efficiency of 60%.[32]
Thermochemical production
There are more than 352[33] thermochemical cycles which can be used for water splitting,[34] around a dozen of these cycles such as the iron oxide cycle, cerium(IV) oxide-cerium(III) oxide cycle, zinc zinc-oxide cycle, sulfur-iodine cycle, copper-chlorine cycle and hybrid sulfur cycle are under research and in testing phase to produce hydrogen and oxygen from water and heat without using electricity.[35] These processes can be more efficient than high-temperature electrolysis, typical in the range from 35 % - 49 % LHV efficiency. Thermochemical production of hydrogen using chemical energy from coal or natural gas is generally not considered, because the direct chemical path is more efficient.
None of the thermochemical hydrogen production processes have been demonstrated at production levels, although several have been demonstrated in laboratories.
Storage
Main article: Hydrogen storageAlthough molecular hydrogen has very high energy density on a mass basis, partly because of its low molecular weight, as a gas at ambient conditions it has very low energy density by volume. If it is to be used as fuel stored on board the vehicle, pure hydrogen gas must be pressurized or liquefied to provide sufficient driving range. Increasing gas pressure improves the energy density by volume, making for smaller, but not lighter container tanks (see pressure vessel). Achieving higher pressures necessitates greater use of external energy to power the compression. Alternatively, higher volumetric energy density liquid hydrogen or slush hydrogen may be used. However, liquid hydrogen is cryogenic and boils at 20.268 K (–252.882 °C or –423.188 °F). Cryogenic storage cuts weight but requires large liquification energies. The liquefaction process, involving pressurizing and cooling steps, is energy intensive. The liquefied hydrogen has lower energy density by volume than gasoline by approximately a factor of four, because of the low density of liquid hydrogen — there is actually more hydrogen in a liter of gasoline (116 grams) than there is in a liter of pure liquid hydrogen (71 grams). Liquid hydrogen storage tanks must also be well insulated to minimize boil off. Ice may form around the tank and help corrode it further if the liquid hydrogen tank insulation fails.
The mass of the tanks needed for compressed hydrogen reduces the fuel economy of the vehicle. Because it is a small molecule, hydrogen tends to diffuse through any liner material intended to contain it, leading to the embrittlement, or weakening, of its container.
Distinct from storing molecular hydrogen, hydrogen can be stored as a chemical hydride or in some other hydrogen-containing compound. Hydrogen gas is reacted with some other materials to produce the hydrogen storage material, which can be transported relatively easily. At the point of use the hydrogen storage material can be made to decompose, yielding hydrogen gas. As well as the mass and volume density problems associated with molecular hydrogen storage, current barriers to practical storage schemes stem from the high pressure and temperature conditions needed for hydride formation and hydrogen release. For many potential systems hydriding and dehydriding kinetics and heat management are also issues that need to be overcome. A French company McPhy Energy [3] is developing the first industrial product, based on Magnesium Hydrate, already sold to some major clients such as Iwatani and ENEL.
A third approach is to absorb molecular hydrogen into a solid storage material. Unlike in the hydrides mentioned above, the hydrogen does not dissociate/recombine upon charging/discharging the storage system, and hence does not suffer from the kinetic limitations of many hydride storage systems. Hydrogen densities similar to liquefied hydrogen can be achieved with appropriate absorption media. Some suggested absorbers include MOFs, nanostructured carbons (including CNTs) and hydrogen clathrate hydrate.
The most common method of on board hydrogen storage in today's demonstration vehicles is as a compressed gas at pressures of roughly 700 bar (70 MPa).
Underground hydrogen storage is the practice of hydrogen storage in underground caverns, salt domes and depleted oil and gas fields. Large quantities of gaseous hydrogen are stored in underground caverns by ICI for many years without any difficulties.[36] The storage of large quantities of hydrogen underground can function as grid energy storage which is essential for the hydrogen economy.
- Alternative storage proposal
It has been proposed in a hypothetical renewable energy dominated energy system to use the excess electricity generated by wind, solar photovoltaic, hydro, marine currents and others to make methane (natural gas) by electrolysis of water.[37][38] The methane could then be injected into the existing gas network to generate electricity and heat on demand to overcome low points of renewable energy production. The process described would be to create hydrogen (which could partly be used directly in fuel cells) and the addition of carbon dioxide CO2 (Sabatier process) to create methane as follows: CO2 + 4H2 → CH4 + 2H2O
Infrastructure
Main article: Hydrogen infrastructure Praxair Hydrogen Plant
Praxair Hydrogen Plant
The hydrogen infrastructure would consist mainly of industrial hydrogen pipeline transport and hydrogen-equipped filling stations like those found on a hydrogen highway. Hydrogen stations which were not situated near a hydrogen pipeline would get supply via hydrogen tanks, compressed hydrogen tube trailers, liquid hydrogen trailers, liquid hydrogen tank trucks or dedicated onsite production.
Because of hydrogen embrittlement of steel, and corrosion[39][40] natural gas pipes require internal coatings or replacement in order to convey hydrogen. Techniques are well-known; over 700 miles of hydrogen pipeline currently exist in the United States. Although expensive, pipelines are the cheapest way to move hydrogen. Hydrogen gas piping is routine in large oil-refineries, because hydrogen is used to hydrocrack fuels from crude oil.
Hydrogen piping can in theory be avoided in distributed systems of hydrogen production, where hydrogen is routinely made on site using medium or small-sized generators which would produce enough hydrogen for personal use or perhaps a neighborhood. In the end, a combination of options for hydrogen gas distribution may succeed.
While millions of tons of elemental hydrogen are distributed around the world each year in various ways, bringing hydrogen to individual consumers would require an evolution of the fuel infrastructure. For example, according to GM, 70% of the U.S. population lives near a hydrogen-generating facility but has little public access to that hydrogen. The same study however, shows that building the infrastructure in a systematic way is much more doable and affordable than most people think. For example, one article has noted that hydrogen stations could be put within every 10 miles in metro Los Angeles, and on the highways between LA and neighboring cities like Palm Springs, Las Vegas, San Diego and Santa Barbara, for the cost of a Starbuck's latte for every one of the 15 million residents living in these areas.[41]
A key tradeoff: centralized vs. distributed production
In a future full hydrogen economy, primary energy sources and feedstock would be used to produce hydrogen gas as stored energy for use in various sectors of the economy. Producing hydrogen from primary energy sources other than coal, oil, and natural gas, would result in lower production of the greenhouse gases characteristic of the combustion of these fossil energy resources.
