- Methane
-
Methane 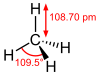

 Other names
Other namesIdentifiers CAS number 74-82-8 
PubChem 297 ChemSpider 291 
EC number 200-812-7 UN number 1971 KEGG C01438 
MeSH Methane ChEBI CHEBI:16183 
ChEMBL CHEMBL17564 
RTECS number PA1490000 Beilstein Reference 1718732 Gmelin Reference 59 3DMet B01450 Jmol-3D images Image 1 - C
Properties Molecular formula CH4 Molar mass 16.04 g mol−1 Exact mass 16.031300128 g mol−1 Appearance Colorless gas Odor Odourless gas Density 716 μg cm−3 Melting point -187 °C, 86 K, -305 °F
Boiling point -161 °C, 112 K, -258 °F
Solubility in water 35 mg dm−3 (at 17 °C) log P 1.09 Thermochemistry Std enthalpy of
formation ΔfHo298-74.87 kJ mol−1 Std enthalpy of
combustion ΔcHo298-891.1—890.3 kJ mol−1 Standard molar
entropy So298186.25 J K−1 mol−1 Specific heat capacity, C 35.69 J K−1 mol−1 Hazards MSDS External MSDS GHS pictograms 
GHS signal word DANGER GHS hazard statements H220, H280 GHS precautionary statements P210, P410+403 EU Index 601-001-00-4 EU classification  F+
F+R-phrases R12 S-phrases (S2), S9, S16, S33 NFPA 704 Flash point –188 °C Autoignition
temperature537 °C Explosive limits 5–15% [2] Related compounds Related alkanes Ethane
Related compounds Chloromethane
Supplementary data page Structure and
propertiesn, εr, etc. Thermodynamic
dataPhase behaviour
Solid, liquid, gasSpectral data UV, IR, NMR, MS  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Methane (pronounced /ˈmɛθeɪn/ or /ˈmiːθeɪn/) is a chemical compound with the chemical formula CH4. It is the simplest alkane, the principal component of natural gas, and probably the most abundant organic compound on earth. The relative abundance of methane makes it an attractive fuel. However, because it is a gas at normal conditions, methane is difficult to transport from its source.
Methane is a relatively potent greenhouse gas. The concentration of methane in the Earth's atmosphere in 1998, expressed as a mole fraction, was 1745 nmol/mol (parts per billion, ppb), up from 700 nmol/mol in 1750. By 2008, however, global methane levels, which had stayed mostly flat since 1998, had risen to 1,800 nmol/mol.[3]
Contents
Properties and bonding
Methane is a tetrahedral molecule with four equivalent C-H bonds. Its electronic structure is described by four bonding molecular orbitals (MOs) resulting from the overlap of the valence orbitals on C and H. The lowest energy MO is the result of the overlap of the 2s orbital on carbon with the in-phase combination of the 1s orbitals on the four hydrogen atoms. Above this level in energy is a triply degenerate set of MOs that involve overlap if the 2p orbitals on carbon with various linear combinations of the 1s orbitals on hydrogen. The resulting "three-over-one" bonding scheme is consistent with photoelectron spectroscopic measurements.
At room temperature and standard pressure, methane is a colorless, odorless gas.[4] The familiar smell of natural gas as used in homes is a safety measure achieved by the addition of an odorant, often methanethiol or ethanethiol. Methane has a boiling point of −161 °C (−257.8 °F) at a pressure of one atmosphere.[5] As a gas it is flammable only over a narrow range of concentrations (5–15%) in air. Liquid methane does not burn unless subjected to high pressure (normally 4–5 atmospheres).[6]
Chemical reactions
Main reactions with methane are: combustion, steam reforming to syngas, and halogenation. In general, methane reactions are difficult to control. Partial oxidation to methanol, for example, is challenging because the reaction typically progresses all the way to carbon dioxide and water even with incomplete amounts of oxygen. The enzymes methane monooxygenase can produce methanol from methane, but they cannot be used for industrial scale reactions.[7]
Acid-base reactions
Like other hydrocarbons, methane is a very weak acid. Its pKa in DMSO is estimated to be 56.[8] It cannot be deprotonated in solution, but the conjugate base methyl lithium is known. Protonation of methane can be achieved with super acids to give CH5+, sometimes called the methanium ion. Despite the strength of its C-H bonds, there is intense interest in catalysts that facilitate C–H bond activation in methane (and other low alkanes).[9]
Combustion
In the combustion of methane, several steps are involved. An early intermediate is formaldehyde (HCHO or H2CO). Oxidation of formaldehyde gives the formyl radical (HCO), which then give carbon monoxide (CO):
- CH4 + O2 → CO + H2 + H2O
The resulting H2 oxidizes to H2O, releasing heat. This reaction occurs very quickly, usually in significantly less than a millisecond.
