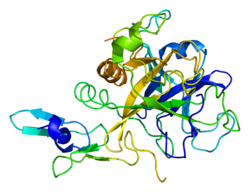- Factor X
-
For other uses, see Factor X (disambiguation).
Factor X, also known by the eponym Stuart-Prower factor or as prothrombinase, is an enzyme (EC 3.4.21.6) of the coagulation cascade. It is a serine endopeptidase (protease group S1).
Contents
Physiology
Factor X is synthesized in the liver and requires vitamin K for its synthesis.
Factor X is activated into factor Xa by both factor IX (with its cofactor, factor VIII in a complex known as intrinsic Xase) and factor VII with its cofactor, tissue factor (a complex known as extrinsic Xase). It is therefore the first member of the final common pathway or thrombin pathway.
It acts by cleaving prothrombin in two places (an arg-thr and then an arg-ile bond), which yields the active thrombin. This process is optimized when factor Xa is complexed with activated co-factor V in the prothrombinase complex.
Factor Xa is inactivated by protein Z-dependent protease inhibitor (ZPI), a serine protease inhibitor (serpin). The affinity of this protein for factor Xa is increased 1000-fold by the presence of protein Z, while it does not require protein Z for inactivation of factor XI. Defects in protein Z lead to increased factor Xa activity and a propensity for thrombosis.
The half life of factor X is 40-45 hours.
Structure of Factor Xa
The first crystal structure of human factor Xa was deposited in May 1993. Till now total 191 crystal structures of factor Xa with various inhibitors have been deposited in the protein data bank. The active site of factor Xa is divided into four sub pockets as S1, S2, S3 and S4. The S1 subpocket determines the major component of selectivity and binding. The S2 sub-pocket is small, shallow and not well defined. It merges with the S4 subpocket. The S3 sub-pocket is located on the rim of the S1 pocket and is quite exposed to solvent. The S4 sub-pocket has 3 ligand binding domains, namely the “hydrophobic box”, the “cationic hole” and the water site. Factor Xa inhibitors generally bind in an L-shaped conformation, where one group of the ligand occupies the anionic S1 pocket lined by residues Asp189, Ser195, and Tyr228, and another group of the ligand occupies the aromatic S4 pocket lined by residues Tyr99, Phe174, and Trp228. Typically, a fairly rigid linker group bridges these two interaction sites.[1]
Genetics
The human factor X gene is located on the thirteenth chromosome (13q34).
Role in disease
Inborn deficiency of factor X is very uncommon (1:500,000), and may present with epistaxis (nosebleeds), hemarthrosis (bleeding into joints) and gastrointestinal blood loss. Apart from congenital deficiency, low factor X levels may occur occasionally in a number of disease states. For example, factor X deficiency may be seen in amyloidosis, where factor X is adsorbed to the amyloid fibrils in the vasculature.
Deficiency of vitamin K or antagonism by warfarin (or similar medication) leads to the production of an inactive factor X. In warfarin therapy, this is desirable to prevent thrombosis. As of late 2007, four out of five emerging anti-coagulation therapeutics targeted this enzyme.[2]
Inhibiting Factor Xa would offer an alternate method for anticoagulation. Direct Xa inhibitors are popular anticoagulants.
Condition Prothrombin time Partial thromboplastin time Bleeding time Platelet count Vitamin K deficiency or warfarin prolonged prolonged unaffected unaffected Disseminated intravascular coagulation prolonged prolonged prolonged decreased von Willebrand disease unaffected prolonged prolonged unaffected Haemophilia unaffected prolonged unaffected unaffected Aspirin unaffected unaffected prolonged unaffected Thrombocytopenia unaffected unaffected prolonged decreased Early Liver failure prolonged unaffected unaffected unaffected End-stage Liver failure prolonged prolonged prolonged decreased Uremia unaffected unaffected prolonged unaffected Congenital afibrinogenemia prolonged prolonged prolonged unaffected Factor V deficiency prolonged prolonged unaffected unaffected Factor X deficiency as seen in amyloid purpura prolonged prolonged unaffected unaffected Glanzmann's thrombasthenia unaffected unaffected prolonged unaffected Bernard-Soulier syndrome unaffected unaffected prolonged unaffected [3] Therapeutic use
Factor X is part of fresh frozen plasma and the prothrombinase complex. The only commercially available concentrate is 'Factor X P Behring' manufactured by CSL Behring. [4]
Use in Biochemistry
The Factor Xa protease can be used in biochemistry to cleave off protein tags that improve expression or purification of a protein of interest. Its preferred cleavage site (after the arginine in ile-glu/asp-gly-arg) can easily be engineered between a tag sequence and the protein of interest. After expression and purification, the tag is then proteolytically removed by Factor Xa.