One key feature of a hydrogen economy would be that in mobile applications (primarily vehicular transport) energy generation and use could be decoupled. The primary energy source would need no longer travel with the vehicle, as it currently does with hydrocarbon fuels. Instead of tailpipes creating dispersed emissions, the energy (and pollution) could be generated from point sources such as large-scale, centralized facilities with improved efficiency. This would allow the possibility of technologies such as carbon sequestration, which are otherwise impossible for mobile applications. Alternatively, distributed energy generation schemes (such as small scale renewable energy sources) could be used, possibly associated with hydrogen stations.
Aside from the energy generation, hydrogen production could be centralized, distributed or a mixture of both. While generating hydrogen at centralized primary energy plants promises higher hydrogen production efficiency, difficulties in high-volume, long range hydrogen transportation (due to factors such as hydrogen damage and the ease of hydrogen diffusion through solid materials) makes electrical energy distribution attractive within a hydrogen economy. In such a scenario, small regional plants or even local filling stations could generate hydrogen using energy provided through the electrical distribution grid. While hydrogen generation efficiency is likely to be lower than for centralized hydrogen generation, losses in hydrogen transport could make such a scheme more efficient in terms of the primary energy used per kilogram of hydrogen delivered to the end user.
The proper balance between hydrogen distribution and long-distance electrical distribution is one of the primary questions that arises about the hydrogen economy.
Again the dilemmas of production sources and transportation of hydrogen can now be overcome using on site (home, business, or fuel station) generation of hydrogen from off grid renewable sources.[4].
Distributed electrolysis
Distributed electrolysis would bypass the problems of distributing hydrogen by distributing electricity instead. It would use existing electrical networks to transport electricity to small, on-site electrolysers located at filling stations. However, accounting for the energy used to produce the electricity and transmission losses would reduce the overall efficiency.
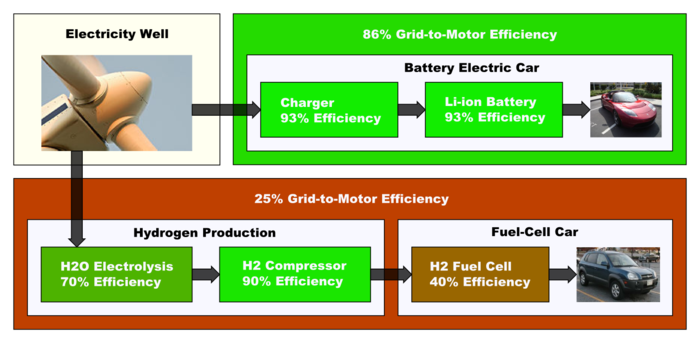
Natural gas combined cycle power plants, which account for almost all construction of new electricity generation plants in the United States, generate electricity at efficiencies of 60 percent or greater. Increased demand for electricity, whether due to hydrogen cars or other demand, would have the marginal impact of adding new combined cycle power plants. On this basis, distributed production of hydrogen would be roughly 40% efficient. However, if the marginal impact is referred to today's power grid, with an efficiency of roughly 40% owing to its mix of fuels and conversion methods, the efficiency of distributed hydrogen production would be roughly 25%.[42]
The distributed production of hydrogen in this fashion would be expected to generate air emissions of pollutants and carbon dioxide at various points in the supply chain, e.g., electrolysis, transportation and storage. Such externalities as pollution must be weighed against the potential advantages of a hydrogen economy.
Fuel cells as alternative to internal combustion
Main article: Fuel cellMain article: Hydrogen vehicleOne of the main offerings of a hydrogen economy is that the fuel can replace the fossil fuel burned in internal combustion engines and turbines as the primary way to convert chemical energy into kinetic or electrical energy; hereby eliminating greenhouse gas emissions and pollution from that engine.
Although hydrogen can be used in conventional internal combustion engines, fuel cells, being electrochemical, have a theoretical efficiency advantage over heat engines. Fuel cells are more expensive to produce than common internal combustion engines, but are becoming cheaper as new technologies and production systems develop.
Some types of fuel cells work with hydrocarbon fuels,[43] while all can be operated on pure hydrogen. In the event that fuel cells become price-competitive with internal combustion engines and turbines, large gas-fired power plants could adopt this technology.
Hydrogen gas must be distinguished as "technical-grade" (five nines pure), which is suitable for applications such as fuel cells, and "commercial-grade", which has carbon- and sulfur-containing impurities, but which can be produced by the much cheaper steam-reformation process. Fuel cells require high purity hydrogen because the impurities would quickly degrade the life of the fuel cell stack.
Much of the interest in the hydrogen economy concept is focused on the use of fuel cells to power electric cars. Current Hydrogen fuel cells suffer from a low power-to-weight ratio.[44] Fuel cells are much more efficient than internal combustion engines, and produce no harmful emissions. If a practical method of hydrogen storage is introduced, and fuel cells become cheaper, they can be economically viable to power hybrid fuel cell/battery vehicles, or purely fuel cell-driven ones. The economic viability of fuel cell powered vehicles will improve as the hydrocarbon fuels used in internal combustion engines become more expensive, because of the depletion of easily accessible reserves or economic accounting of environmental impact through such measures as carbon taxes.
Other fuel cell technologies based on the exchange of metal ions (i.e. zinc-air fuel cells) are typically more efficient at energy conversion than hydrogen fuel cells, but the widespread use of any electrical energy → chemical energy → electrical energy systems would necessitate the production of electricity.
