- Striatum
-
Brain: Striatum 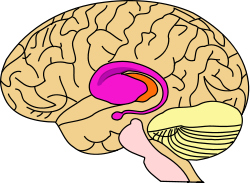
purple=caudate and putamen, orange=thalamus 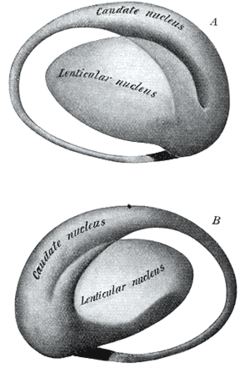
Two views of a model of the striatum. A: lateral aspect, B: medial aspect. (The lenticular nucleus is an alternative name for the putamen and globus pallidus.) NeuroNames hier-207 The striatum, also known as the neostriatum or striate nucleus, is a subcortical (i.e., inside, rather than on the outside) part of the forebrain. It is the major input station of the basal ganglia system. The striatum, in turn, gets input from the cerebral cortex. In primates (including humans), the striatum is divided by a white matter tract called the internal capsule into two sectors called the caudate nucleus and putamen. The term corpus striatum sometimes refers to the striatum combined with the globus pallidus, a structure closely related to the putamen.[1]
Contents
History
In the seventeenth and eighteenth centuries, the term "corpus striatum" was used to designate many distinct, deep, infracortical elements of the hemisphere.[2] In 1941, Cécile and Oskar Vogt simplified the nomenclature by proposing the term striatum for all elements built with striatal elements (see primate basal ganglia system): the caudate, the putamen, and the fundus striati, that ventral part linking the two preceding together ventrally to the inferior part of the internal capsule.
The term neostriatum was forged by comparative anatomists comparing the subcortical structures between vertebrates, because it was thought to be a phylogenetically newer section of the corpus striatum. The term is still used by some sources, including Medical Subject Headings.[3]
Cell types
The striatum is heterogeneous in terms of neurons. It is composed of these neuronal cell types:[4]
- Medium spiny neurons so called due to the presence of spines on the dendrites, and making up 96% of the striatum.
- Deiters' neurons (2%) with large, minimally branched arborizations looking like pallidonigral neurons.
- Cholinergic interneurons (1%). In primates, they are the tonically active neurons, and morphologically entirely different from those observed in rodents. These briefly stop firing in response to behaviorally-salient and reward-related events.
- GABAergic parvalbumin expressing interneurons, which are fast-spiking, and express dopamine receptors.
- GABAergic calretinin expressing interneurons.
- GABAergic somatostatin expressing interneurons, which are low-threshold-spiking and express dopamine receptors.
Organization
Anatomical subdivisions
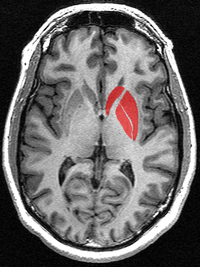 This is a transverse section of the striatum from a structural MR image. The striatum includes the caudate nucleus (red, top) and putamen (right) and the globus pallidus (lower left).
This is a transverse section of the striatum from a structural MR image. The striatum includes the caudate nucleus (red, top) and putamen (right) and the globus pallidus (lower left).
The striatum is spatially organized according to several criteria. It is important to note that the distinction between a dorsal and ventral striatum on the basis of specific cortical, thalamic, and dopaminergic inputs does not provide sharply defined borders between these striatal areas. To date, few structural or functional markers uniquely characteristic for either of the two regions have been identified. In the literature, dorsal and ventral striatum have been virtually equalized with the distinction between the caudate-putamen complex (dorsal) and the nucleus accumbens (ventral), respectively. However, the ventral striatum as defined on the basis of the aforementioned limbic inputs, as well as based on cyto- and chemoarchitectonic characteristics, occupies a more extensive striatal area than the nucleus accumbens alone and extends more dorsally and caudally into the ventral parts of the caudate nucleus and putamen.
The striatum can also be differentiated based on immunochemical characteristics—particularly with regard to acetylcholinesterase—into "compartments", consisting of 'striosomes' and 'matrisomes'.
