- Rifaximin
-
Rifaximin 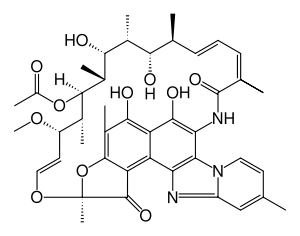
Systematic (IUPAC) name (2S,16Z,18E,20S,21S,22R,23R,24R,25S,26S,27S,28E)-5,6,21,23,25-pentahydroxy-27-methoxy-2,4,11,16,20,22,24,26-octamethyl-2,7-(epoxypentadeca-[1,11,13]trienimino)benzofuro
[4,5-e]pyrido[1,2-a]-benzimida-zole-1,15(2H)-dione,25-acetateClinical data Trade names Xifaxan AHFS/Drugs.com monograph MedlinePlus a604027 Pregnancy cat. C(US) Legal status ℞ Prescription only Routes Oral Pharmacokinetic data Bioavailability < 0.4% Metabolism Hepatic Half-life 6 hours Excretion Fecal (97%) Identifiers CAS number 80621-81-4 
ATC code A07AA11 D06AX11 QG51AA06 QJ51XX01 PubChem CID 6436173 DrugBank APRD01218 ChemSpider 10482302 
UNII L36O5T016N 
KEGG D02554 
ChEMBL CHEMBL1617 
Chemical data Formula C43H51N3O11 Mol. mass 785.879 g/mol SMILES eMolecules & PubChem - InChI=1S/C43H51N3O11/c1-19-14-16-46-28(18-19)44-32-29-30-37(50)25(7)40-31(29)41(52)43(9,57-40)55-17-15-27(54-10)22(4)39(56-26(8)47)24(6)36(49)23(5)35(48)20(2)12-11-13-21(3)42(53)45-33(34(32)46)38(30)51/h11-18,20,22-24,27,35-36,39,48-51H,1-10H3,(H,45,53)/b12-11+,17-15+,21-13-/t20-,22+,23+,24+,27-,35-,36+,39+,43-/m0/s1

Key:NZCRJKRKKOLAOJ-XRCRFVBUSA-N
 (what is this?) (verify)
(what is this?) (verify)Rifaximin is a semisynthetic, rifamycin-based non-systemic antibiotic, meaning that very little of the drug will pass the gastrointestinal wall into the circulation as is common for other types of orally administered antibiotics. It is used in the treatment of traveler's diarrhea and hepatic encephalopathy, for which it received orphan drug status from the U.S. Food and Drug Administration in 1998.
Contents
Uses
Rifaximin is licensed by the U.S. Food and Drug Administration to treat traveler's diarrhea caused by E. coli.[1] Clinical trials have shown that rifaximin is highly effective at preventing and treating traveler's diarrhea among travelers to Mexico, with few side effects and low risk of developing antibiotic resistance.[2] [3] It is not effective against Campylobacter jejuni, and there is no evidence of efficacy against Shigella or Salmonella species.
It may be efficacious in relieving chronic functional symptoms of bloating and flatulence that are common in irritable bowel syndrome.[4] There was recently a pilot-study done on the efficacy of rifaximin as a means of treatment for rosacea, according to the study, induced by the co-presence of small intestinal bacterial overgrowth.[5]
In the United States, rifaximin has orphan drug status for the treatment of hepatic encephalopathy.[6] Although high-quality evidence is still lacking, rifaximin appears to be as effective as or more effective than other available treatments for hepatic encephalopathy (such as lactulose), is better tolerated, and may work faster.[7] The drawbacks to rifaximin are increased cost and lack of robust clinical trials for HE without combination lactulose therapy.
A recent study suggests that treatment with rifaximin relieves symptoms for some sufferers of irritable bowel syndrome. [8]
Availability
Rifaximin is currently sold in the U.S. under the brand name Xifaxan by Salix Pharmaceuticals. It is also sold in Europe under the names Spiraxin, Zaxine, Normix, Rifacol and Colidur, and in India under the name Rixmin, Rcifax, Rifagut and Torfix.