Efficiency as an automotive fuel
Hydrogen has been called one of the least efficient and most expensive possible replacements for gasoline (petrol) in terms of reducing greenhouse gases; other technologies may be less expensive and more quickly implemented.[45][46] A comprehensive study of hydrogen in transportation applications has found that "there are major hurdles on the path to achieving the vision of the hydrogen economy; the path will not be simple or straightforward".[3] The Ford Motor Company has dropped its plans to develop hydrogen cars, stating that "The next major step in Ford’s plan is to increase over time the volume of electrified vehicles".[47]
An accounting of the energy utilized during a thermodynamic process, known as an energy balance, can be applied to automotive fuels. With today's technology, the manufacture of hydrogen via steam reforming can be accomplished with a thermal efficiency of 75 to 80 percent. Additional energy will be required to liquefy or compress the hydrogen, and to transport it to the filling station via truck or pipeline. The energy that must be utilized per kilogram to produce, transport and deliver hydrogen (i.e., its well-to-tank energy use) is approximately 50 megajoules using technology available in 2004. Subtracting this energy from the enthalpy of one kilogram of hydrogen, which is 141 megajoules, and dividing by the enthalpy, yields a thermal energy efficiency of roughly 60%.[48] Gasoline, by comparison, requires less energy input, per gallon, at the refinery, and comparatively little energy is required to transport it and store it owing to its high energy density per gallon at ambient temperatures. Well-to-tank, the supply chain for gasoline is roughly 80% efficient (Wang, 2002). The most efficient distribution however is electrical, which is typically 95% efficient. Electric vehicles are typically 3 to 4 times as efficient as hydrogen powered vehicles.[49]
A study of the well-to-wheels efficiency of hydrogen vehicles compared to other vehicles in the Norwegian energy system indicates that hydrogen fuel-cell vehicles tend to be about a third as efficient as EVs when electrolysis is used, with hydrogen Internal Combustion Engines (ICE) being barely a sixth as efficient. Even in the case where hydrogen fuel cells get their hydrogen from natural gas reformation rather than electrolysis, and EVs get their power from a natural gas power plant, the EVs still come out ahead 35% to 25% (and only 13% for a H2 ICE). This compares to 14% for a gasoline ICE, 27% for a gasoline ICE hybrid, and 17% for a diesel ICE, also on a well-to-wheels basis.[50]
Hydrogen safety
Main article: Hydrogen safetyHydrogen has one of the widest explosive/ignition mix range with air of all the gases with few exceptions such as acetylene, silane, and ethylene oxide. That means that whatever the mix proportion between air and hydrogen, a hydrogen leak will most likely lead to an explosion, not a mere flame, when a flame or spark ignites the mixture. This makes the use of hydrogen particularly dangerous in enclosed areas such as tunnels or underground parking.[51] Pure hydrogen-oxygen flames burn in the ultraviolet color range and are nearly invisible to the naked eye, so a flame detector is needed to detect if a hydrogen leak is burning. Hydrogen is odorless and leaks cannot be detected by smell.
Hydrogen codes and standards are codes and standards for hydrogen fuel cell vehicles, stationary fuel cell applications and portable fuel cell applications. There are codes and standards for the safe handling and storage of hydrogen, for example the Standard for the installation of stationary fuel cell power systems from the National Fire Protection Association.
Codes and standards have repeatedly been identified as a major institutional barrier to deploying hydrogen technologies and developing a hydrogen economy. To enable the commercialization of hydrogen in consumer products, new model building codes and equipment and other technical standards are developed and recognized by federal, state, and local governments.[52]
One of the measures on the roadmap is to implement higher safety standards like early leak detection with hydrogen sensors.[53] The Canadian Hydrogen Safety Program concluded that hydrogen fueling is as safe as, or safer than, CNG fueling.[54] The European Commission has funded the first higher educational program in the world in hydrogen safety engineering at the University of Ulster. It is expected that the general public will be able to use hydrogen technologies in everyday life with at least the same level of safety and comfort as with today's fossil fuels.
Environmental concerns
There are many concerns regarding the environmental effects of the manufacture of hydrogen. Hydrogen is made either by electrolysis of water, or by fossil fuel reforming. Reforming a fossil fuel leads to a higher emissions of carbon dioxide compared with direct use of the fossil fuel in an internal combustion engine. Similarly, if hydrogen is produced by electrolysis from fossil-fuel powered generators, increased carbon dioxide is emitted in comparison with direct use of the fossil fuel.
Using renewable energy source to generate hydrogen by electrolysis would require greater energy input than direct use of the renewable energy to operate electric vehicles, because of the extra conversion stages and losses in distribution.
Like any internal combustion engine, an ICE running on hydrogen may produce nitrous oxides and other pollutants. Air input into the combustion cylinder is approximately 78% nitrogen, and the N2 molecule has a binding energy of approximately 226 kilocalories per mole. The hydrogen reaction has sufficient energy to break this bond and produce unwanted components such as nitric acid (HNO3), and hydrogen cyanide gas (HCN), both toxic byproducts.[55] Nitrogen compound emissions from internal combustion engines are a root cause of smog.[56] Hydrogen as transportation fuel, however, is mainly used for fuel cells that do not produce greenhouse gas emission, but water.
There have also been some concerns over possible problems related to hydrogen gas leakage.[57] Molecular hydrogen leaks slowly from most containment vessels. It has been hypothesized that if significant amounts of hydrogen gas (H2) escape, hydrogen gas may, because of ultraviolet radiation, form free radicals (H) in the stratosphere. These free radicals would then be able to act as catalysts for ozone depletion. A large enough increase in stratospheric hydrogen from leaked H2 could exacerbate the depletion process. However, the effect of these leakage problems may not be significant. The amount of hydrogen that leaks today is much lower (by a factor of 10–100) than the estimated 10–20% figure conjectured by some researchers; for example, in Germany, the leakage rate is only 0.1% (less than the natural gas leak rate of 0.7%). At most, such leakage would likely be no more than 1–2% even with widespread hydrogen use, using present technology.[57]
Costs
When evaluating costs, fossil fuels are generally used as the reference. The energy content of these fuels is not a product of human effort and so has no cost assigned to it. Only the extraction, refining, transportation and production costs are considered. On the other hand, the energy content of a unit of hydrogen fuel must be manufactured, and so has a significant cost, on top of all the costs of refining, transportation, and distribution. Systems which use renewably generated electricity more directly, for example in trolleybuses, or in battery electric vehicles may have a significant economic advantage because there are fewer conversion processes required between primary energy source and point of use.
The barrier to lowering the price of high purity hydrogen is a cost of more than 35 kWh of electricity used to generate each kilogram of hydrogen gas.
Demonstrated advances in electrolyzer and fuel cell technology by ITM Power [58] are claimed to have made significant in-roads into addressing the cost of electrolysing water to make hydrogen. Cost reduction would make hydrogen from off-grid renewable sources economic for refueling vehicles.