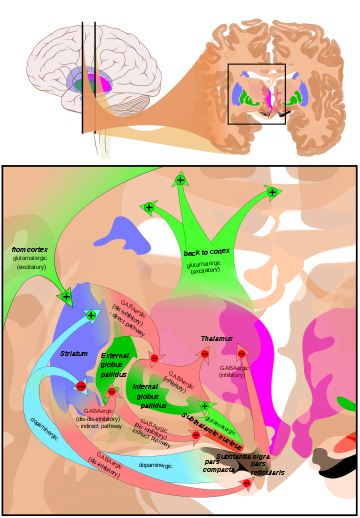 Overview of the main circuits of the basal ganglia. Striatum is shown in blue. Picture shows 2 coronal slices that have been superimposed to include the involved basal ganglia structures. + and - signs at the point of the arrows indicate respectively whether the pathway is excitatory or inhibitory in effect. Green arrows refer to excitatory glutamatergic pathways, red arrows refer to inhibitory GABAergic pathways and turquoise arrows refer to dopaminergic pathways that are excitatory on the direct pathway and inhibitory on the indirect pathway.
Overview of the main circuits of the basal ganglia. Striatum is shown in blue. Picture shows 2 coronal slices that have been superimposed to include the involved basal ganglia structures. + and - signs at the point of the arrows indicate respectively whether the pathway is excitatory or inhibitory in effect. Green arrows refer to excitatory glutamatergic pathways, red arrows refer to inhibitory GABAergic pathways and turquoise arrows refer to dopaminergic pathways that are excitatory on the direct pathway and inhibitory on the indirect pathway.
Dorsal striatum
The dorsal striatum forms a continuous and large mass, topographically separated by the internal capsule into:
- The caudate (medially)
- The putamen (laterally)
- The fundus (ventrally), linking the caudate nucleus and the putamen.
The dorsal striatum is a single entity closed and continuous.[5] However, the observable anatomical subdivisions of the dorsal striatum (caudate nucleus and putamen) essentially induced by the internal capsule do not completely overlap with now accepted anatomo-functional subdivisions. The selective distribution of the axonal terminal arborisations of cortical sources differentiate the sensorimotor striatum, mainly putaminal but located in its dorsal part and in the lateroinferior part of the caudate. A great part of the remaining of the volume (essentially caudate) receiving from axonal endings from the frontal, parietal, temporal cortex forms the associative striatum. The separation between these two territories is rather clearcut and observable using calbindin immunochemistry. A third entity, the most inferomedial, raises more problems as there is no general agreement about its border with the associative striatum.
Current evidence suggests that the dorsal striatum contributes directly to decision-making, especially to action selection and initiation, through the integration of sensorimotor, cognitive, and motivational/emotional information within specific corticostriatal circuits involving discrete regions of striatum. Studies in humans corroborate the research in animals suggesting that the dorsal striatum is an integral part of a circuit involved in decision-making. Accumulating evidence, primarily from neuroimaging but also neuropsychological investigations, has implicated the dorsal striatum in different aspects of motivational and learning processes that support goal-directed action. For instance, positron emission tomography (PET) studies report increases in dopamine release in the dorsal striatum (as measured by displacement of endogenous dopamine by radioligands) when participants are presented with potential rewards, such as the opportunity to gain money (Koepp et al., 1998; Zald et al., 2004) or even when presented with food stimuli while in a state of hunger (Volkow et al., 2002). Similarly, fMRI studies typically report increases in blood oxygenation level dependent (BOLD) responses in the dorsal striatum during anticipation of either primary (O'Doherty et al., 2002) or secondary (Knutson et al., 2001) rewards, much like the ventral striatum.
Ventral striatum
The ventral striatum is strongly associated with emotional and motivational aspects of behavior.
