- Thrombin
-
Thrombin is a "trypsin-like" serine protease protein that in humans is encoded by the F2 gene.[2][3] Prothrombin (coagulation factor II) is proteolytically cleaved to form thrombin in the first step of the coagulation cascade, which ultimately results in the stemming of blood loss. Thrombin in turn acts as a serine protease that converts soluble fibrinogen into insoluble strands of fibrin, as well as catalyzing many other coagulation-related reactions.
Contents
History
After the description of fibrinogen and fibrin, Alexander Schmidt hypothesised the existence of an enzyme that converts fibrinogen into fibrin in 1872.[4]
Physiology
Synthesis
Thrombin is produced by the enzymatic cleavage of two sites on prothrombin by activated Factor X (Xa). The activity of factor Xa is greatly enhanced by binding to activated Factor V (Va), termed the prothrombinase complex. Prothrombin is produced in the liver and is post-translationally modified in a vitamin K-dependent reaction that converts ten glutamic acids on prothrombin to gamma-carboxyglutamic acid (Gla). In the presence of calcium, the Gla residues promote the binding of prothrombin to phospholipid bilayers (see the picture). Deficiency of vitamin K or administration of the anticoagulant warfarin inhibits the production of gamma-carboxyglutamic acid residues, slowing the activation of the coagulation cascade.
In human adults, the normal blood level of antithrombin activity has been measured to be around 1.1 units/mL. Newborn levels of thrombin steadily increase after birth to reach normal adult levels, from a level of around 0.5 units/mL 1 day after birth, to a level of around 0.9 units/mL after 6 months of life.[5]
Mechanism of action
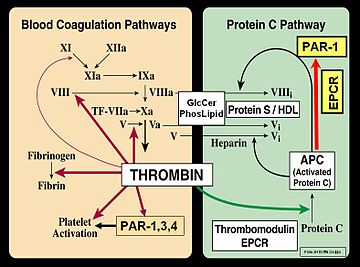
In the blood coagulation pathway, thrombin acts to convert factor XI to XIa, VIII to VIIIa, V to Va, and fibrinogen to fibrin.
Factor XIIIa[clarification needed] is a transglutaminase that catalyzes the formation of covalent bonds between lysine and glutamine residues in fibrin. The covalent bonds increase the stability of the fibrin clot. Thrombin interacts with thrombomodulin.[6][7]
As part of its activity in the coagulation cascade, thrombin also promotes platelet activation and aggregation via activation of protease-activated receptors on the cell membrane of the platelet.
Negative feedback
Thrombin bound to thrombomodulin activates protein C, an inhibitor of the coagulation cascade. The activation of protein C is greatly enhanced following the binding of thrombin to thrombomodulin, an integral membrane protein expressed by endothelial cells. Activated protein C inactivates factors Va and VIIIa. Binding of activated protein C to protein S leads to a modest increase in its activity. Thrombin is also inactivated by antithrombin, a serine protease inhibitor.
Protein
The molecular weight of prothrombin is approximately 72,000 Da. The catalytic domain is released from prothrombin fragment 1.2 to create the active enzyme thrombin, which has a molecular weight of 36,000 Da.
Gene
The Thrombin (prothrombin) gene is located on the eleventh chromosome (11p11-q12).[2]
There are an estimated 30 people in the world that have been diagnosed with the congenital form of Factor II deficiency,[9] which should not be confused with a mutation of prothrombin. The prothrombin gene mutation is called Factor II mutation. Factor II mutation is congenital.[10]
The Factor II mutated gene is not usually accompanied by other factor mutations (i.e., the most common is Factor V Leiden). The gene may be inherited heterozygous (1 pair), or much more rarely, homozygous (2 pairs), and is not related to gender or blood type. Homozygous mutations increase the risk of thrombosis more than heterozygous mutations, but the relative increased risk is not well documented. Other potential risks for thrombosis, such as oral contraceptives may be additive. The previously reported relationship of inflammatory bowel disease (i.e., Crohn's disease or Ulcerative Colitis) and prothrombin mutation or Factor V Leiden mutation have been contradicted by research.[11]
Role in disease
Activation of prothrombin is crucial in physiological and pathological coagulation. Various rare diseases involving prothrombin have been described (e.g., hypoprothrombinemia). Anti-prothrombin antibodies in autoimmune disease may be a factor in the formation of the lupus anticoagulant also known as (antiphospholipid syndrome). Hyperprothrombinemia can be caused by a mutation at 20210a.