Hydrogen pipelines are more expensive[59] than even long-distance electric lines. Hydrogen is about three times bulkier in volume than natural gas for the same enthalpy. Hydrogen accelerates the cracking of steel (hydrogen embrittlement), which increases maintenance costs, leakage rates, and material costs. The difference in cost is likely to expand with newer technology: wires suspended in air can use higher voltage with only marginally increased material costs, but higher pressure pipes require proportionally more material.
Setting up a hydrogen economy would require huge investments in the infrastructure to store and distribute hydrogen to vehicles. In contrast, battery electric vehicles, which are already publicly available, would not necessitate immediate expansion of the existing infrastructure for electricity transmission and distribution. Power plant capacity that now goes unused at night could be used for recharging electric vehicles. A study conducted by the Pacific Northwest National Laboratory for the US Department of Energy in December 2006 found that the idle off-peak grid capacity in the US would be sufficient to power 84% of all vehicles in the US if they all were immediately replaced with electric vehicles.[60]
Different production methods each have differing associated investment and marginal costs. The energy and feedstock could originate from a multitude of sources i.e. natural gas, nuclear, solar, wind, biomass, coal, other fossil fuels, and geothermal.
- Natural Gas at Small Scale
- Uses steam reformation. Requires 15.9 million cubic feet (450,000 m3) of gas, which, if produced by small 500 kg/day reformers at the point of dispensing (i.e., the filling station), would equate to 777,000 reformers costing $1 trillion dollars and producing 150 million tons of hydrogen gas annually. Obviates the need for distribution infrastructure dedicated to hydrogen. $3.00 per GGE (Gallons of Gasoline Equivalent)
- Nuclear
- Provides energy for electrolysis of water. Would require 240,000 tons of unenriched uranium — that's 2,000 600-megawatt power plants, which would cost $840 billion, or about $2.50 per GGE.[61]
- Solar
- Provides energy for electrolysis of water. Would require 2,500 kWh of sun per square meter, 113 million 40-kilowatt systems, which would cost $22 trillion, or about $9.50 per GGE.
- Wind
- Provides energy for electrolysis of water. At 7 meters per second average wind speed, it would require 1 million 2-MW wind turbines, which would cost $3 trillion dollars, or about $3.00 per GGE.
- Biomass
- Gasification plants would produce gas with steam reformation. 1.5 billion tons of dry biomass, 3,300 plants which would require 113.4 million acres (460,000 km²) of farm to produce the biomass. $565 billion dollars in cost, or about $1.90 per GGE
- Coal
- FutureGen plants use coal gasification then steam reformation. Requires 1 billion tons of coal or about 1,000 275-megawatt plants with a cost of about $500 billion, or about $1 per GGE.
- DOE Cost targets[62]
Examples and pilot programs
Several domestic U.S. automobile manufactures have committed to develop vehicles using hydrogen. The distribution of hydrogen for the purpose of transportation is currently being tested around the world, particularly in Portugal, Iceland, Norway, Denmark, Germany, California, Japan and Canada, but the cost is very high.
Some hospitals have installed combined electrolyzer-storage-fuel cell units for local emergency power. These are advantageous for emergency use because of their low maintenance requirement and ease of location compared to internal combustion driven generators.[citation needed]
Iceland has committed to becoming the world's first hydrogen economy by the year 2050.[63] Iceland is in a unique position. Presently, it imports all the petroleum products necessary to power its automobiles and fishing fleet. Iceland has large geothermal resources, so much that the local price of electricity actually is lower than the price of the hydrocarbons that could be used to produce that electricity.
Iceland already converts its surplus electricity into exportable goods and hydrocarbon replacements. In 2002, it produced 2,000 tons of hydrogen gas by electrolysis—primarily for the production of ammonia (NH3) for fertilizer. Ammonia is produced, transported, and used throughout the world, and 90% of the cost of ammonia is the cost of the energy to produce it. Iceland is also developing an aluminium -smelting industry. Aluminium costs are primarily driven by the cost of the electricity to run the smelters. Either of these industries could effectively export all of Iceland's potential geothermal electricity.
Neither industry directly replaces hydrocarbons. Reykjavík, Iceland, had a small pilot fleet of city buses running on compressed hydrogen,[64] and research on powering the nation's fishing fleet with hydrogen is under way. For more practical purposes, Iceland might process imported oil with hydrogen to extend it, rather than to replace it altogether.
The Reykjavík buses are part of a larger program, HyFLEET:CUTE,[65] operating hydrogen fueled buses in eight European cities. HyFLEET:CUTE buses also operate in Beijing and Perth (see below).
A pilot project demonstrating a hydrogen economy is operational on the Norwegian island of Utsira. The installation combines wind power and hydrogen power. In periods when there is surplus wind energy, the excess power is used for generating hydrogen by electrolysis. The hydrogen is stored, and is available for power generation in periods when there is little wind.
A joint venture between NREL and Xcel Energy is combining wind power and hydrogen power in the same way in Colorado.[66]
In the Netherlands, one fossil fuel power plant (with full carbon capture) is already scheduled to incorporate the generating of hydrogen. The project is called CGEN and the power plant wil be built in the port of Rotterdam.[67]
Hydro in Newfoundland and Labrador are converting the current wind-diesel Power System on the remote island of Ramea into a Wind-Hydrogen Hybrid Power Systems facility.[68]
A similar pilot project on Stuart Island uses solar power, instead of wind power, to generate electricity. When excess electricity is available after the batteries are full, hydrogen is generated by electrolysis and stored for later production of electricity by fuel cell.[69]
The UK started a fuel cell pilot program in January 2004, the program ran two Fuel cell buses on route 25 in London until December 2005, and switched to route RV1 until January 2007.[70]
The Hydrogen Expedition is currently working to create a hydrogen fuel cell-powered ship and using it to circumnavigate the globe, as a way to demonstrate the capability of hydrogen fuel cells.[71]
Western Australia's Department of Planning and Infrastructure currently operates three Daimler Chrysler Citaro fuel cell buses as part of its Sustainable Transport Energy for Perth Fuel Cells Bus Trial in Perth.[72] The buses are operated by Path Transit on regular Transperth public bus routes. The trial began in September 2004 and concluded in September 2006. The buses' fuel cells use a proton exchange membrane system and are supplied with raw hydrogen from a BP refinery in Kwinana, south of Perth. The hydrogen is a byproduct of the refinery's industrial process. The buses are refueled at a station in the northern Perth suburb of Malaga.