- 2 H2 + O2 → 2 H2O
Finally, the CO oxidizes, forming CO2 and releasing more heat. This process is generally slower than the other chemical steps, and typically requires a few to several milliseconds to occur.
- 2 CO + O2 → 2 CO2
The result of the above is the following total equation:
Reactions with halogens
Methane reacts with halogens given appropriate conditions as follows:
- CH4 + X2 → CH3X + HX
where X is a halogen: fluorine (F), chlorine (Cl), bromine (Br), or iodine (I). This mechanism for this process is called free radical halogenation, beginning with the attach of Cl· radicals on methane to produce CH3·, which combines with a second Cl· to give methyl chloride (CH3Cl). Similar reactions will produce dichloromethane (CH2Cl2), chloroform(CHCl3), and, ulitimately, carbon tetrachloride (CCl4). The energy requied to start this reaction comes from UV radiation or heating.[10]
Uses
Methane is used in industrial chemical processes and may be transported as a refrigerated liquid (liquefied natural gas, or LNG). While leaks from a refrigerated liquid container are initially heavier than air due to the increased density of the cold gas, the gas at ambient temperature is lighter than air. Gas pipelines distribute large amounts of natural gas, of which methane is the principal component.
Fuel
Main article: natural gasMethane is important for electrical generation by burning it as a fuel in a gas turbine or steam boiler. Compared to other hydrocarbon fuels, burning methane produces less carbon dioxide for each unit of heat released. At about 891 kJ/mol, methane's heat of combustion is lower than any other hydrocarbon but the ratio of the heat of combustion (891 kJ/mol) to the molecular mass (16.0 g/mol, of which 12.0 g/mol is carbon) shows that methane, being the simplest hydrocarbon, produces more heat per mass unit (55.7 kJ/g) than other complex hydrocarbons. In many cities, methane is piped into homes for domestic heating and cooking purposes. In this context it is usually known as natural gas, and is considered to have an energy content of 39 megajoules per cubic meter, or 1,000 BTU per standard cubic foot.
Methane in the form of compressed natural gas is used as a vehicle fuel and is claimed to be more environmentally friendly than other fossil fuels such as gasoline/petrol and diesel.[11]
Research into adsorption methods of methane storage for this purpose has been conducted.[12]
Developing technologies
Research is being conducted by NASA on methane's potential as a rocket fuel.[13]
Methane emitted from coal mines has been converted to electricity.[14]
Chemical feedstock
Although there would is great interest in converting methane into useful or more easily liquified compounds, the only practical processes are relatively unselective. In the chemical industry, methane is converted to synthesis gas, a mixture of carbon monoxide and hydrogen, by steam reforming. This endergonic process (requiring energy) utilizes nickel catalysts and requires high temperatures, around 700–1100 °C:
- CH4 + 2 H2O → CO2 + 4 H2
Related chemistries are exploited in the Haber-Bosch Synthesis of ammonia from air, which is reduced with natural gas to a mixture of carbon dioxide, water, and ammonia.