Thrombin, a potent vasoconstrictor and mitogen, is implicated as a major factor in vasospasm following subarachnoid hemorrhage. Blood from a ruptured cerebral aneurysm clots around a cerebral artery, releasing thrombin. This can induce an acute and prolonged narrowing of the blood vessel, potentially resulting in cerebral ischemia and infarction (stroke).
Beyond its key role in the dynamic process of thrombus formation, thrombin has a pronounced pro-inflammatory character, which may influence the onset and progression of atherosclerosis. Acting via its specific cell membrane receptors (protease activated receptors: PAR-1, PAR-3 and PAR-4), which are abundantly expressed in all arterial vessel wall constituents, thrombin has the potential to exert pro-atherogenic actions such as inflammation, leukocyte recruitment into the atherosclerotic plaque, enhanced oxidative stress, migration and proliferation of vascular smooth muscle cells, apoptosis and angiogenesis.[12][13][14]
Applications
Research tool
Due to its high proteolytic specificity, thrombin is a valuable biochemical tool. The thrombin cleavage site (Leu-Val-Pro-Arg-Gly-Ser) is commonly included in linker regions of recombinant fusion protein constructs. Following purification of the fusion protein, thrombin can be used to selectively cleave between the Arginine and Glycine residues of the cleavage site, effectively removing the purification tag from the protein of interest with a high degree of specificity.
Medicine and surgery
Prothrombin complex concentrate and fresh frozen plasma are prothrombin-rich coagulation factor preparations that can be used to correct deficiencies (usually due to medication) of prothrombin. Indications include intractable bleeding due to warfarin.
Manipulation of prothrombin is central to the mode of action of most anticoagulants. Warfarin and related drugs inhibit vitamin K-dependent carboxylation of several coagulation factors, including prothrombin. Heparin increases the affinity of antithrombin to thrombin (as well as factor Xa). The direct thrombin inhibitors, a newer class of medication, directly inhibit thrombin by binding to its active site.
Recombinant thrombin is available as a powder for reconstitution into aqueous solution. It can be applied topically during surgery, as an aid to hemostasis. It can be useful for controlling minor bleeding from capillaries and small venules, but ineffective and not indicated for massive or brisk arterial bleeding.[15][16][17] It is marketed by ZymoGenetics in the US under the trade name Recothrom.
Food production
Thrombin is sold under the brand name Fibrimex for use as a binding agent for meat. The thrombin in Fibrimex derives from pigs or bovine blood.[18] According to the manufacturer it can be used to produce new kinds of mixed meats (for example combining beef and fish seamlessly). The manufacturer also states that it can be used to combine scrap meat into whole steaks that can then be sold to a higher price, thus cutting down on production costs.[19]
General secretary Jan Bertoft of Sveriges Konsumenter (The Consumer Coalition of Sweden) has stated that "there is danger of misleading the consumers since there is no way to tell this reconstituted meat from real meat"[18]
See also
- The Proteolysis Map
- Fibrin glue
References
- ^ PDB 2C93; Howard N, Abell C, Blakemore W, Chessari G, Congreve M, Howard S, Jhoti H, Murray CW, Seavers LC, van Montfort RL (February 2006). "Application of fragment screening and fragment linking to the discovery of novel thrombin inhibitors". J. Med. Chem. 49 (4): 1346–55. doi:10.1021/jm050850v. PMID 16480269.
- ^ a b Royle NJ, Irwin DM, Koschinsky ML, MacGillivray RT, Hamerton JL (May 1987). "Human genes encoding prothrombin and ceruloplasmin map to 11p11-q12 and 3q21-24, respectively". Somat. Cell Mol. Genet. 13 (3): 285–92. doi:10.1007/BF01535211. PMID 3474786.
- ^ Degen SJ, Davie EW (September 1987). "Nucleotide sequence of the gene for human prothrombin". Biochemistry 26 (19): 6165–77. doi:10.1021/bi00393a033. PMID 2825773.
