- Tranexamic acid
-
Tranexamic acid 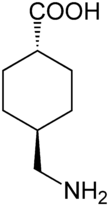
Systematic (IUPAC) name trans-4-(aminomethyl)cyclohexanecarboxylic acid Clinical data AHFS/Drugs.com Consumer Drug Information Pregnancy cat. B Legal status P (UK) Routes Injection and oral Pharmacokinetic data Bioavailability 34% Half-life 3.1 h Identifiers CAS number 1197-18-8 
ATC code B02AA02 PubChem CID 5526 DrugBank APRD01270 ChemSpider 10482000 
UNII 6T84R30KC1 
KEGG D01136 
ChEBI CHEBI:48669 
ChEMBL CHEMBL877 
Chemical data Formula C8H15NO2 Mol. mass 157.21 g/mol SMILES eMolecules & PubChem  (what is this?) acid (verify)
(what is this?) acid (verify)Tranexamic acid (commonly marketed in tablet form as Lysteda and in IV form as Cyklokapron in the U.S. and Australia and as Transamin,Transcam in Asia, and Espercil in South America. Also marketed as Cyclo-F and Femstrual in UK.) is often prescribed for excessive bleeding. It is an antifibrinolytic that competitively inhibits the activation of plasminogen to plasmin, a molecule responsible for the degradation of fibrin. Fibrin is the basic framework for the formation of a blood clot in hemostasis. It has roughly 8 times the antifibrinolytic activity of an older analogue, ε-aminocaproic acid.
Contents
Therapeutic uses
Menstrual bleeding
Tranexamic acid is a synthetic derivative of the amino acid lysine. It exerts its antifibrinolytic effect through the reversible blockade of lysine-binding sites on plasminogen molecules. It inhibits endometrial (or endothelial[citation needed]) plasminogen activator and thus prevents fibrinolysis and the breakdown of blood clots. Side effects are uncommon. While prolonged treatment may heighten the risk of an increased thrombotic tendency, such as deep vein thrombosis, large scale studies reveal the incidence of thrombosis in women treated by tranexamic acid is no different from the spontaneous incidence of thrombosis in untreated women. Tranexamic acid is used as firstline nonhormonal treatment of dysfunctional uterine bleeding, and heavy bleeding associated with uterine fibroids. A recent study showed patients treated with tranexamic acid are more likely to develop thrombosis and necrosis in their fibroids, and may result in pain and fever. Moreover, the histological appearance of the necrosis in women treated by tranexamic acid is no different from the spontaneous incidence of thrombosis. The U.S. Food and Drug Administration (FDA) approved tranexamic acid oral tablets (brand name Lysteda) for treatment of heavy menstrual bleeding on 13 November 2009.
In March 2011 the status of Tranexamic acid for treatment of heavy menstrual bleeding was changed in the UK, from PoM (Prescription only Medicines) to P (Pharmacy Medicines)[1] and became available over the counter in UK pharmacies under the brand names of Cyclo-F and Femstrual, initially exclusively for Boots pharmacy, which has sparked some discussion about availability [2]. (In parts of Europe - like for example Sweden - it had then been available OTC for over a decade.)
Orthopedic Surgery
Tranexamic acid is used in orthopedic surgery to reduce bloodloss. It is of proven value in clearing the field of surgery and reducing pre- and postoperative blood loss. Drain and number of transfusions are reduced. However, the hidden blood loss is not reduced. Still, it is becoming an important tool in the anaesthetist's arsenal. It is commonly used in joint replacement surgery.
Trauma
The 20,000 patient CRASH-2 trial provides strong evidence of benefit in patients with traumatic haemorrhage [3]. All-cause mortality was significantly reduced with tranexamic acid (1463 [14·5%] tranexamic acid group vs. 1613 [16·0%] placebo group; relative risk 0·91, 95% CI 0·85–0·97; p=0·0035). The risk of death due to bleeding was significantly reduced (489 [4·9%] vs 574 [5·7%]; relative risk 0·85, 95% CI 0·76–0·96; p=0·0077). No number needed to treat (NNT) is quoted in the paper, as the authors are not of the opinion it is a useful measure. However, the NNT is easily calculated. As the Absolute Risk Reduction for all-cause mortality is 1.5%, the number of patients needed to treat to save 1 life is 1/1.5% or 1/.015 = 67 patients. The authors suggest tranexamic acid should be considered for use in bleeding trauma patients.
Dentistry
Tranexamic acid is used in dentistry in the form of a 5% mouth rinse after extractions or surgery in patients with prolonged bleeding time, e.g. from acquired or inherited disorders.
Other uses
- In obstetrics, tranexamic acid is used after delivery to reduce bleeding, often with syntocinon/oxytocin and fundal massage. A major trial is in progress in Nigeria to establish the efficacy of the drug to arrest postpartum haemorrhage (PPH). Since the drug can be administered orally, it has great potential to reduce maternal mortality rates in developing countries where primary healthcare is often unavailable.
- In cardiac surgery, e.g. coronary artery bypass surgery, it is used to prevent excessive blood loss.
- In hemophilia - Tranexamic acid is also useful in the treatment of bleeding as a second line treatment after factor VIII in patients (e.g. tooth extraction).
- In hereditary angioedema[4]
References
- ^ Tranexamic Acid to be available OtC
- ^ In defence of multiple pharmacies
- ^ Roberts I (2007). "The CRASH-2 trial of an antifibrinolytic agent in traumatic haemorrhage: an international collaboration". Indian J Med Res 125 (1): 5–7. PMID 17332649.
- ^ Rod Flower; Humphrey P. Rang; Maureen M. Dale; Ritter, James M. (2007). Rang & Dale's pharmacology. Edinburgh: Churchill Livingstone. ISBN 0-443-06911-5.
External links
- [1] (PoM to P in the UK)
- [2] (Exclusivity controversy)
- [3] (Final results from the Crash2 study)
- Patient Experience with Tranexamic Acid (Patient Experience)
- Tranexamic acid (Dr. P.L.F. Giangrande, Oxford Haemophilia Centre)
- Tranexamic acid (UK patient information leaflet)
- Effects of tranexamic acid on death, vascular occlusive events, and blood transfusion in trauma patients with significant haemorrhage (CRASH-2): a randomised, placebo-controlled trial (The Lancet)
- Drug will save lives of accident victims, says study (BBC News article)
- Types of Angioedema and treatments (Hereditary Angioedema Association)
Antihemorrhagics (B02) Hemostatics
(coagulation)SystemicOther systemicEtamsylate • Carbazochrome • Batroxobin • thrombopoietin receptor agonist (Romiplostim, Eltrombopag)LocalAntifibrinolytics amino acids (Aminocaproic acid, Tranexamic acid, Aminomethylbenzoic acid)
serpins (Aprotinin, Alfa1 antitrypsin, C1-inhibitor, Camostat)
This drug article relating to the blood and blood forming organs is a stub. You can help Wikipedia by expanding it.
