- Nystatin
-
Nystatin 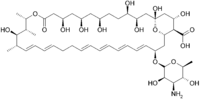
Systematic (IUPAC) name (1S,3R,4R,7R,9R,11R,15S,16R,17R,18S,19E,21E, 25E,27E,29E,31E,33R,35S,36R,37S)-33-[(3-amino-3, Clinical data Trade names Mycostatin, Nilstat AHFS/Drugs.com monograph MedlinePlus a682758 Pregnancy cat. ? Legal status ? Routes topical & oral (but not absorbed) Pharmacokinetic data Bioavailability 0% on oral ingestion Metabolism ? Half-life ? Excretion ? Identifiers CAS number 1400-61-9 
ATC code A07AA02 D01AA01 G01AA01 PubChem CID 14960 DrugBank APRD01146 ChemSpider 10468627 
UNII BDF1O1C72E 
KEGG D00202 
ChEBI CHEBI:473992 
ChEMBL CHEMBL229383 
Chemical data Formula C47H75NO17 Mol. mass 926.09 SMILES eMolecules & PubChem - InChI=1S/C47H75NO17/c1-27-17-15-13-11-9-7-5-6-8-10-12-14-16-18-34(64-46-44(58)41(48)43(57)30(4)63-46)24-38-40(45(59)60)37(54)26-47(61,65-38)25-36(53)35(52)20-19-31(49)21-32(50)22-33(51)23-39(55)62-29(3)28(2)42(27)56/h5-6,8,10-18,27-38,40-44,46,49-54,56-58,61H,7,9,19-26,48H2,1-4H3,(H,59,60)/b6-5+,10-8+,13-11+,14-12+,17-15+,18-16+/t27-,28-,29-,30+,31?,32?,33?,34?,35?,36?,37?,38?,40?,41-,42+,43+,44-,46+,47+/m0/s1

Key:VQOXZBDYSJBXMA-RKEBNKJGSA-N
 (what is this?) (verify)
(what is this?) (verify)Nystatin is a polyene antifungal drug to which many molds and yeast infections are sensitive, including Candida. Due to its toxicity profile, there are currently no injectable formulations of this drug on the US market.[1] However, nystatin may be safely given orally as well as applied topically due to its minimal absorption through mucocutaneous membranes such as the gut and the skin.[2][3]
Contents
Uses
Cutaneous, vaginal, mucosal and esophageal Candida infections usually respond well to treatment with nystatin. Cryptococcus is also sensitive to nystatin. In the UK its licence for treating neonatal oral thrush is restricted to those over the age of one month (miconazole is an appropriate alternative for younger babies).[citation needed]
Nystatin is often used as prophylaxis in patients who are at risk for fungal infections, such as AIDS patients with a low CD4+ count and patients receiving chemotherapy.
It is prescribed in units, with doses varying from 100,000 (for oral infections) to 1 million (for intestinal ones). As it is not absorbed from the gut, it is safe for oral use and does not have problems of drug interactions.
It is also used in cellular biology as an inhibitor of the lipid raft-caveolae endocytosis pathway on mammalian cells, at concentrations around 3 µg/mL.
 Penicillium-infested tangerine. The spot absent of growth had nystatin applied to it before the fungus covered the fruit.
Penicillium-infested tangerine. The spot absent of growth had nystatin applied to it before the fungus covered the fruit.
In certain cases, nystatin has been used to prevent the spread of mold on objects such as works of art. For example, it was applied to wood panel paintings damaged as a result of the Arno River Flood of 1966 in Florence, Italy.
Nystatin is also used as a tool by scientists performing "perforated" patch-clamp electrophysiologic recordings of cells. When loaded in the recording pipette, it allows for measurement of electrical currents without washing out the intracellular contents, because it forms pores in the cell membrane that are permeable to only monovalent ions.[4]
Mechanism of action
Like amphotericin B and natamycin, nystatin binds to ergosterol, a major component of the fungal cell membrane. When present in sufficient concentrations, it forms pores in the membrane that lead to K+ leakage and death of the fungus. Ergosterol is fairly unique to fungi, so the drug does not have such catastrophic effects on animals.
Origin
Like many other antifungals and antibiotics, nystatin is of bacterial origin. It was isolated from Streptomyces noursei in 1950 by Elizabeth Lee Hazen and Rachel Fuller Brown, who were doing research for the Division of Laboratories and Research of the New York State Department of Health. Hazen found a promising micro-organism in the soil of a friend's dairy farm. She named it Streptomyces noursei, after William Nourse, the farm's owner.[5] Hazen and Brown named nystatin after the New York State Public Health Department (now known as the Wadsworth Center) in 1954.