- ^ Schmidt A (1872). "Neue Untersuchungen ueber die Fasserstoffesgerinnung". Pflüger's Archiv für die gesamte Physiologie 6: 413–538. doi:10.1007/BF01612263.
- ^ Andrew M, Paes B et al. (1987). "Development of the human coagulation system in the full-term infant". Blood 70 (1): 165–172. PMID 3593964.
- ^ Bajzar L, Morser J, Nesheim M (July 1996). "TAFI, or plasma procarboxypeptidase B, couples the coagulation and fibrinolytic cascades through the thrombin-thrombomodulin complex". J. Biol. Chem. 271 (28): 16603–8. doi:10.1074/jbc.271.28.16603. PMID 8663147.
- ^ Jakubowski HV, Owen WG (July 1989). "Macromolecular specificity determinants on thrombin for fibrinogen and thrombomodulin". J. Biol. Chem. 264 (19): 11117–21. PMID 2544585.
- ^ PDB 1nl2; Huang M, Rigby AC, Morelli X, Grant MA, Huang G, Furie B, Seaton B, Furie BC (September 2003). "Structural basis of membrane binding by Gla domains of vitamin K-dependent proteins". Nat. Struct. Biol. 10 (9): 751–6. doi:10.1038/nsb971. PMID 12923575.
- ^ Degen SJ, McDowell SA, Sparks LM, Scharrer I (February 1995). "Prothrombin Frankfurt: a dysfunctional prothrombin characterized by substitution of Glu-466 by Ala". Thromb. Haemost. 73 (2): 203–9. PMID 7792730.
- ^ Varga EA, Moll S (July 2004). "Cardiology patient pages. Prothrombin 20210 mutation (factor II mutation)". Circulation 110 (3): e15–8. doi:10.1161/01.CIR.0000135582.53444.87. PMID 15262854.
- ^ Bernstein CN, Sargent M, et al. (2007). "Mutations in clotting factors and inflammatory bowel disease". Am. J. Gastroenterol. 102 (2): 338–343. doi:10.1111/j.1572-0241.2006.00974.x. PMID 17156138.
- ^ Borissoff JI, Spronk HM, Heeneman S, ten Cate H. Is thrombin a key player in the 'coagulation-atherogenesis' maze? Cardiovasc Res. 2009;82(3):392-403. PMID19228706.http://cardiovascres.oxfordjournals.org/content/82/3/392.long
- ^ Borissoff JI, Heeneman S, Kilinc E, Kassak P, Van Oerle R, Winckers K, Govers-Riemslag JW, Hamulyak K, Hackeng TM, Daemen MJ, ten Cate H, Spronk HM. Early atherosclerosis exhibits an enhanced procoagulant state. Circulation. 2010;122(8):821-830. PMID20697022. http://circ.ahajournals.org/cgi/content/full/122/8/821
- ^ Borissoff JI, Spronk HM, ten Cate H. The hemostatic system as a modulator of atherosclerosis. N Engl J Med. 2011;364(18):1746-1760. PMID21542745. http://www.nejm.org/doi/full/10.1056/NEJMra1011670
- ^ Chapman WC, Singla N, Genyk Y, McNeil JW, Renkens KL, Reynolds TC, Murphy A, Weaver FA (August 2007). "A phase 3, randomized, double-blind comparative study of the efficacy and safety of topical recombinant human thrombin and bovine thrombin in surgical hemostasis". J. Am. Coll. Surg. 205 (2): 256–65. doi:10.1016/j.jamcollsurg.2007.03.020. PMID 17660072.
- ^ Singla NK, Ballard JL, Moneta G, Randleman CD, Renkens KL, Alexander WA (July 2009). "A phase 3b, open-label, single-group immunogenicity and safety study of topical recombinant thrombin in surgical hemostasis". J. Am. Coll. Surg. 209 (1): 68–74. doi:10.1016/j.jamcollsurg.2009.03.016. PMID 19651065.
- ^ Greenhalgh DG, Gamelli RL, Collins J, Sood R, Mozingo DW, Gray TE, Alexander WA (2009). "Recombinant thrombin: safety and immunogenicity in burn wound excision and grafting". J Burn Care Res 30 (3): 371–9. doi:10.1097/BCR.0b013e3181a28979. PMID 19349898.