Methane is also subjected to free-radical chlorination in the production of chloromethanes, although methanol is a more typical precursor.[10]
Production
Biological routes
Main article: methanogenesisNaturally occurring methane is mainly produced by the process of methanogenesis. This multistep process is used by microorganisms as an energy source. The net reaction is:
- CO2 + 8 H+ + 8 e- → CH4 + 2 H2O
The final step in the process is catalysed by the enzyme methyl-coenzyme M reductase. Methanogenesis is a form of anaerobic respiration used by organisms that occupy landfill, ruminants (e.g., cattle), and the guts of termites.
It is uncertain if plants are a source of methane emissions.[15][16][17]
Industrial routes
Natural gas is so abundant that the intentional production of methane would be unusual. Methane can be produced by hydrogenation carbon dioxide through the Sabatier process. It is also a side product of the hydrogenation of carbon monoxide in the Fischer-Tropsch process. This technology is practiced on a large scale to produce longer chain molecules than methane.
Occurrence
Methane was discovered and isolated by Alessandro Volta between 1776 and 1778 when studying marsh gas from Lake Maggiore. It is the major component of natural gas, about 87% by volume. The major source of methane is extraction from geological deposits known as natural gas fields, with coal seam gas extraction becoming a major source (see Coal bed methane extraction, a method for extracting methane from a coal deposit, while enhanced coal bed methane recovery is a method of recovering methane from an non-minable coal seams). It is associated with other hydrocarbon fuels, and sometimes accompanied by helium and nitrogen. The gas at shallow levels (low pressure) forms by anaerobic decay of organic matter and reworked methane from deep under the Earth's surface. In general, sediments buried deeper and at higher temperatures than those that contain oil generate natural gas.
It is generally transported in bulk by pipeline in its natural gas form, or LNG carriers in its liquefied form; few countries transport it by truck.
Alternative sources
Apart from gas fields, an alternative method of obtaining methane is via biogas generated by the fermentation of organic matter including manure, wastewater sludge, municipal solid waste (including landfills), or any other biodegradable feedstock, under anaerobic conditions. Rice fields also generate large amounts of methane during plant growth. Methane hydrates/clathrates (ice-like combinations of methane and water on the sea floor, found in vast quantities) are a potential future source of methane. Cattle belch methane accounts for 16% of the world's annual methane emissions to the atmosphere.[18] One study reported that the livestock sector in general (primarily cattle, chickens, and pigs) produces 37% of all human-induced methane.[19] Early research has found a number of medical treatments and dietary adjustments that help slightly limit the production of methane in ruminants.[20] [21] A more recent study, in 2009, found that at a conservative estimate, at least 51% of global greenhouse gas emissions were attributable to the life cycle and supply chain of livestock products, meaning all meat, dairy, and by-products, and their transportation.[22]
Atmospheric methane
Main article: Atmospheric methane 2006–2009 methane concentration in the upper troposphere
2006–2009 methane concentration in the upper troposphere
Methane is created near the Earth's surface, primarily by microorganisms by the process of methanogenesis. It is carried into the stratosphere by rising air in the tropics. Uncontrolled build-up of methane in the atmosphere is naturally checked — although human influence can upset this natural regulation — by methane's reaction with hydroxyl radicals formed from singlet oxygen atoms and with water vapor. It has a net lifetime of about 10 years,[23] and is primarily removed by conversion to carbon dioxide and water
Methane also affects the degradation of the ozone layer.[24][25]
In addition, there is a large (but unknown) amount of methane in methane clathrates in the ocean floors as well as the Earth's crust. Most methane is the result of biological process called methanogenesis.
In 2010, methane levels in the Arctic were measured at 1850 nmol/mol, a level scientists described as being higher than at any time in the previous 400,000 years. Historically, methane concentrations in the world's atmosphere have ranged between 300 and 400 nmol/mol during glacial periods commonly known as ice ages, and between 600 to 700 nmol/mol during the warm interglacial periods. It has a high global warming potential: 72 times that of carbon dioxide over 20 years, and 25 times over 100 years,[26] and the levels are rising.