- The nucleus accumbens
- The olfactory tubercle
The ventral striatum is clearly delineated by tracing the subicular (subiculum) territory. This corresponds to the olfactory tubercle and the nucleus accumbens, which is not a nucleus, but is a striatal part made up of striatal elements. The ventral striatum is generally associated with limbic structures, such as the amygdala, hippocampus, midline thalamus, and certain regions of the prefrontal cortex. In addition, the ventral striatum is strongly innervated by dopaminergic fibers from the ventral tegmental area (VTA), known as the mesolimbic dopamine system, and has the highest density of serotonergic inputs in the striatum.
In its present connotation, the term “ventral striatum” was introduced in 1975 by Heimer and Wilson to differentiate it from the dorsal, sensorimotor-related part of the striatum (i.e., the caudate-putamen complex). This inclusion of ventrally located striatal tissue in a “unified” striatum, along with the recognition of connectionally associated pallidal elements in the substantia innominata and deep layers of the olfactory tubercle (i.e., the ventral pallidum), has had great impact on the functional-anatomical concept of the basal ganglia. Whereas traditionally, the basal ganglia were thought to be primarily involved in sensory-motor functions, it has now become accepted that the basal ganglia, as a result of their involvement in a set of parallel, functionally segregated basal ganglia-thalamocortical circuits, which primarily entertain the premotor and prefrontal cortical cortices, are also involved in cognitive and “limbic” functions.
Thus, in line with the characteristics of its inputs, the ventral striatum is functionally strongly associated with emotional and motivational aspects of behavior. Moreover, structural and functional disturbances of ventral striatal areas have been shown to be correlated with various forms of psychopathology, such as schizophrenia, addictive behavior, and obsessive-compulsive disorder.
Inputs (afferent connections)
The most important afferent in terms of quantity of axons is the corticostriatal connection. Many parts of the neocortex innervate the dorsal striatum. The cortical pyramidal neurons projecting to the striatum are located in layers II-VI, but the most dense projections come from layer V.[6] They end mainly on the spines of the spiny neurons. They are glutamatergic, exciting striatal neurons. Another well known afferent is the nigrostriatal connection arising from the neurons of the substantia nigra pars compacta. While cortical axons synapse mainly on spine heads of spiny neurons, nigral axons synapse mainly on spine shafts. In primates, the thalamostriatal afferent essentially comes from the center median-parafascicular complex of the thalamus (see primate basal ganglia system). This afferent is glutamatergic. The participation of truly intralaminar neurons is much more limited. The striatum also receives afferents from other elements of the basal ganglia such as the subthalamic nucleus (glutamatergic) or the external globus pallidus (GABAergic).
Targets (efferent connections)
The basal ganglia core is made up of the striatum along with the regions to which it projects directly, via the striato-pallidonigral bundle. The striato-pallidonigral bundle is a very dense bundle of sparsely myelinated axons, giving a whitish appearance. This projection comprises successively the external globus pallidus (GPe), the internal globus pallidus (GPi), the pars compacta of the substantia nigra (SNc) and the pars reticulata of substantia nigra (SNr). The neurons of this projection are inhibited by GABAergic synapses from the dorsal striatum. Among these targets, the GPe does not send axons outside the system. Others send axons to the superior colliculus. Two others comprise the output to the thalamus, forming two separate channels: one through the internal segment of the globus pallidus to the ventral oralis nuclei of the thalamus and from there to the cortical supplementary motor area (SMA) and another through the substantia nigra to the ventral anterior nuclei of the thalamus and from there to the frontal cortex and the occulomotor cortex.
Function
Metabotropic dopamine receptors are present both on spiny neurons and on cortical axon terminals. Second messenger cascades triggered by activation of these dopamine receptors can modulate pre- and postsynaptic function, both in the short term and in the long term. The striatum is best known for its role in the planning and modulation of movement pathways but is also involved in a variety of other cognitive processes involving executive function. In humans the striatum is activated by stimuli associated with reward, but also by aversive, novel, unexpected or intense stimuli, and cues associated with such events. Recent fMRI evidence[citation needed] suggests that the common property linking these stimuli, to which the striatum is reacting, is saliency under the conditions of presentation. A number of other brain areas and circuits are also related to reward such as frontal areas. Research at the Wellcome Trust Centre for Neuroimaging at UCL (University College London) shows it to be associated with novelty-related decision making behaviors.