- ^ a b "Sverige röstade ja till köttklister [Sweden voted in favor of the meat paste]" (in Swedish). Dagens Nyheter. 2010-02-09. http://www.dn.se/nyheter/politik/sverige-rostade-ja-till-kottklister-1.1042136. Retrieved 2010-10-17.
- ^ "Welcome to Fibrimex". Fibrimex website. FX Technology. http://www.fibrimex.com. Retrieved 2010-10-17.
Further reading
- Esmon CT (1995). "Thrombomodulin as a model of molecular mechanisms that modulate protease specificity and function at the vessel surface". FASEB J 9 (10): 946–55. PMID 7615164.
- Lenting PJ, van Mourik JA, Mertens K (1999). "The life cycle of coagulation factor VIII in view of its structure and function". Blood 92 (11): 3983–96. PMID 9834200.
- Plow EF, Cierniewski CS, Xiao Z, et al. (2002). "AlphaIIbbeta3 and its antagonism at the new millennium". Thromb. Haemost 86 (1): 34–40. PMID 11487023.
- Maragoudakis ME, Tsopanoglou NE, Andriopoulou P (2002). "Mechanism of thrombin-induced angiogenesis". Biochem. Soc. Trans 30 (2): 173–7. doi:10.1042/BST0300173. PMID 12023846.
- Howell DC, Laurent GJ, Chambers RC (2002). "Role of thrombin and its major cellular receptor, protease-activated receptor-1, in pulmonary fibrosis". Biochem. Soc. Trans 30 (2): 211–6. doi:10.1042/BST0300211. PMID 12023853.
- Firth SM, Baxter RC (2003). "Cellular actions of the insulin-like growth factor binding proteins". Endocr. Rev 23 (6): 824–54. doi:10.1210/er.2001-0033. PMID 12466191.
- Minami T, Sugiyama A, Wu SQ, et al. (2004). "Thrombin and phenotypic modulation of the endothelium". Arterioscler. Thromb. Vasc. Biol 24 (1): 41–53. doi:10.1161/01.ATV.0000099880.09014.7D. PMID 14551154.
- De Cristofaro R, De Candia E (2004). "Thrombin domains: structure, function and interaction with platelet receptors". J. Thromb. Thrombolysis 15 (3): 151–63. doi:10.1023/B:THRO.0000011370.80989.7b. PMID 14739624.
- Tsopanoglou NE, Maragoudakis ME (2004). "Role of thrombin in angiogenesis and tumor progression". Semin. Thromb. Hemost 30 (1): 63–9. doi:10.1055/s-2004-822971. PMID 15034798.
- Bode W (2007). "Structure and interaction modes of thrombin". Blood Cells Mol. Dis 36 (2): 122–30. doi:10.1016/j.bcmd.2005.12.027. PMID 16480903.
- Wolberg AS (2007). "Thrombin generation and fibrin clot structure". Blood Rev 21 (3): 131–42. doi:10.1016/j.blre.2006.11.001. PMID 17208341.
- Degen S: Prothrombin. In: High K, Roberts H, eds. Molecular Basis of Thrombosis and Hemostasis. New York, NY: Marcel Dekker; 1995:75.