The United Nations Industrial Development Organization (UNIDO) and the Turkish Ministry of Energy and Natural Resources have signed in 2003 a $40M Trust Fund Agreement for the creation in Istanbul of the International Centre for Hydrogen Energy Technologies (UNIDO-ICHET), which started operation in 2004.[73] A hydrogen forklift, a hydrogen cart and a mobile house powered by renewable energies are being demonstrated in UNIDO-ICHET's premises. An uninterruptible power supply system has been working since April 2009 in the headquarters of Istanbul Sea Buses company.
Hydrogen-using alternatives to a fully distributive hydrogen economy
For other energy alternatives, see
Main article: Energy developmentHydrogen is simply a method to store and transmit energy. Various alternative energy transmission and storage scenarios which begin with hydrogen production, but do not use it for all parts of the store and transmission infrastructure, may be more economic, in both near and far term. These include:
Ammonia economy
An alternative to gaseous hydrogen as an energy carrier is to bond it with nitrogen from the air to produce ammonia, which can be easily liquefied, transported, and used (directly or indirectly) as a clean and renewable fuel.[74][75]
Hydrogen production of greenhouse-neutral alcohol
Main articles: Methanol economy and ethanol economyThe methanol economy is a synfuel production energy plan which may begin with hydrogen production. Hydrogen in a full "hydrogen economy" was initially suggested as a way to make renewable energy, in non-polluting form, available to automobiles. However, a theoretical alternative to address the same problem is to produced hydrogen centrally and immediately use it to make liquid fuels from a CO2 source. This would eliminate the requirement to transport and store the hydrogen. The source could be CO2 that is produced by fuel-burning power plants. In order to be greenhouse-neutral, the source for CO2 in such a plan would need to be from air, biomass, or other source of CO2 which is already in, or to be released into, the air.)[citation needed]. Direct methanol fuel cells are in commercial use, though they are not presently efficient.[citation needed]
The electrical grid plus synthetic methanol fuel cells
Many of the hybrid strategies described above, using captive hydrogen to generate other more easily usable fuels, might be more effective than hydrogen-production alone. Short term energy storage (meaning the energy is used not long after it has been captured) may be best accomplished with battery or even ultracapacitor storage. Longer term energy storage (meaning the energy is used weeks or months after capture) may be better done with synthetic methane or alcohols, which can be stored indefinitely at relatively low cost, and even used directly in some type of fuel cells, for electric vehicles. These strategies dovetail well with the recent interest in Plug-in Hybrid Electric Vehicles, or PHEVs, which use a hybrid strategy of electrical and fuel storage for their energy needs. Hydrogen storage has been proposed by some[citation needed] to be optimal in a narrow range of energy storage time, probably somewhere between a few days and a few weeks. This range is subject to further narrowing with any improvements in battery technology. It is always possible that some kind of breakthrough in hydrogen storage or generation could occur, but this is unlikely given the physical and chemical limitations of the technical choices are fairly well understood.
Captive hydrogen synthetic methane production
In a similar way as with synthetic alcohol production, hydrogen can be used on-site to directly (nonbiologically) produce greenhouse-neutral gaseous fuels. Thus, captive-hydrogen-mediated production of greenhouse-neutral methane has been proposed (note that this is the reverse of the present method of acquiring hydrogen from natural methane, but one that does not require ultimate burning and release of fossil fuel carbon). Captive hydrogen (and carbon dioxide) may be used onsite to synthesize methane, using the Sabatier reaction. This process is about 80% efficient, reducing the round trip efficiency to about 20 to 30%, depending on the method of fuel utilization. This is even lower than hydrogen, but the storage costs drop by at least a factor of 3, because of methane's higher boiling point and higher energy density. Liquid methane has 3.2 times the energy density of liquid hydrogen and is easier to store. Additionally, the pipe infrastructure (natural gas pipelines) are already in place. Natural-gas-powered vehicles already exist, and are known to be easier to adapt from existing internal engine technology, than internal combustion autos running directly on hydrogen. Experience with natural gas powered vehicles shows that methane storage is inexpensive, once one has accepted the cost of conversion to store the fuel. However, the cost of alcohol storage is even lower, so this technology would need to produce methane at a considerable savings with regard to alcohol production. Ultimate mature prices of fuels in the competing technologies are not presently known, but both are expected to offer substantial infrastructural savings over attempts to transport and use hydrogen directly.
See also
- Alternative fuel
- Energy development
- Fuel Cells and Hydrogen Joint Technology Initiative
- HOPE Curriculum (Hydrogen Outreach Program for Education)
- Hydrogen energy plant in Denmark
- Qazvin hydrogen power plant
- Hydrogen internal combustion engine vehicle
- Hydrogen prize
- Hydrogen vehicle
- International Centre for Hydrogen Energy Technologies
- International Journal of Hydrogen Energy
- Lolland Hydrogen Community
References
- ^ National Hydrogen Association; United States Department of Energy. "The History of Hydrogen". hydrogenassociation.org. National Hydrogen Association. p. 1. http://www.hydrogenassociation.org/general/factSheet_history.pdf. Retrieved 17 December 2010.
- ^ Jones, Lawrence W (13 March 1970). "Toward a liquid hydrogen fuel economy" (PDF). University of Michigan Environmental Action for Survival Teach In. Ann Arbor, Michigan: University of Michigan. http://hdl.handle.net/2027.42/5800. Retrieved 17 December 2010.
- ^ a b National Academy of Engineering (2004). The Hydrogen Economy: Opportunities, Costs, Barriers, and R&D Needs. Washington, D.C.: The National Academies Press. ISBN 0-309-53068-7. http://www.nap.edu/catalog.php?record_id=10922. Retrieved 17 December 2010.
- ^ BMW Group Clean Energy ZEV Symposium. September 2006, p. 12
- ^ "EPA mileage estimates". Honda FCX Clarity - Vehicle Specifications. American Honda Motor Company. http://automobiles.honda.com/fcx-clarity/specifications.aspx?group=epa. Retrieved 17 December 2010.