For sources regarding saliency of the reward pathway (thought to be related to dopamine) one can look to the work of Dr. John D. Salamone (early to late 1990s) and Wolfram Schultz.[7] The ventral tegmental dopaminergic neurons that innervate portions of the striatum have long been accepted to be the site of rewarding feeling. Intracranial stimulation studies from the 1960s show implants in this brain area will elicit bar pressing from rats for many hours at a time. However the collective works of researchers in the 1990s show that blocking dopamine receptors does not remove rewarding sensations, rather it affects how much the animal is willing to work, triggering more motivation to seek eventual reward rather than immediate reward.
Pathology
Parkinson's disease results in loss of dopaminergic innervation to the striatum (and other basal ganglia) and a cascade of subsequent consequences. Atrophy of the striatum is also involved in Huntington's disease, choreas, choreoathetosis, and dyskinesias.[8] It is also thought that addiction involves plasticity at striatal synapses.
See also
- Striatopallidal fibres
- List of regions in the human brain
References
- ^ "DBS: The Basal Ganglia". http://biomed.brown.edu/Courses/BI108/BI108_2003_Groups/Deep_Brain_Stimulation/basalganglia.html.
- ^ Raymond Vieussens, 1685
- ^ MeSH Neostriatum
- ^ Yelnik J, Francois C, Percheron G. Spatial relationships between striatal axonal endings and pallidal neurons in macaque monkeys. Adv Neurol. 1997;74:45-56. PMID 9348401.
- ^ Pieter Voorn, Louk J. M. J. Vanderschuren, Henk J. Groenewegen, Trevor W. Robbins and Cyriel M. A. Pennartz, Putting a spin on the dorsal-ventral divide of the striatum, Trends in Neurosciences, Volume 27, Issue 8, 1 August 2004, Pages 468-474.
- ^ Rosell A, Giménez-Amaya JM (1999). "Anatomical re-evaluation of the corticostriatal projections to the caudate nucleus: a retrograde labeling study in the cat". Neurosci Res 34 (4): 257–69. doi:10.1016/S0168-0102(99)00060-7. PMID 10576548.
- ^ "Department of Physiology, Development and Neuroscience: About the Department". http://www.pdn.cam.ac.uk/staff/schultz/.
- ^ Walker FO (January 2007). "Huntington's disease". Lancet 369 (9557): 218–28. doi:10.1016/S0140-6736(07)60111-1. PMID 17240289.
External links
Human brain, cerebrum, Interior of the cerebral hemispheres—Rostral Basal ganglia and associated structures (TA A14.1.09.321–552, GA 9.832–837) Basal ganglia Ventral striatumOtherInternal capsule (Anterior limb · Genu · Posterior limb, Optic radiation)
Corona radiata · External capsule · Extreme capsule
Pallidothalamic tracts: Thalamic fasciculus (Ansa lenticularis, Lenticular fasciculus) · Subthalamic fasciculusRhinencephalon Other basal forebrain Diagonal band of Broca · Stria terminalisArchicortex:
Hippocampal formation/
Hippocampus anatomyAnxiety disorder: Obsessive–compulsive disorder (F42, 300.3) History Yale–Brown Obsessive Compulsive ScaleBiology NeuroanatomyReceptorsSymptoms Obsessions (associative, diagnostic, injurious, scrupulous, pathogenic, sexual) · Compulsions (impulses, rituals, tics) · Thought suppression (avoidance) · Hoarding (animals, books, possessions)Treatment Mu opioidergicsNMDA glutamatergicsNK-1 tachykininergicsOtherBehavioralOrganizations Notable people Edna B. Foa · Stanley Rachman · Adam S. Radomsky · Jeffrey M. Schwartz · Susan Swedo · Jeff Bell · Emily ColasPopular culture LiteratureFictionalNonfictionEverything in Its Place · Just CheckingMediaRelated Obsessive–compulsive personality disorder · Obsessional jealousy · Purely Obsessional OCD · Social anxiety disorder · Tourette syndromeCategories:
Wikimedia Foundation. 2010.