Methane in the Earth's atmosphere is an important greenhouse gas with a global warming potential of 25 compared to CO2 over a 100-year period (although accepted figures probably represents an underestimate[27]). This means that a methane emission will have 25 times the effect on temperature of a carbon dioxide emission of the same mass over the following 100 years. Methane has a large effect for a brief period (a net lifetime of 8.4 years in the atmosphere), whereas carbon dioxide has a small effect for a long period (over 100 years). Because of this difference in effect and time period, the global warming potential of methane over a 20 year time period is 72. The Earth's atmospheric methane concentration has increased by about 150% since 1750, and it accounts for 20% of the total radiative forcing from all of the long-lived and globally mixed greenhouse gases (these gases don't include water vapour which is by far the largest component of the greenhouse effect).[28] Usually, excess methane from landfills and other natural producers of methane is burned so CO2 is released into the atmosphere instead of methane, because methane is a more effective greenhouse gas. Recently, methane emitted from coal mines has been successfully utilized to generate electricity.
Clathrates
Arctic methane release from permafrost and methane clathrates is an expected consequence of global warming.[29]
Safety
Methane is not toxic; however, it is extremely flammable and may form explosive mixtures with air. Methane is violently reactive with oxidizers, halogens, and some halogen-containing compounds. Methane is also an asphyxiant and may displace oxygen in an enclosed space. Asphyxia may result if the oxygen concentration is reduced to below about 16% by displacement, as most people can tolerate a reduction from 21% to 16% without ill effects. The concentration of methane at which asphyxiation risk becomes significant is much higher than the 5–15% concentration in a flammable or explosive mixture. Methane off-gas can penetrate the interiors of buildings near landfills and expose occupants to significant levels of methane. Some buildings have specially engineered recovery systems below their basements to actively capture this gas and vent it away from the building.
References
- ^ a b "methane (CHEBI:16183)". Chemical Entities of Biological Interest. UK: European Bioinformatics Institute. 17 October 2009. Main. https://www.ebi.ac.uk/chebi/searchId.do?chebiId=16183. Retrieved 10 October 2011.
- ^ MSDS Methane
- ^ Carbon Dioxide, Methane Rise Sharply in 2007
- ^ David A. Hensher, Kenneth J. Button (2003). Handbook of transport and the environment. Emerald Group Publishing. p. 168. ISBN 0080441033. http://books.google.com/books?id=yp3qEgHrsJ4C&pg=PA168.
- ^ NIST Chemistry Webbook
- ^ Ayhan Demirbas (2010). Methane Gas Hydrate. Springer. p. 102. ISBN 1848828713. http://books.google.com/books?id=WFefWAq1Sh0C&pg=PA102.
- ^ Mu-Hyun Baik, Martin Newcomb, Richard A. Friesner, and Stephen J. Lippard "Mechanistic Studies on the Hydroxylation of Methane by Methane Monooxygenase" Chem. Rev., 2003, vol. 103, pp 2385–2420. doi:10.1021/cr950244f
- ^ Equilibrium acidities in dimethyl sulfoxide solution Frederick G. Bordwell Acc. Chem. Res.; 1988; 21(12) pp 456 - 463; doi:10.1021/ar00156a004
- ^ Wesley H. Bernskoetter, Cynthia K. Schauer, Karen I. Goldberg and Maurice Brookhart "Characterization of a Rhodium(I) σ-Methane Complex in Solution" Science 2009, Vol. 326, pp. 553-556. doi:10.1126/science.1177485
- ^ a b M. Rossberg et al. “Chlorinated Hydrocarbons” in Ullmann’s Encyclopedia of Industrial Chemistry 2006, Wiley-VCH, Weinheim. doi:10.1002/14356007.a06_233.pub2
- ^ Clayton B. Cornell (April 29, 2008). "Natural Gas Cars: CNG Fuel Almost Free in Some Parts of the Country". http://www.gas2.org/2008/04/29/natural-gas-cars-cng-fuel-almost-free-in-some-parts-of-the-country/. "Compressed natural gas is touted as the 'cleanest burning' alternative fuel available, since the simplicity of the methane molecule reduces tailpipe emissions of different pollutants by 35 to 97%. Not quite as dramatic is the reduction in net greenhouse-gas emissions, which is about the same as corn-grain ethanol at about a 20% reduction over gasoline"
- ^ Düren, Tina; Sarkisov, Lev; Yaghi, Omar M.; Snurr, Randall Q. (2004). "Design of New Materials for Methane Storage". Langmuir 20 (7): 2683–9. doi:10.1021/la0355500. PMID 15835137.