Factor Xa
Factor Xa is the activated form of the coagulation factor thrombokinase, known eponymously as Stuart-Prower factor. Factor X is an enzyme, a serine endopeptidase, which plays a key role at several stages of the coagulation system. Factor X is synthesized in the liver. The most commonly used anticoagulants in clinical practice, warfarin and the heparin series of anticoagulants and fondaparinux, act to inhibit the action of Factor Xa in various degrees.
Traditional models of coagulation developed in the 1960’s envisaged two separate cascades, the extrinsic (tissue factor (TF)) pathway and the intrinsic pathway. These pathways converge to a common point, the formation of the Factor Xa/Va complex which together with calcium and bound on a phospholipids surface generate thrombin (Factor IIa) from prothrombin (Factor II).
A new model, the cell-based model of anticoagulation appears to explain more fully the steps in coagulation. This model has three stages: 1) initiation of coagulation on TF-bearing cells, 2) amplification of the procoagulant signal by thrombin generated on the TF-bearing cell and 3) propagation of thrombin generation on the platelet surface. Factor Xa plays a key role in all three of these stages.[5]
In stage 1, Factor VII binds to the transmembrane protein TF on the surface of cells and is converted to Factor VIIa. The result is a Factor VIIa/TF complex which catalyzes the activation of Factor X and Factor IX. Factor Xa formed on the surface of the TF-bearing cell interacts with Factor Va to form the prothrombinase complex which generates small amounts of thrombin on the surface of TF-bearing cells.
In stage 2, the amplification stage, if enough thrombin has been generated, then activation of platelets and platelet associated cofactors occurs.
In stage 3, thrombin generation, Factor XIa activates free Factor IX on the surface of activated platelets. The activated Factor IXa with Factor VIIIa forms the ”tenase” complex. This “tenase” complex activates more Factor X, which in turn forms new prothrombinase complexes with Factor Va. Factor Xa is the prime component of the prothrombinase complex which converts large amounts of prothrombin – the “thrombin burst”. Each molecule of Factor Xa can generate 1000 molecules of thrombin. This large burst of thrombin is responsible for fibrin polymerization to form a thrombus.
Inhibition of the synthesis or activity of Factor X is the mechanism of action for many anticoagulants in use today. Warfarin, a synthetic derivative of coumarin, is the most widely used oral anticoagulant in the US. In some European countries, other coumarin derivatives (phenprocoumon and acenocoumarol) are used. These agents are vitamin K antagonists (VKA). Vitamin K is essential for the hepatic synthesis of Factors II (prothrombin), VII, IX and X.
Heparin (unfractionated heparin) and its derivatives low molecular weight heparin (LMWH) bind to a plasma cofactor, antithrombin (AT) to inactivate several coagulation factors IIa, Xa, XIa and XIIa. The affinity of unfractionated heparin and the various LMWHs for Factor Xa varies considerably. The efficacy of heparin-based anticoagulants increases as selectivity for Factor Xa increases. LMWH shows increased inactivation of Factor Xa compared to unfractionated heparin, and fondaparinux, an agent based on the critical pentasacharide sequence of heparin, shows more selectivity than LMWH. This inactivation of Factor Xa by heparins is termed “indirect” since it relies on the presence of AT and not a direct interaction with Factor Xa.
Recently a new series of specific, direct acting inhibitors of Factor Xa has been developed. These include the drugs rivaroxaban, apixaban, betrixaban, LY517717, YM150, edoxaban and 813893. These agents have several theoretical advantages over current therapy. They may be given orally. They have rapid Onset of action. And they may be more effective against Factor Xa in that they inhibit both free Factor Xa and Factor Xa in the prothrombinase complex.[6]
History
American and British scientists described deficiency of factor X independently in 1953 and 1956, respectively. As with some other coagulation factors, the factor was initially named after these patients, a Mr Rufus Stuart (1921) and a Miss Audrey Prower (1934).
External links
- The MEROPS online database for peptidases and their inhibitors: S01.216
- med/3495 at eMedicine
- Factor X deficiency
Interactions
Factor X has been shown to interact with Tissue factor pathway inhibitor.[7]
References
- ^ Presentation on Direct Factor Xa Inhibitors
- ^ Ron Winslow; Avery Johnson (2007-12-10). "Race Is on for the Next Blood Thinner". Wall Street Journal: p. A12. http://online.wsj.com/article/SB119725064671318856.html. Retrieved 2008-01-06. "The flurry of interest reflects increasing understanding of what doctors call the coagulation cascade... Four new blood thinners target an enzyme called factor Xa, one of several enzymes that play an important role in the cascade."