External links
- The MEROPS online database for peptidases and their inhibitors: S01.217
- GeneReviews/NCBI/NIH/UW entry on Prothrombin Thrombophilia
- [3] Anti-coagulation & proteases by The Proteolysis Map-animation
- [4] PMAP: The Proteolysis Map/Thrombin
PDB gallery 1a2c: STRUCTURE OF THROMBIN INHIBITED BY AERUGINOSIN298-A FROM A BLUE-GREEN ALGA1a3b: COMPLEX OF HUMAN ALPHA-THROMBIN WITH THE BIFUNCTIONAL BORONATE INHIBITOR BOROLOG11a3e: COMPLEX OF HUMAN ALPHA-THROMBIN WITH THE BIFUNCTIONAL BORONATE INHIBITOR BOROLOG21a46: THROMBIN COMPLEXED WITH HIRUGEN AND A BETA-STRAND MIMETIC INHIBITOR1a4w: CRYSTAL STRUCTURES OF THROMBIN WITH THIAZOLE-CONTAINING INHIBITORS: PROBES OF THE S1' BINDING SITE1a5g: HUMAN THROMBIN COMPLEXED WITH NOVEL SYNTHETIC PEPTIDE MIMETIC INHIBITOR AND HIRUGEN1a61: THROMBIN COMPLEXED WITH A BETA-MIMETIC THIAZOLE-CONTAINING INHIBITOR1abi: STRUCTURE OF THE HIRULOG 3-THROMBIN COMPLEX AND NATURE OF THE S' SUBSITES OF SUBSTRATES AND INHIBITORS1abj: STRUCTURE OF THE HIRULOG 3-THROMBIN COMPLEX AND NATURE OF THE S' SUBSITES OF SUBSTRATES AND INHIBITORS1ad8: COMPLEX OF THROMBIN WITH AND INHIBITOR CONTAINING A NOVEL P1 MOIETY1ae8: HUMAN ALPHA-THROMBIN INHIBITION BY EOC-D-PHE-PRO-AZALYS-ONP1aht: CRYSTAL STRUCTURE OF HUMAN ALPHA-THROMBIN COMPLEXED WITH HIRUGEN AND P-AMIDINOPHENYLPYRUVATE) AT 1.6 ANGSTROMS RESOLUTION1ai8: HUMAN ALPHA-THROMBIN TERNARY COMPLEX WITH THE EXOSITE INHIBITOR HIRUGEN AND ACTIVE SITE INHIBITOR PHCH2OCO-D-DPA-PRO-BOROMPG1aix: HUMAN ALPHA-THROMBIN TERNARY COMPLEX WITH EXOSITE INHIBITOR HIRUGEN AND ACTIVE SITE INHIBITOR PHCH2OCO-D-DPA-PRO-BOROVAL1awf: NOVEL COVALENT THROMBIN INHIBITOR FROM PLANT EXTRACT1awh: NOVEL COVALENT THROMBIN INHIBITOR FROM PLANT EXTRACT1ay6: THROMBIN INHIBITOR FROM THEONALLA, CYCLOTHEANAMIDE-BASED MACROCYCLIC TRIPEPTIDE MOTIF1b5g: HUMAN THROMBIN COMPLEXED WITH NOVEL SYNTHETIC PEPTIDE MIMETIC INHIBITOR AND HIRUGEN1b7x: STRUCTURE OF HUMAN ALPHA-THROMBIN Y225I MUTANT BOUND TO D-PHE-PRO-ARG-CHLOROMETHYLKETONE1ba8: THROMBIN INHIBITOR WITH A RIGID TRIPEPTIDYL ALDEHYDES1bb0: THROMBIN INHIBITORS WITH RIGID TRIPEPTIDYL ALDEHYDES1bbr: THE STRUCTURE OF RESIDUES 7-16 OF THE A ALPHA CHAIN OF HUMAN FIBRINOGEN BOUND TO BOVINE THROMBIN AT 2.3 ANGSTROMS RESOLUTION1bcu: ALPHA-THROMBIN COMPLEXED WITH HIRUGEN AND PROFLAVIN1bhx: X-RAY STRUCTURE OF THE COMPLEX OF HUMAN ALPHA THROMBIN WITH THE INHIBITOR SDZ 229-3571bth: STRUCTURE OF THROMBIN COMPLEXED WITH BOVINE PANCREATIC TRYPSIN INHIBITOR1c1u: RECRUITING ZINC TO MEDIATE POTENT, SPECIFIC INHIBITION OF SERINE PROTEASES1c1v: RECRUITING ZINC TO MEDIATE POTENT, SPECIFIC INHIBITION OF SERINE PROTEASES1c1w: RECRUITING ZINC TO MEDIATE POTENT, SPECIFIC INHIBITION OF SERINE PROTEASES1c4u: SELECTIVE NON ELECTROPHILIC THROMBIN INHIBITORS WITH CYCLOHEXYL MOIETIES.1c4v: SELECTIVE NON ELECTROPHILIC THROMBIN INHIBITORS WITH CYCLOHEXYL MOIETIES.1c4y: SELECTIVE NON-ELECTROPHILIC THROMBIN INHIBITORS1c5l: STRUCTURAL BASIS FOR SELECTIVITY OF A SMALL MOLECULE, S1-BINDING, SUB-MICROMOLAR INHIBITOR OF UROKINASE TYPE PLASMINOGEN ACTIVATOR1c5n: STRUCTURAL BASIS FOR SELECTIVITY OF A SMALL MOLECULE, S1-BINDING, SUB-MICROMOLAR INHIBITOR OF UROKINASE TYPE PLASMINOGEN ACTIVATOR1c5o: STRUCTURAL BASIS FOR SELECTIVITY OF A SMALL MOLECULE, S1-BINDING, SUB-MICROMOLAR INHIBITOR OF UROKINASE TYPE PLASMINOGEN ACTIVATOR1ca8: THROMBIN INHIBITORS WITH RIGID TRIPEPTIDYL ALDEHYDES1d3d: CRYSTAL STRUCTURE OF HUMAN ALPHA THROMBIN IN COMPLEX WITH BENZOTHIOPHENE INHIBITOR 41d3p: CRYSTAL STRUCTURE OF HUMAN APLHA-THROMBIN IN COMPLEX WITH BENZO[B]THIOPHENE INHIBITOR 31d3q: CRYSTAL STRUCTURE OF HUMAN ALPHA THROMBIN IN COMPLEX WITH BENZO[B]THIOPHENE INHIBITOR 21d3t: CRYSTAL STRUCTURE OF HUMAN ALPHA THROMBIN IN COMPLEX WITH BENZO[B]THIOPHENE INHIBITOR 11d4p: CRYSTAL STRUCTURE OF HUMAN ALPHA THROMBIN IN COMPLEX WITH 5-AMIDINOINDOLE-4-BENZYLPIPERIDINE INHIBITOR1d6w: STRUCTURE OF THROMBIN COMPLEXED WITH SELECTIVE NON-ELECTROPHILIC INHIBITORS HAVING CYCLOHEXYL MOIETIES AT P11d9i: STRUCTURE OF THROMBIN COMPLEXED WITH SELECTIVE NON-ELECTOPHILIC INHIBITORS HAVING CYCLOHEXYL MOIETIES AT P11de7: INTERACTION OF FACTOR XIII ACTIVATION PEPTIDE WITH ALPHA-THROMBIN: CRYSTAL STRUCTURE OF THE ENZYME-SUBSTRATE COMPLEX1dit: COMPLEX OF A DIVALENT INHIBITOR WITH THROMBIN1dm4: SER195ALA MUTANT OF HUMAN THROMBIN COMPLEXED WITH FIBRINOPEPTIDE A (7-16)1doj: Crystal structure of human alpha-thrombin*RWJ-51438 complex at 1.7 A1dwb: CRYSTALLOGRAPHIC ANALYSIS AT 3.0-ANGSTROMS RESOLUTION OF THE BINDING TO HUMAN THROMBIN OF FOUR ACTIVE SITE-DIRECTED INHIBITORS1dwc: CRYSTALLOGRAPHIC ANALYSIS AT 3.0-ANGSTROMS RESOLUTION OF THE BINDING TO HUMAN THROMBIN OF FOUR ACTIVE SITE-DIRECTED INHIBITORS1dwd: CRYSTALLOGRAPHIC ANALYSIS AT 3.0-ANGSTROMS RESOLUTION OF THE BINDING TO HUMAN THROMBIN OF FOUR ACTIVE SITE-DIRECTED INHIBITORS1dwe: CRYSTALLOGRAPHIC ANALYSIS AT 3.0-ANGSTROMS RESOLUTION OF THE BINDING TO HUMAN THROMBIN OF FOUR ACTIVE SITE-DIRECTED INHIBITORS1dx5: CRYSTAL STRUCTURE OF THE THROMBIN-THROMBOMODULIN COMPLEX1e0f: CRYSTAL STRUCTURE OF THE HUMAN ALPHA-THROMBIN-HAEMADIN COMPLEX: AN EXOSITE II-BINDING INHIBITOR1eb1: COMPLEX STRUCTURE OF HUMAN THROMBIN WITH N-METHYL-ARGININE INHIBITOR1eoj: DESIGN OF P1' AND P3' RESIDUES OF TRIVALENT THROMBIN INHIBITORS