- ^ Lunar Engines, Aviation Week & Space Technology, 171, 2 (13 July 2009), p. 16: "Aerojet has completed assembly of a 5,500-pound-thrust liquid oxygen/liquid methane rocket engine—a propulsion technology under consideration as the way off the Moon for human explorers" One advantage of methane is that it is abundant in many parts of the solar system and it could potentially be harvested in situ (i.e. on the surface of another solar-system body), providing fuel for a return journey.Methane Blast, NASA, May 4, 2007. Current methane engines in development produce a thrust of 7,500 pounds-force (33 kN), which is far from the 7,000,000 lbf (31 MN) needed to launch the Space Shuttle. Instead, such engines will most likely propel voyages from the Moon or send robotic expeditions to other planets in the solar system.Green, V. (September). Hit the Gas: NASA's methane rocket could make long distance space travel possible, on the cheap. 271. Popular Science magazine. pp. 16–17. ISSN 0161-7370. http://books.google.com/?id=3AClY8pMg-EC&pg=PA16&lpg=PA16.
- ^ A Global First: Coal Mine Turns Greenhouse Gas into Green Energy
- ^ Hamilton JT, McRoberts WC, Keppler F, Kalin RM, Harper DB (July 2003). "Chloride methylation by plant pectin: an efficient environmentally significant process". Science 301 (5630): 206–9. Bibcode 2003Sci...301..206H. doi:10.1126/science.1085036. PMID 12855805.
- ^ "Methane Emissions? Don't Blame Plants", ScienceNOW, 14 January 2009
- ^ "Plants do emit methane after all". New Scientist. 2 December 2007. http://environment.newscientist.com/article/mg19626322.900-plants-do-emit-methane-after-all.html.
- ^ Miller, G. Tyler. Sustaining the Earth: An Integrated Approach. U.S.A.: Thomson Advantage Books, 2007. 160.
- ^ FAO (2006). Livestock’s Long Shadow–Environmental Issues and Options. Rome: Food and Agriculture Organization of the United Nations (FAO). http://www.fao.org/docrep/010/a0701e/a0701e00.HTM. Retrieved 2009-10-27.
- ^ John Roach (2002-05-13). "New Zealand Tries to Cap Gaseous Sheep Burps". National Geographic. http://news.nationalgeographic.com/news/2002/05/0509_020509_belch.html. Retrieved 2011-03-02.
- ^ Research on use of bacteria from the stomach lining of kangaroos (who don't emit methane) to reduce methane in cattle
- ^ Goodland, Robert, and Anhang, Jeff. (November/ December 2009), Livestock and Climate Change., Washington, D.C.: World Watch, www.worldwatch.org, http://en.wikipedia.org/wiki/Worldwatch_institute, http://www.worldwatch.org/files/pdf/Livestock%20and%20Climate%20Change.pdf
- ^ Boucher, Olivier; Friedlingstein, Pierre; Collins, Bill; Shine, Keith P (2009). "The indirect global warming potential and global temperature change potential due to methane oxidation". Environmental Research Letters 4 (4): 044007. Bibcode 2009ERL.....4d4007B. doi:10.1088/1748-9326/4/4/044007.
- ^ Ozon – wpływ na życie człowieka, Ozonowanie/Ewa Sroka, Group: Freony i inne związki, Reakcje rozkładu ozonu.
