- Mafenide
-
Mafenide 
Systematic (IUPAC) name 4-(aminomethyl)benzenesulfonamide Clinical data AHFS/Drugs.com monograph Pregnancy cat. ? Legal status ? Routes Topical Identifiers CAS number 138-39-6 
ATC code D06BA03 PubChem CID 3998 ChemSpider 3858 
UNII 58447S8P4L 
KEGG D02351 
ChEMBL CHEMBL419 
Chemical data Formula C7H10N2O2S Mol. mass 186.233 g/mol SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Mafenide (INN; usually as mafenide acetate, trade name Sulfamylon) is a sulfonamide.
Contents
Uses
It is used to treat severe burns.[1][2]
Mafenide (Sulfamylon) can interfere with the kidney's role in hydrogen ion excretion, resulting in metabolic acidosis.
Mafenide is a topical sulfonamide used as adjunctive therapy of second- and third-degree burns. It is bacteriostatic against many gram-positive and gram-negative organisms including Pseudomonas aeruginosa. Mafenide is metabolized to a carbonic anhydrase inhibitor, which could result in metabolic acidosis. This drug was approved by the FDA in 1948. Some sources state that mafenide is more appropriate for non-facial burns, while chloramphenicol/prednisolone or bacitracin are more appropriate for facial burns.[3]Mechanism of Action
Mechanism of Action: Mafenide works by reducing the bacterial population present in the avascular tissues of burns and permits spontaneous healing of deep partial-thickness burns.
Contraindications/Precautions:Sulfonamide hypersensitivity; renal impairment.Adverse Reactions
Superinfection, pain or burning upon application, rash, pruritus, tachypnea, hyperventilation, metabolic acidosis.
Drug interaction
Drug Interactions: No significant interactions
Dosage:
For use adjunctive therapy of second- and third-degree burns to prevent infection: Topical dosage: Adults and children: Apply topically to a thickness of approximately 1.6 mm, to cleaned and debrided wound once or twice per day with a sterile gloved hand. Burned area should be covered with cream at all times.
See also
References
- ^ Siuda J. F., Cihonski C. D. (1972). "New compounds: carbamate derivatives of mafenide (homosulfanilamide)". J. Pharm. Sci. 61 (11): 1856–1857. doi:10.1002/jps.2600611143. PMID 4652670.
- ^ Haynes B. (1971). "Mafenide acetate in Burn Treatment". New England J. Medic. 284 (23): 1324. doi:10.1056/NEJM197106102842310. PMID 5576444.
- ^ Haik J, Ashkenazy O, Sinai S, et al. (November 2005). "Burn care standards in Israel: lack of consensus". Burns 31 (7): 845–9. doi:10.1016/j.burns.2005.04.012. PMID 15967581. http://linkinghub.elsevier.com/retrieve/pii/S0305-4179(05)00130-0.
External links
Antibiotics and chemotherapeutics for dermatological use (D06) Antibiotics OthersChemotherapeutics Aciclovir • Penciclovir • Idoxuridine • Edoxudine
Imiquimod/Resiquimod • Podophyllotoxin
Docosanol • Tromantadine • Inosine • Lysozyme • IbacitabineOtherAntibacterials: nucleic acid inhibitors (J01E, J01M) Antifolates
(inhibits
purine metabolism,
thereby inhibiting
DNA and RNA synthesis)Other/ungroupedCombinationsTopoisomerase
inhibitors/
quinolones/
(inhibits
DNA replication)1st g.2nd g.Ciprofloxacin# • Enoxacin‡ • Fleroxacin‡ • Lomefloxacin • Nadifloxacin • Ofloxacin • Norfloxacin • Pefloxacin • Rufloxacin3rd g.4th g.Besifloxacin • Clinafloxacin† • Garenoxacin • Gemifloxacin • Moxifloxacin • Gatifloxacin‡ • Sitafloxacin • Trovafloxacin‡/Alatrofloxacin‡ • PrulifloxacinVet.Related (DG)Anaerobic DNA
inhibitorsNitrofuran derivativesRNA synthesis #WHO-EM. ‡Withdrawn from market. Clinical trials: †Phase III. §Never to phase III 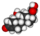
This dermatologic drug article is a stub. You can help Wikipedia by expanding it.