- ^ "Fuel Cells: Cost". California Distributed Energy Resource Guide. California Energy Commission. 18 January 2002. http://www.energy.ca.gov/distgen/equipment/fuel_cells/cost.html. Retrieved 17 December 2010.
- ^ Tester, Jefferson W; Drake, Elisabeth M; Driscoll, Michael J; Golay, Michael W; Peters, William A (July 2005). Sustainable Energy: Choosing Among Options. Cambridge, MA: MIT Press. ISBN 978-0-262-20153-7. OCLC 58451915.
- ^ "Integrated Hydrogen Production, Purification and Compression System; DOE Hydrogen Program FY 2008 Annual Progress Report" (PDF). United States Department of Energy. http://www.hydrogen.energy.gov/pdfs/progress08/ii_a_7_tamhankar.pdf. Retrieved 2009-09-19.
- ^ "Arno A. Evers FAIR-PR". Fair-pr.de. http://www.fair-pr.de/background/worldwide-hydrogen-production-analysis.php. Retrieved 2009-09-19.
- ^ "Leeds researchers fuelling the ‘hydrogen economy’". University of Leeds. 26 November 2007. http://reporter.leeds.ac.uk/press_releases/current/biodiesel.htm. Retrieved 2008-05-09.
- ^ "Configuration and Technology Implications of Potential Nuclear Hydrogen System Applications" (PDF). Argonne National Laboratory. July 31, 2005. p. 16. Archive copy at the Wayback Machine
- ^ "Global Hydrogen Production". Hydrogenassociation.org. 2004-08-13. http://www.hydrogenassociation.org/general/faqs.asp#howmuchproduced. Retrieved 2010-07-05.
- ^ "Assessing Current, Near-term, and Long-term U.S. Hydrogen Markets". Argonne National Laboratory. Archive copy at the Wayback Machine
- ^ a b "Vehicle Technologies Program: Fact #205: February 25, 2002 Hydrogen Cost and Worldwide Production". .eere.energy.gov. http://www1.eere.energy.gov/vehiclesandfuels/facts/favorites/fcvt_fotw205.html. Retrieved 2009-09-19.
- ^ a b "Bellona-HydrogenReport". Interstatetraveler.us. http://www.interstatetraveler.us/Reference-Bibliography/Bellona-HydrogenReport.html. Retrieved 2010-07-05.
- ^ "Actual Worldwide Hydrogen Production from …". Arno A Evers. December 2008. http://www.fair-pr.de/background/worldwide-hydrogen-production-analysis.php. Retrieved 2008-05-09.
- ^ https://www.hfpeurope.org/infotools/energyinfos__e/hydrogen/main03.html
- ^ "High hydrogen yield from a two-step process of dark-and photo-fermentation of sucrose". Cat.inist.fr. http://cat.inist.fr/?aModele=afficheN&cpsidt=18477081. Retrieved 2010-07-05.
- ^ "Hydrogen production from organic solid matter". Biohydrogen.nl. http://www.biohydrogen.nl/hyvolution/25446/5/0/20. Retrieved 2010-07-05.
- ^ "Power from plants using microbial fuel cell" (in (Dutch)). Translate.google.com. http://translate.google.com/translate?js=n&prev=_t&hl=nl&ie=UTF-8&u=http%3A%2F%2Fwww.resource-online.nl%2Fachtergrond.php%3Fid%3D147&sl=nl&tl=en&history_state0=. Retrieved 2010-07-05.
- ^ Werner Zittel; Reinhold Wurster (1996-07-08). "Chapter 3: Production of Hydrogen. Part 4: Production from electricity by means of electrolysis". HyWeb: Knowledge - Hydrogen in the Energy Sector. Ludwig-Bölkow-Systemtechnik GmbH. http://www.hyweb.de/Knowledge/w-i-energiew-eng3.html#3.4.
- ^ Bjørnar Kruse; Sondre Grinna, Cato Buch (2002-02-13). "Hydrogen—Status and Possibilities" (pdf). The Bellona Foundation. Archived from the original on 2002-02-13. http://www.bellona.org/filearchive/fil_Hydrogen_6-2002.pdf. "Efficiency factors for PEM electrolysers up to 94% are predicted, but this is only theoretical at this time."
- ^ high-rate and high efficiency 3D water electrolysis
- ^ Crabtree, George W.; Mildred S. Dresselhaus, and Michelle V. Buchanan (December 2004). "The Hydrogen Economy". Physics Today. pp. 39. http://scitation.aip.org/journals/doc/PHTOAD-ft/vol_57/iss_12/39_1.shtml. Retrieved 2008-05-09.
- ^ "2001-High pressure electrolysis - The key technology for efficient H.2". http://www.fz-juelich.de/scientific-report-2001/docs/patente/26_11600.pdf?web_session=e4af90eba1518be519b2c1b61fa42cfb. Retrieved 2010-07-05.
- ^ "2003-PHOEBUS-Pag.9" (PDF). http://www.fz-juelich.de/ief/ief-3/datapool/page/214/solar%20energy%2075%20469-478.pdf. Retrieved 2010-07-05.
- ^ "Steam heat: researchers gear up for full-scale hydrogen plant" (Press release). Science Daily. 2008-09-18. http://www.sciencedaily.com/releases/2008/09/080918170624.htm. Retrieved 2008-09-19.
- ^ "Nuclear Hydrogen R&D Plan" (PDF). U.S. Dept. of Energy. March 2004. http://www.hydrogen.energy.gov/pdfs/nuclear_energy_h2_plan.pdf. Retrieved 2008-05-09.
- ^ del Valle, F. et al; Álvarez Galván, M. Consuelo; Del Valle, F.; Villoria De La Mano, José A.; Fierro, José L. G. (Jun 2009). "Water Splitting on Semiconductor Catalysts under Visible-Light Irradiation". Chemsuschem (WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim) 2 (6): 471–485. doi:10.1002/cssc.200900018. PMID 19536754.
- ^ del Valle, F. et al; Del Valle, F.; Villoria De La Mano, J.A.; Álvarez-Galván, M.C.; Fierro, J.L.G. (2009). "Photocatalytic water splitting under visible Light: concept and materials requirements". Advances in Chemical Engineering (Academic Press) 36: 111–143. doi:10.1016/S0065-2377(09)00404-9.