AND THEIR CRYSTAL STRUCTURES1eol: DESIGN OF P1' AND P3' RESIDUES OF TRIVALENT THROMBIN INHIBITORS AND THEIR CRYSTAL STRUCTURES1fpc: ACTIVE SITE MIMETIC INHIBITION OF THROMBIN1fph: THE INTERACTION OF THROMBIN WITH FIBRINOGEN: A STRUCTURAL BASIS FOR ITS SPECIFICITY1g30: THROMBIN INHIBITOR COMPLEX1g32: THROMBIN INHIBITOR COMPLEX1g37: CRYSTAL STRUCTURE OF HUMAN ALPHA-THROMBIN COMPLEXED WITH BCH-10556 AND EXOSITE-DIRECTED PEPTIDE1ghv: A NOVEL SERINE PROTEASE INHIBITION MOTIF INVOLVING A MULTI-CENTERED SHORT HYDROGEN BONDING NETWORK AT THE ACTIVE SITE1ghw: A NOVEL SERINE PROTEASE INHIBITION MOTIF INVOLVING A MULTI-CENTERED SHORT HYDROGEN BONDING NETWORK AT THE ACTIVE SITE1ghx: A NOVEL SERINE PROTEASE INHIBITION MOTIF INVOLVING A MULTI-CENTERED SHORT HYDROGEN BONDING NETWORK AT THE ACTIVE SITE1ghy: A NOVEL SERINE PROTEASE INHIBITION MOTIF INVOLVING A MULTI-CENTERED SHORT HYDROGEN BONDING NETWORK AT THE ACTIVE SITE1gj4: SELECTIVITY AT S1, H2O DISPLACEMENT, UPA, TPA, SER190/ALA190 PROTEASE, STRUCTURE-BASED DRUG DESIGN1gj5: SELECTIVITY AT S1, H2O DISPLACEMENT, UPA, TPA, SER190/ALA190 PROTEASE, STRUCTURE-BASED DRUG DESIGN1h8d: X-RAY STRUCTURE OF THE HUMAN ALPHA-THROMBIN COMPLEX WITH A TRIPEPTIDE PHOSPHONATE INHIBITOR.1h8i: X-RAY CRYSTAL STRUCTURE OF HUMAN ALPHA-THROMBIN WITH A TRIPEPTIDE PHOSPHONATE INHIBITOR.1hag: THE ISOMORPHOUS STRUCTURES OF PRETHROMBIN2, HIRUGEN-AND PPACK-THROMBIN: CHANGES ACCOMPANYING ACTIVATION AND EXOSITE BINDING TO THROMBIN1hah: THE ISOMORPHOUS STRUCTURES OF PRETHROMBIN2, HIRUGEN-AND PPACK-THROMBIN: CHANGES ACCOMPANYING ACTIVATION AND EXOSITE BINDING TO THROMBIN1hai: THE ISOMORPHOUS STRUCTURES OF PRETHROMBIN2, HIRUGEN-AND PPACK-THROMBIN: CHANGES ACCOMPANYING ACTIVATION AND EXOSITE BINDING TO THROMBIN1hao: COMPLEX OF HUMAN ALPHA-THROMBIN WITH A 15MER OLIGONUCLEOTIDE GGTTGGTGTGGTTGG (BASED ON NMR MODEL OF DNA1hap: COMPLEX OF HUMAN ALPHA-THROMBIN WITH A 15MER OLIGONUCLEOTIDE GGTTGGTGTGGTTGG (BASED ON X-RAY MODEL OF DNA)1hbt: HUMAN ALPHA-THROMBIN COMPLEXED WITH A PEPTIDYL PYRIDINIUM METHYL KETONE CONTAINING BIVALENT INHIBITOR1hdt: STRUCTURE OF A RETRO-BINDING PEPTIDE INHIBITOR COMPLEXED WITH HUMAN ALPHA-THROMBIN1hgt: STRUCTURE OF THE HIRUGEN AND HIRULOG 1 COMPLEXES OF ALPHA-THROMBIN1hlt: THE STRUCTURE OF A NONADECAPEPTIDE OF THE FIFTH EGF DOMAIN OF THROMBOMODULIN COMPLEXED WITH THROMBIN1hut: THE STRUCTURE OF ALPHA-THROMBIN INHIBITED BY A 15-MER SINGLE-STRANDED DNA APTAMER1hxe: SERINE PROTEASE1hxf: HUMAN THROMBIN COMPLEX WITH HIRUDIN VARIANT1ihs: CRYSTAL STRUCTURE OF THE COMPLEX OF HUMAN ALPHA-THROMBIN AND NON-HYDROLYZABLE BIFUNCTIONAL INHIBITORS, HIRUTONIN-2 AND