- Aqueous solution
-
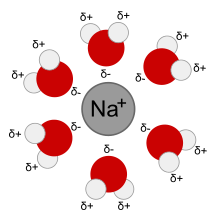
An aqueous solution is a solution in which the solvent is water. It is usually shown in chemical equations by appending aq to the relevant formula, such as NaCl(aq). The word aqueous means pertaining to, related to, similar to, or dissolved in water. As water is an excellent solvent and is also naturally abundant, it is a ubiquitous solvent in chemistry.
Substances which are hydrophobic ('water fearing') often do not dissolve well in water whereas those that hydrophilic ('water-loving') do. An example of a hydrophilic substance would be sodium chloride (ordinary table salt). Acids and bases are aqueous solutions, as part of their Arrhenius definitions.
The ability of a substance to dissolve in water is determined by whether the substance can match or exceed the strong attractive forces that water molecules generate between themselves. If the substance lacks the ability to dissolve in water the molecules form a precipitate.
Aqueous solutions that conduct electric current efficiently contain strong electrolytes, while ones that conduct poorly are considered to have weak electrolytes. Those strong electrolytes are substances that are completely ionized in water, whereas the weak electrolytes exhibit only a small degree of ionization in water.
Nonelectrolytes are substances that dissolve in water, but which maintain their molecular integrity (do not dissociate into ions). Examples include sugar, urea, glycerol, and methylsulfonylmethane (MSM).
When performing calculations regarding the reacting of one or more aqueous solutions, one generally must know the concentration, or molarity, of the aqueous solutions. Solution concentration is given in terms of the form of the solute prior to it dissolving.
See also
- Solubility
- Dissociation (chemistry)
- Acid-base reaction theories
References
- Zumdahl S. 1997. Chemistry. 4th ed. Boston: Houghton Mifflin Company. p 133-145.
Articles related to solutions Solution Ideal solution · Aqueous solution · Solid solution · Buffer solution · Flory-Huggins · Mixture · Suspension · Colloid · Phase diagram · Eutectic point · Alloy · Saturation · Supersaturation · Serial dilution · Dilution (equation) · Apparent molar propertyConcentration
and related quantitiesMolar concentration · Mass concentration · Number concentration · Volume concentration · Normality · Percentage solution · Molality · Mole fraction · Mass fraction · Mixing ratioSolubility Solubility equilibrium · Total dissolved solids · Solvation · Solvation shell · Enthalpy of solution · Lattice energy · Raoult's law · Henry's law · Solubility table (data) · Solubility chartSolvent Categories:- Chemical properties
- Solutions
- Chemistry stubs
Wikimedia Foundation. 2010.