- ^ Kaplan QBook - USMLE Step 1 - 5th edition - page 254
- ^ Mark Brooker (2008): "Registry of Clotting Factor Concentrates". Eighth Edition, 2008, World Federation of Hemophilia
- ^ Hoffman M; Monroe MM (2007). "Coagulation 2006: A Modern View of Hemostasis". Hematology/Oncology Clinics of North America 21 (1):1-11. PMID 17258114. http://www.ncbi.nlm.nih.gov/pubmed/17258114.
- ^ Turpie AG (2007). "Oral, Direct Factor Xa Inhibitors in Development for the Prevention and Treatment of Thromboembolic Diseases". Arterioscl Thromb Vasc Biol, 27 (6):1238-47, PMID 17379841. http://www.ncbi.nlm.nih.gov/pubmed/17379841.
- ^ Broze, G J; Warren L A, Novotny W F, Higuchi D A, Girard J J, Miletich J P (Feb. 1988). "The lipoprotein-associated coagulation inhibitor that inhibits the factor VII-tissue factor complex also inhibits factor Xa: insight into its possible mechanism of action". Blood (UNITED STATES) 71 (2): 335–43. ISSN 0006-4971. PMID 3422166.
Further reading
- Cooper DN, Millar DS, Wacey A, et al. (1997). "Inherited factor X deficiency: molecular genetics and pathophysiology". Thromb. Haemost. 78 (1): 161–72. PMID 9198147.
- Hassan HJ, Leonardi A, Chelucci C, et al. (1990). "Blood coagulation factors in human embryonic-fetal development: preferential expression of the FVII/tissue factor pathway". Blood 76 (6): 1158–64. PMID 1698100.
- Messier TL, Pittman DD, Long GL, et al. (1991). "Cloning and expression in COS-1 cells of a full-length cDNA encoding human coagulation factor X". Gene 99 (2): 291–4. doi:10.1016/0378-1119(91)90141-W. PMID 1902434.
- Krishnaswamy S (1990). "Prothrombinase complex assembly. Contributions of protein-protein and protein-membrane interactions toward complex formation". J. Biol. Chem. 265 (7): 3708–18. PMID 2303476.
- España F, Berrettini M, Griffin JH (1989). "Purification and characterization of plasma protein C inhibitor". Thromb. Res. 55 (3): 369–84. doi:10.1016/0049-3848(89)90069-8. PMID 2551064.
- Fung MR, Hay CW, MacGillivray RT (1985). "Characterization of an almost full-length cDNA coding for human blood coagulation factor X". Proc. Natl. Acad. Sci. U.S.A. 82 (11): 3591–5. doi:10.1073/pnas.82.11.3591. PMC 397831. PMID 2582420. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=397831.
- Jagadeeswaran P, Reddy SV, Rao KJ, et al. (1990). "Cloning and characterization of the 5' end (exon 1) of the gene encoding human factor X". Gene 84 (2): 517–9. doi:10.1016/0378-1119(89)90529-5. PMID 2612918.
- Reddy SV, Zhou ZQ, Rao KJ, et al. (1989). "Molecular characterization of human factor XSan Antonio". Blood 74 (5): 1486–90. PMID 2790181.
- Kaul RK, Hildebrand B, Roberts S, Jagadeeswaran P (1986). "Isolation and characterization of human blood-coagulation factor X cDNA". Gene 41 (2–3): 311–4. doi:10.1016/0378-1119(86)90112-5. PMID 3011603.
- Broze GJ, Warren LA, Novotny WF, et al. (1988). "The lipoprotein-associated coagulation inhibitor that inhibits the factor VII-tissue factor complex also inhibits factor Xa: insight into its possible mechanism of action". Blood 71 (2): 335–43. PMID 3422166.
- Gilgenkrantz S, Briquel ME, André E, et al. (1986). "Structural genes of coagulation factors VII and X located on 13q34". Ann. Genet. 29 (1): 32–5. PMID 3487272.
- Leytus SP, Foster DC, Kurachi K, Davie EW (1986). "Gene for human factor X: a blood coagulation factor whose gene organization is essentially identical with that of factor IX and protein C". Biochemistry 25 (18): 5098–102. doi:10.1021/bi00366a018. PMID 3768336.
- Leytus SP, Chung DW, Kisiel W, et al. (1984). "Characterization of a cDNA coding for human factor X". Proc. Natl. Acad. Sci. U.S.A. 81 (12): 3699–702. doi:10.1073/pnas.81.12.3699. PMC 345286. PMID 6587384. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=345286.