HIRUTONIN-61iht: CRYSTAL STRUCTURE OF THE COMPLEX OF HUMAN ALPHA-THROMBIN AND NON-HYDROLYZABLE BIFUNCTIONAL INHIBITORS, HIRUTONIN-2 AND HIRUTONIN-61jmo: Crystal Structure of the Heparin Cofactor II-S195A Thrombin Complex1jou: Crystal Structure of Native S195A Thrombin with an Unoccupied Active Site1jwt: CRYSTAL STRUCTURE OF THROMBIN IN COMPLEX WITH A NOVEL BICYCLIC LACTAM INHIBITOR1k21: HUMAN THROMBIN-INHIBITOR COMPLEX1k22: HUMAN THROMBIN-INHIBITOR COMPLEX1kts: Thrombin Inhibitor Complex1ktt: Thrombin inhibitor complex1lhc: HUMAN ALPHA-THROMBIN COMPLEXED WITH AC-(D)PHE-PRO-BOROARG-OH1lhd: HUMAN ALPHA-THROMBIN COMPLEXED WITH AC-(D)PHE-PRO-BOROLYS-OH1lhe: HUMAN ALPHA-THROMBIN COMPLEXED WITH AC-(D)PHE-PRO-BORO-N-BUTYL-AMIDINO-GLYCINE-OH1lhf: HUMAN ALPHA-THROMBIN COMPLEXED WITH AC-(D)PHE-PRO-BORO-HOMOLYS-OH1lhg: HUMAN ALPHA-THROMBIN COMPLEXED WITH AC-(D)PHE-PRO-BOROORNITHINE-OH1mh0: Crystal structure of the anticoagulant slow form of thrombin1mu6: Crystal Structure of Thrombin in Complex with L-378,6221mu8: thrombin-hirugen_l-378,6501mue: Thrombin-Hirugen-L405,4261nm6: thrombin in complex with selective macrocyclic inhibitor at 1.8A1no9: Design of weakly basic thrombin inhibitors incorporating novel P1 binding functions: molecular and X-ray crystallographic studies.The maximum number of images (100) is exceeded ! Proteins: coagulation Coagulation factors Coagulation inhibitors Thrombolysis/fibrinolysis Digestive enzymes Coagulation factors: Thrombin · Factor VIIa · Factor IXa · Factor Xa · Factor XIa · Factor XIIa · Kallikrein (PSA, KLK1, KLK2, KLK3, KLK4, KLK5, KLK6, KLK7, KLK8, KLK9, KLK10, KLK11, KLK12, KLK13, KLK14, KLK15)
fibrinolysis: Plasmin · Plasminogen activator (Tissue plasminogen activator · Urinary plasminogen activator)Complement system Other immune system Venombin Other Acrosin · Prolyl endopeptidase · Pronase · Proprotein convertases (1, 2) · Reelin · Subtilisin/Furin · Streptokinase · S1P · Cathepsin (A, G)Antigens: Autoantigens Dehydrogenase Transglutaminase Nucleoporins Other Acetylcholine receptor · Actin · Apolipoprotein H · Cardiolipin · Centromere · Filaggrin(Citrullinate) · Gangliosides · Sp100 nuclear antigen · Thrombin · TopoisomeraseAntihemorrhagics (B02) Hemostatics
(coagulation)Systemicintrinsic: IX/Nonacog alfa • VIII
extrinsic: VII/Eptacog alfa
common: X • II/Thrombin • I/FibrinogenOther systemicEtamsylate • Carbazochrome • Batroxobin • thrombopoietin receptor agonist (Romiplostim, Eltrombopag)LocalAntifibrinolytics Categories:- Human proteins
- Coagulation system
- Acute phase proteins
- EC 3.4.21
- Peripheral membrane proteins
Wikimedia Foundation. 2010.