- ^ Twenty Questions And Answers About The Ozone Layer, UNEP/D.W. Fahey 2002, pp. 12, 34, 38
- ^ IPCC Fourth Assessment Report, Working Group 1, Chapter 2
- ^ Shindell, D. T.; Faluvegi, G.; Koch, D. M.; Schmidt, G. A.; Unger, N.; Bauer, S. E. (2009). "Improved Attribution of Climate Forcing to Emissions". Science 326 (5953): 716–8. Bibcode 2009Sci...326..716S. doi:10.1126/science.1174760. PMID 19900930.
- ^ "Technical summary". Climate Change 2001. United Nations Environment Programme. http://www.grida.no/climate/ipcc_tar/wg1/017.htm.
- ^ "Methane Releases From Arctic Shelf May Be Much Larger and Faster Than Anticipated". Press Release. National Science Foundation. http://www.nsf.gov/news/news_summ.jsp?cntn_id=116532&org=NSF&from=news.
Appendix: extraterrestrial methane
Methane has been detected or is believed to exist in several locations of the solar system. In most cases, it is believed to have been created by abiotic processes. Possible exceptions are Mars and Titan.
- Moon – traces are outgassed from the surface[1]
- Mars – the atmosphere contains 10 nmol/mol methane. In January 2009, NASA scientists announced that they had discovered that the planet often vents methane into the atmosphere in specific areas, leading some to speculate this may be a sign of biological activity going on below the surface.[2]
- Jupiter – the atmosphere contains about 0.3% methane
- Saturn – the atmosphere contains about 0.4% methane
- Iapetus
- Titan — the atmosphere contains 1.6% methane and thousands of methane lakes have been detected on the surface[3] In the upper atmosphere the methane is converted into more complex molecules including acetylene, a process that also produces molecular hydrogen. There is evidence that acetylene and hydrogen are recycled into methane near the surface. This suggests the presence either of an exotic catalyst, or an unfamiliar form of methanogenic life.[4]
- Enceladus – the atmosphere contains 1.7% methane[5]
- Uranus – the atmosphere contains 2.3% methane
- Ariel – methane is believed to be a constituent of Ariel's surface ice
- Miranda
- Oberon – about 20% of Oberon's surface ice is composed of methane-related carbon/nitrogen compounds
- Titania – about 20% of Titania's surface ice is composed of methane-related organic compounds
- Umbriel – methane is a constituent of Umbriel's surface ice
- Neptune – the atmosphere contains 1.6% methane
- Pluto – spectroscopic analysis of Pluto's surface reveals it to contain traces of methane[8][9]
- Eris – infrared light from the object revealed the presence of methane ice
- Comet Halley
- Comet Hyakutake – terrestrial observations found ethane and methane in the comet[11]
- Extrasolar planet HD 189733b – This is the first detection of an organic compound on a planet outside the solar system. Its origin is unknown, since the planet's high temperature (700 °C) would normally favor the formation of carbon monoxide instead.[12]
- Interstellar clouds[13]
References
- ^ Stern, S.A. (1999). "The Lunar atmosphere: History, status, current problems, and context". Rev. Geophys. 37 (4): 453–491. Bibcode 1999RvGeo..37..453S. doi:10.1029/1999RG900005.
- ^ Mars Vents Methane in What Could Be Sign of Life, Washington Post, January 16, 2009
- ^ Niemann, HB; Atreya, SK; Bauer, SJ; Carignan, GR; Demick, JE; Frost, RL; Gautier, D; Haberman, JA et al. (2005). "The abundances of constituents of Titan’s atmosphere from the GCMS instrument on the Huygens probe". Nature 438 (7069): 779–784. Bibcode 2005Natur.438..779N. doi:10.1038/nature04122. PMID 16319830.
- ^ Chris Mckay (2010). "Have We Discovered Evidence For Life On Titan". SpaceDaily. http://www.spacedaily.com/reports/Have_We_Discovered_Evidence_For_Life_On_Titan_999.html. Retrieved 2010-06-10. Space.com. March 23, 2010.