- ^ "DLR Portal - DLR scientists achieve solar hydrogen production in a 100-kilowatt pilot plant". Dlr.de. 2008-11-25. http://www.dlr.de/en/desktopdefault.aspx/tabid-1/86_read-14380/. Retrieved 2009-09-19.
- ^ http://www3.interscience.wiley.com/journal/123275459/abstract
- ^ "353 Thermochemical cycles" (PDF). http://www.hydrogen.energy.gov/pdfs/review06/pd_10_weimer.pdf. Retrieved 2010-07-05.
- ^ UNLV Thermochemical cycle automated scoring database (public)
- ^ "Development of Solar-powered Thermochemical Production of Hydrogen from Water" (PDF). http://www.hydrogen.energy.gov/pdfs/review05/pd28_weimer.pdf. Retrieved 2010-07-05.
- ^ "1994 - ECN abstract". Hyweb.de. http://www.hyweb.de/Knowledge/Ecn-h2a.html. Retrieved 2010-07-05.
- ^ Bioenergy and renewable power methane in integrated 100% renewable energy system, [1]
- ^ scénario négaWatt 2011 (France), [2],
- ^ Idaho National Engineering Laboratory's recommendation for Gaseous Hydrogen: Stainless steel Accessed 2010-10-13
- ^ Stuart Island Energy Initiative Website Accessed 2010-10-13: Hydrogen has an active electron, and therefore behaves somewhat like a Halogen. The recommended pipe material is stainless steel.
- ^ Gross Britta K, Sutherland Ian J, Mooiweer Henk (December 2007). "Hydrogen fueling infrastructure assessment" (PDF). General Motors Research & Development Center. http://www.h2andyou.org/pdf/10Things.pdf. Retrieved 2008-09-19.[dead link]
- ^ Nakicenovic, 1998.
- ^ Electricity from wood through the combination of gasification and solid oxide fuel cells, Ph.D. Thesis by Florian Nagel, Swiss Federal Institute of Technology Zurich, 2008
- ^ "Power-to-weight ratio". .eere.energy.gov. 2009-06-23. http://www1.eere.energy.gov/hydrogenandfuelcells/fuelcells/fc_types.html. Retrieved 2010-07-05.
- ^ Boyd, Robert S. (May 15, 2007). "Hydrogen cars may be a long time coming". McClatchy Newspapers. http://www.mcclatchydc.com/staff/robert_boyd/story/16179.html. Retrieved 2008-05-09.
- ^ Squatriglia, Chuck (May 12, 2008). "Hydrogen Cars Won't Make a Difference for 40 Years". Wired. CondéNet, Inc. http://www.wired.com/cars/energy/news/2008/05/hydrogen. Retrieved 2008-05-13.
- ^ "Ford Motor Company Business Plan", December 2, 2008
- ^ Kreith, 2004
- ^ "The 21st Century Electric Car" (PDF). Tesla Motors. http://www.teslamotors.com/display_data/twentyfirstcenturycar.pdf.
- ^ Marisvensson, A; Ann Mari Svensson, Steffen Møller-Holst, Ronny Glöckner and Ola Maurstad (September 2006). "Well-to-wheel study of passenger vehicles in the Norwegian energy system". Energy 32 (4): 437–45. doi:10.1016/j.energy.2006.07.029. http://www.sciencedirect.com/science?_ob=ArticleURL&_udi=B6V2S-4M04DW9-1&_user=440026&_rdoc=1&_fmt=&_orig=search&_sort=d&view=c&_acct=C000020939&_version=1&_urlVersion=0&_userid=440026&md5=e7b3b8bfb288aaed28ee38d247e49a24. Retrieved 2008-07-22.
- ^ Utgikar, Vivek P; Thiesen, Todd (2005). "Safety of compressed hydrogen fuel tanks: Leakage from stationary vehicles". Technology in Society 27 (3): 315–320. doi:10.1016/j.techsoc.2005.04.005.
- ^ "DOE codes and standards". Hydrogen.energy.gov. http://www.hydrogen.energy.gov/codes_standards.html. Retrieved 2010-07-05.
- ^ "Hydrogen Sensor: Fast, Sensitive, Reliable, and Inexpensive to Produce" (PDF). Argonne National Laboratory. September 2006. http://www.anl.gov/techtransfer/pdf/Profile_HydrogenSensor9_06.pdf. Retrieved 2008-05-09.
- ^ "Canadian Hydrogen Safety Program testing H2/CNG". Hydrogenandfuelcellsafety.info. http://www.hydrogenandfuelcellsafety.info/2007/jun/h2cng.asp. Retrieved 2010-07-05.
- ^ "The Hidden Danger of Hydrogen Power". http://www.sparkplugengineering.com/. Retrieved 2009-06-04.
- ^ "Photochemical Ozone Production or Summer Smog". http://www.scienceinthebox.com/en_UK/sustainability/summersmog_en.html. Retrieved 2005.
- ^ a b "Assessing the Future Hydrogen Economy (letters)" (PDF). Science. 10 October 2003. http://rael.berkeley.edu/files/2003/Kammen-Tromp-Science-2003.pdf. Retrieved 2008-05-09.
- ^ "ITM Power makes bi-fuel breakthrough | Energy Archive". Businessweekly.co.uk. 2007-07-04. http://www.businessweekly.co.uk/2007070421235/energy-archive/itm-power-makes-bi-fuel-breakthrough.html. Retrieved 2009-09-19.[dead link]
- ^ Keith, Geoffrey; William Leighty (28 September 2002). "Transmitting 4,000 MW of New Windpower from North Dakota to Chicago: New HVDC Electric Lines or Hydrogen Pipeline" (PDF). http://www.ef.org/documents/NDakotaWindPower.pdf. Retrieved 2008-05-09.
- ^ "Mileage From Megawatts: Study Finds Enough Electric Capacity to 'Fill Up' Plug-In Vehicles Across Much of the Nation". December 11, 2006. http://newswire.ascribe.org/cgi-bin/behold.pl?ascribeid=20061211.105149&time=11%2005%20PST&year=2006&public=0. Retrieved 2008-05-09.