References
- ^ Xifaxan label informationPDF Retrieved November 15, 2008.
- ^ DuPont, H (2007). "Therapy for and Prevention of Traveler's Diarrhea". Clinical Infectious Diseases 45 (45 (Suppl 1)): S78–S84. doi:10.1086/518155. PMID 17582576.
- ^ Ruiz J, Mensa L, Pons MJ, Vila J, Gascon J (May 2008). "Development of Escherichia coli rifaximin-resistant mutants: frequency of selection and stability". The Journal of antimicrobial chemotherapy 61 (5): 1016–9. doi:10.1093/jac/dkn078. PMID 18325895. http://jac.oxfordjournals.org/cgi/pmidlookup?view=long&pmid=18325895.
- ^ Sharara A, Aoun E, Abdul-Baki H, Mounzer R, Sidani S, ElHajj I. (2006). "A randomized double-blind placebo-controlled trial of rifaximin in patients with abdominal bloating and flatulence". Am J Gastroenterol 101 (2): 326–33. doi:10.1111/j.1572-0241.2006.00458.x. PMID 16454838.
- ^ Small intestinal bacterial overgrowth in rosacea: clinical effectiveness of its eradication. Parodi A, Paolino S, Greco A, Drago F, Mansi C, Rebora A, Parodi A, Savarino V.
- ^ Wolf, David C. (2007-01-09). "Hepatic Encephalopathy". eMedicine. WebMD. http://www.emedicine.com/med/TOPIC3185.HTM. Retrieved 2007-02-15.
- ^ Lawrence KR, Klee JA (2008). "Rifaximin for the treatment of hepatic encephalopathy". Pharmacotherapy 28 (8): 1019–32. doi:10.1592/phco.28.8.1019. PMID 18657018. Free full text with registration at Medscape.
- ^ Antibiotic May Help Ease Irritable Bowel, Businessweek, January 05, 2011
External links
- Micromedex information on rifaximin
- FDA label approved for Xifaxan (PDF warning)
- Xifaxan (manufacturer's website)
Antibiotics and chemotherapeutics for dermatological use (D06) Antibiotics OthersAminoglycosides: Neomycin • Gentamicin • Amikacin
Rifamycin: Rifaximin
other: Fusidic acid • Bacitracin • Tyrothricin • MupirocinChemotherapeutics Aciclovir • Penciclovir • Idoxuridine • Edoxudine
Imiquimod/Resiquimod • Podophyllotoxin
Docosanol • Tromantadine • Inosine • Lysozyme • IbacitabineOtherAntibacterials: nucleic acid inhibitors (J01E, J01M) Antifolates
(inhibits
purine metabolism,
thereby inhibiting
DNA and RNA synthesis)Other/ungroupedCombinationsTopoisomerase
inhibitors/
quinolones/
(inhibits
DNA replication)1st g.2nd g.Ciprofloxacin# • Enoxacin‡ • Fleroxacin‡ • Lomefloxacin • Nadifloxacin • Ofloxacin • Norfloxacin • Pefloxacin • Rufloxacin3rd g.4th g.Besifloxacin • Clinafloxacin† • Garenoxacin • Gemifloxacin • Moxifloxacin • Gatifloxacin‡ • Sitafloxacin • Trovafloxacin‡/Alatrofloxacin‡ • PrulifloxacinVet.Related (DG)Anaerobic DNA
inhibitorsNitrofuran derivativesRNA synthesis 
This systemic antibacterial-related article is a stub. You can help Wikipedia by expanding it. - InChI=1S/C43H51N3O11/c1-19-14-16-46-28(18-19)44-32-29-30-37(50)25(7)40-31(29)41(52)43(9,57-40)55-17-15-27(54-10)22(4)39(56-26(8)47)24(6)36(49)23(5)35(48)20(2)12-11-13-21(3)42(53)45-33(34(32)46)38(30)51/h11-18,20,22-24,27,35-36,39,48-51H,1-10H3,(H,45,53)/b12-11+,17-15+,21-13-/t20-,22+,23+,24+,27-,35-,36+,39+,43-/m0/s1