- ^ Waite, J. H.; et al.; (2006); Cassini Ion and Neutral Mass Spectrometer: Enceladus Plume Composition and Structure, Science, Vol. 311, No. 5766, pp. 1419–1422
- ^ Shemansky, DF; Yelle, RV; Linick; Lunine (December 15, 1989). "Ultraviolet Spectrometer Observations of Neptune and Triton". Science 246 (4936): 1459–1466. Bibcode 1989Sci...246.1459B. doi:10.1126/science.246.4936.1459. PMID 17756000.
- ^ Ron Miller; William K. Hartmann (2005). The Grand Tour: A Traveler's Guide to the Solar System (3rd ed.). Thailand: Workman Publishing. pp. 172–73. ISBN 0-7611-3547-2.
- ^ Tobias C. Owen, Ted L. Roush et al. (6 August 1993). "Surface Ices and the Atmospheric Composition of Pluto". Science 261 (5122): 745–748. Bibcode 1993Sci...261..745O. doi:10.1126/science.261.5122.745. PMID 17757212. http://www.sciencemag.org/cgi/content/abstract/261/5122/745. Retrieved 2007-03-29.
- ^ "Pluto". SolStation. 2006. http://www.solstation.com/stars/pluto.htm. Retrieved 2007-03-28.
- ^ Sicardy, B; Bellucci, A; Gendron, E; Lacombe, F; Lacour, S; Lecacheux, J; Lellouch, E; Renner, S et al. (2006). "Charon’s size and an upper limit on its atmosphere from a stellar occultation". Nature 439 (7072): 52–4. Bibcode 2006Natur.439...52S. doi:10.1038/nature04351. PMID 16397493. http://www.nature.com/nature/journal/v439/n7072/abs/nature04351.html.
- ^ Mumma, M.J.; Disanti, M.A., dello Russo, N., Fomenkova, M., Magee-Sauer, K., Kaminski, C.D., and D.X. Xie (1996). "Detection of Abundant Ethane and Methane, Along with Carbon Monoxide and Water, in Comet C/1996 B2 Hyakutake: Evidence for Interstellar Origin". Science 272 (5266): 1310–4. Bibcode 1996Sci...272.1310M. doi:10.1126/science.272.5266.1310. PMID 8650540.
- ^ Stephen Battersby (2008-02-11). "Organic molecules found on alien world for first time". http://space.newscientist.com/article/dn13303-organic-molecules-found-on-alien-world-for-first-time.html. Retrieved 2008-02-12.
- ^ J. H. Lacy, J. S. Carr, N. J. Evans, II, F. Baas, J. M. Achtermann, J. F. Arens (1991). "Discovery of interstellar methane — Observations of gaseous and solid CH4 absorption toward young stars in molecular clouds". Astrophysical Journal 376: 556–560. Bibcode 1991ApJ...376..556L. doi:10.1086/170304.
See also
- 2007 Zasyadko mine disaster
- Abiogenic petroleum origin
- Aerobic methane production
- Anaerobic digestion
- Anaerobic respiration
- Arctic methane release
- Biogas
- Coal Oil Point seep field
- Energy density
- Greenhouse gas
- Halomethane, halogenated methane derivatives
- List of alkanes
- Methanation
- Methane clathrate, form of water ice that contains methane
- Methanogen, archaea that produce methane as a metabolic by-product
- Methanogenesis, the formation of methane by microbes
- Methanotroph, bacteria that are able to grow using methane as their only source of carbon and energy
- Methyl group, a functional group similar to methane
- Organic gas
- Thomas Gold
External links
- Gavin Schmidt, Methane: A Scientific Journey from Obscurity to Climate Super-Stardom, NASA Goddard, September 2004
- Methane thermodynamics
- International Chemical Safety Card 0291
- Methane Hydrates
- Safety data for methane
- Methane-eating bug holds promise for cutting greenhouse gas. Media Release, GNS Science, New Zealand]
- Catalytic conversion of methane to more useful chemicals and fuels
- Methane as a Savior of the Dairy Industry
Alkanes Higher alkanes · List of alkanes Categories:- Methane
- Anaerobic digestion
- Greenhouse gases
- Fuels
- Fuel gas
Wikimedia Foundation. 2010.