Brand names
- Nystan (oral tablets, topical ointment, and pessaries, formerly from Bristol-Myers Squibb)
- Infestat
- Nystalocal from Medinova AG
- Nystamont
- Nystop (topical powder, Paddock)
- Nystex
- Mykinac
- Nysert (vaginal suppositories, Procter & Gamble)
- Nystaform (topical cream, and ointment and cream combined with iodochlorhydroxyquine and hydrocortisone; formerly Bayer now Typharm Ltd)
- Nilstat (vaginal tablet, oral drops, Lederle)
- Korostatin (vaginal tablets, Holland Rantos)
- Mycostatin (vaginal tablets, topical powder, Bristol-Myers Squibb)
- Mycolog-II (topical ointment, combined with triamcinolone; Apothecon)
- Mytrex (topical ointment, combined with triamcinolone)
- Mykacet (topical ointment, combined with triamcinolone)
- Myco-Triacet II (topical ointment, combined with triamcinolone)
- Flagystatin II (cream, combined with metronidazole)
- Nistatina (oral tablets, Antibiotice Iaşi)
- Nidoflor (cream, combined with neomycin sulfate and triamcinolone acetonide, Antibiotice Iaşi)
- Stamicin (oral tablets, Antibiotice Iaşi) see http://www.farmaline.ro/index.php?cid=1&pid=357
- Lystin
- Animax (veterinary topical ointment or cream; combined with neomycin sulfate, thiostrepton and triamcinolone acetonide)
References
- ^ Nystatin on RxList.com http://www.rxlist.com/script/main/srchcont_rxlist.asp?src=nystatin
- ^ Nystatin on Micromedex http://www.thomsonhc.com/hcs/librarian/ND_T/HCS/ND_PR/Main/CS/BCFBA8/DUPLICATIONSHIELDSYNC/6B2A9E/ND_PG/PRIH/ND_B/HCS/SBK/3/ND_P/Main/PFActionId/hcs.common.RetrieveDocumentCommon/DocId/0105/ContentSetId/31#pharmacokineticsSection
- ^ Nystatin on UpToDate http://www.utdol.com/online/content/topic.do?topicKey=drug_l_z/184320&selectedTitle=1%7E44&source=search_result#F202808
- ^ Akaike N, Harata N (1994). "Nystatin perforated patch recording and its applications to analyses of intracellular mechanisms" ([dead link]). Jpn. J. Physiol. 44 (5): 433–73. doi:10.2170/jjphysiol.44.433. PMID 7534361. http://joi.jlc.jst.go.jp/JST.JSTAGE/jjphysiol/44.433?from=PubMed.
- ^ Ana Espinel-Ingroff, Medical mycology in the United States: a historical analysis (1894-1996), Springer, 2003, p. 62.
Antifungals (D01 and J02) Wall/
membraneErgosterol
inhibitorstopical: Bifonazole, Clomidazole, Clotrimazole#, Croconazole, Econazole, Fenticonazole, Ketoconazole, Isoconazole, Miconazole#, Neticonazole, Oxiconazole, Sertaconazole, Sulconazole, Tioconazoletopical: (Fluconazole#, Fosfluconazole)
systemic: (Fluconazole, Hexaconazole, Itraconazole, Posaconazole, Voriconazole)topical: Thiabendazoleβ-glucan synthase
inhibitorsIntracellular Pyrimidine analogues/
Thymidylate synthase inhibitorsOthers Bromochlorosalicylanilide • Methylrosaniline • Tribromometacresol • Undecylenic acid • Polynoxylin • Chlorophetanol • Chlorphenesin • Ticlatone • Sulbentine • Ethyl hydroxybenzoate • Haloprogin • Salicylic acid • Selenium sulfide# • Ciclopirox • Amorolfine • Dimazole • Tolnaftate • Tolciclate • Sodium thiosulfate# • Whitfield's ointment# • Potassium iodide#
Tea tree oil • citronella oil • lemon grass • orange oil • patchouli • lemon myrtle
PCP: Pentamidine • Dapsone • AtovaquoneGynecological anti-infectives and antiseptics (G01) Antibiotics polyene antimycotic (Nystatin, Natamycin, Amphotericin B) • Candicidin • Chloramphenicol • Hachimycin • Oxytetracycline • Carfecillin • Mepartricin • Clindamycin • PentamycinArsenic compounds Quinoline derivatives Organic acids Sulfonamides SulfatolamideImidazole derivatives Triazole derivatives Other Clodantoin • Inosine • Policresulen • Nifuratel • Furazolidone • Methylrosaniline • Povidone-iodine • Ciclopirox • Protiofate • Lactobacillus fermentum • Copper usnateCategories:- Antifungals
- World Health Organization essential medicines
- InChI=1S/C47H75NO17/c1-27-17-15-13-11-9-7-5-6-8-10-12-14-16-18-34(64-46-44(58)41(48)43(57)30(4)63-46)24-38-40(45(59)60)37(54)26-47(61,65-38)25-36(53)35(52)20-19-31(49)21-32(50)22-33(51)23-39(55)62-29(3)28(2)42(27)56/h5-6,8,10-18,27-38,40-44,46,49-54,56-58,61H,7,9,19-26,48H2,1-4H3,(H,59,60)/b6-5+,10-8+,13-11+,14-12+,17-15+,18-16+/t27-,28-,29-,30+,31?,32?,33?,34?,35?,36?,37?,38?,40?,41-,42+,43+,44-,46+,47+/m0/s1
Wikimedia Foundation. 2010.