- ^ Wise, Jeff (November 2006). "The Truth About Hydrogen". Popular Mechanics. pp. 3. http://www.popularmechanics.com/technology/industry/4199381.html?page=3. Retrieved 2008-05-09.
- ^ "DOE Announces New Hydrogen Cost Goal". U.S. DoE. July 14, 2005. http://www1.eere.energy.gov/hydrogenandfuelcells/news_cost_goal.html. Retrieved 2008-05-09.
- ^ Hannesson, Hjálmar W. (2.8.2007). "Climate change as a global challenge". Iceland Ministry for Foreign Affairs. http://www.mfa.is/speeches-and-articles/nr/3800. Retrieved 2008-05-09.
- ^ Doyle, Alister (January 14, 2005). "Iceland's hydrogen buses zip toward oil-free economy". Reuters. http://www.detnews.com/2005/autosinsider/0501/14/autos-60181.htm. Retrieved 2008-05-09.[dead link]
- ^ "What is HyFLEET:CUTE?". http://www.global-hydrogen-bus-platform.com/index.php. Retrieved 2008-05-09.
- ^ "Experimental 'wind to hydrogen' system up and running". Physorg.com. January 8, 2007. http://www.physorg.com/news87494382.html. Retrieved 2008-05-09.
- ^ CGEN
- ^ "Hydrogen Engine Center Receives Order for Hydrogen Power Generator 250kW Generator for Wind/Hydrogen Demonstration" (PDF). Hydrogen Engine Center, Inc.. May 16, 2006. http://www.hydrogenenginecenter.com/userdocs/NRCan_Press_Release_Final_05.16.06.pdf. Retrieved 2008-05-09.
- ^ "Stuart Island Energy Initiative". http://www.siei.org. Retrieved 2008-05-09.
- ^ "Hydrogen buses". Transport for London. Archived from the original on March 23, 2008. http://web.archive.org/web/20080323064054/http://www.tfl.gov.uk/corporate/projectsandschemes/environment/2017.aspx#routes. Retrieved 2008-05-09.
- ^ "The Hydrogen Expedition" (PDF). January 2005. http://www.atti-info.org/HydrogenVeh/prospectus.pdf. Retrieved 2008-05-09.
- ^ "Perth Fuel Cell Bus Trial". Department for Planning and Infrastructure, Government of Western Australia. 13 April 2007. http://www.dpi.wa.gov.au/ecobus/1206.asp. Retrieved 2008-05-09.
- ^ "Independent Mid-Term Review of the UNIDO Project: Establishment and operation of the International Centre for Hydrogen Energy Technologies (ICHET), TF/INT/03/002". UNIDO. 31 August 2009. http://www.unido.org/fileadmin/user_media/About_UNIDO/Evaluation/TORs/TOR%20ICHET%20final.PDF. Retrieved 2010-07-20.
- ^ Agosta, Vito (July 10, 2003). "The Ammonia Economy". http://www.memagazine.org/contents/current/webonly/webex710.html. Retrieved 2008-05-09.
- ^ "Renewable Energy". Iowa Energy Center. http://www.energy.iastate.edu/Renewable/ammonia/index.htm. Retrieved 2008-05-09.
Further reading
- Arno A. Evers (2010). The Hydrogen Society. Hydrogeit Verlag. ISBN 978-3-937863-31-3.
- Jeremy Rifkin (2002). The Hydrogen Economy. Penguin Putnam Inc. ISBN 1-58542-193-6.
- Roy McAlister (2003). The Solar Hydrogen Civilization. American Hydrogen Association. ISBN 0-9728375-0-7.
- Joseph J. Romm (2004). The Hype about Hydrogen, Fact and Fiction in the Race to Save the Climate. Island Press. ISBN 1-55963-703-X. Author interview at Global Public Media.
- James Howard Kunstler (2006). The LONG EMERGENCY. Grove Press. ISBN 0-8021-4249-4. Hydrogen economy = "laughable a fantasy" p. 115
- M. Wang (2002). "Fuel Choices for Fuel Cell Vehicles: Well-to-Wheels Energy and Emissions Impact". Journal of Power Sources 112: 307–321. doi:10.1016/S0378-7753(02)00447-0.
- F. Kreith; West, Ron (2004). "Fallacies of a Hydrogen Economy: A Critical Analysis of Hydrogen Production and Utilization". Journal of Energy Resources Technology 126 (4): 249–257. doi:10.1115/1.1834851.
- Nakicenovic (1998). Global Energy Perspectives. Cambridge University Press. Summary
- National Research Council (2004). The Hydrogen Economy: Opportunities, Costs, Barriers, and R&D Needs. National Academy Press. This book is available online in full text: "The Hydrogen Economy: Opportunities, Costs, Barriers, and R&D Needs". Books.nap.edu. http://books.nap.edu/openbook.php?record_id=10922&page=1.
- Novelli, P.C., P.M. Lang, K.A. Masarie, D.F. Hurst, R. Myers, and J.W. Elkins. (1999). "Molecular Hydrogen in the troposphere: Global distribution and budget". J. Geophys. Res. 104 (30): 427–30.
- T. K. Tromp; Shia, RL; Allen, M; Eiler, JM; Yung, YL (2003). "Potential Environmental Impact of a Hydrogen Economy on the Stratosphere". Science 300 (5626): 1740–1742. doi:10.1126/science.1085169. PMID 12805546.
External links
- Hydrogen Society
- International Partnership for the Hydrogen Economy
- European Hydrogen Association
- European Network of Excellence Safety of Hydrogen as an Energy Carrier (HySafe)
- World's First Higher Educational Programme in Hydrogen Safety Engineering
- Canada
- U.S.-Department of Energy
- European Projects 2002-2006 FP6
- European Projects 2007-2013 FP7
- 20 Hydrogen myths - Published by the Rocky Mountain Institute, a major hydrogen economy proponent.
- Does a Hydrogen Economy Make Sense?
- Hydrogen and Fuel Cell Wiki
- ITM Power - Economic renewable hydrogen from low cost materials (non platinum, fluorocarbon free) & manufacturing processes - electrolyzers & fuel cells
- The Hydrogen Hoax article by Robert Zubrin in The New Atlantis
- Resources on hydrogen plants
Categories:
Wikimedia Foundation. 2010.