- McMullen BA, Fujikawa K, Kisiel W, et al. (1983). "Complete amino acid sequence of the light chain of human blood coagulation factor X: evidence for identification of residue 63 as beta-hydroxyaspartic acid". Biochemistry 22 (12): 2875–84. doi:10.1021/bi00281a016. PMID 6871167.
- Marchetti G, Castaman G, Pinotti M, et al. (1995). "Molecular bases of CRM+ factor X deficiency: a frequent mutation (Ser334Pro) in the catalytic domain and a substitution (Glu102Lys) in the second EGF-like domain". Br. J. Haematol. 90 (4): 910–5. doi:10.1111/j.1365-2141.1995.tb05214.x. PMID 7669671.
- Morgenstern KA, Sprecher C, Holth L, et al. (1994). "Complementary DNA cloning and kinetic characterization of a novel intracellular serine proteinase inhibitor: mechanism of action with trypsin and factor Xa as model proteinases". Biochemistry 33 (11): 3432–41. doi:10.1021/bi00177a037. PMID 8136380.
- Heeb MJ, Rosing J, Bakker HM, et al. (1994). "Protein S binds to and inhibits factor Xa". Proc. Natl. Acad. Sci. U.S.A. 91 (7): 2728–32. doi:10.1073/pnas.91.7.2728. PMC 43443. PMID 8146182. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=43443.
- Inoue K, Morita T (1993). "Identification of O-linked oligosaccharide chains in the activation peptides of blood coagulation factor X. The role of the carbohydrate moieties in the activation of factor X". Eur. J. Biochem. 218 (1): 153–63. doi:10.1111/j.1432-1033.1993.tb18361.x. PMID 8243461.
- Padmanabhan K, Padmanabhan KP, Tulinsky A, et al. (1993). "Structure of human des(1-45) factor Xa at 2.2 A resolution". J. Mol. Biol. 232 (3): 947–66. doi:10.1006/jmbi.1993.1441. PMID 8355279.
- Sinha U, Wolf DL (1993). "Carbohydrate residues modulate the activation of coagulation factor X". J. Biol. Chem. 268 (5): 3048–51. PMID 8428982.
PDB gallery {{Gallery |lines=4 |Image:PDB_1c5m_EBI.jpg|1c5m: STRUCTURAL BASIS FOR SELECTIVITY OF A SMALL MOLECULE, S1-BINDING, SUB-MICROMOLAR INHIBITOR OF UROKINASE TYPE PLASMINOGEN ACTIVATOR |Image:PDB_1ezq_EBI.jpg|1ezq: CRYSTAL STRUCTURE OF HUMAN COAGULATION FACTOR XA COMPLEXED WITH RPR128515 |Image:PDB_1f0r_EBI.jpg|1f0r: CRYSTAL STRUCTURE OF HUMAN COAGULATION FACTOR XA COMPLEXED WITH RPR208815 |Image:PDB_1f0s_EBI.jpg|1f0s: Crystal Structure of Human Coagulation Factor XA Complexed with RPR208707 |Image:PDB_1fax_EBI.jpg|1fax: COAGULATION FACTOR XA INHIBITOR COMPLEX |Image:PDB_1fjs_EBI.jpg|1fjs: CRYSTAL STRUCTURE OF THE INHIBITOR ZK-807834 (CI-1031) COMPLEXED WITH FACTOR XA |Image:PDB_1g2l_EBI.jpg|1g2l: FACTOR XA INHIBITOR COMPLEX |Image:PDB_1g2m_EBI.jpg|1g2m: FACTOR XA INHIBITOR COMPLEX |Image:PDB_1hcg_EBI.jpg|1hcg: STRUCTURE OF HUMAN DES(1-45) FACTOR XA AT 2.2 ANGSTROMS RESOLUTION |Image:PDB_1ioe_EBI.jpg|1ioe: Human coagulation factor Xa in complex with M55532 |Image:PDB_1iqe_EBI.jpg|1iqe: Human coagulation factor Xa in complex with M55590 |Image:PDB_1iqf_EBI.jpg|1iqf: Human coagulation factor Xa in complex with M55165 |Image:PDB_1iqg_EBI.jpg|1iqg: Human coagulation factor Xa in complex with M55159 |Image:PDB_1iqh_EBI.jpg|1iqh: Human coagulation factor Xa in complex with M55143 |Image:PDB_1iqi_EBI.jpg|1iqi: Human coagulation factor Xa in complex with M55125 |Image:PDB_1iqj_EBI.jpg|1iqj: Human coagulation factor Xa in complex with M55124 |Image:PDB_1iqk_EBI.jpg|1iqk: Human coagulation factor Xa in complex with M55113 |Image:PDB_1iql_EBI.jpg|1iql: Human coagulation factor Xa in complex with M54476 |Image:PDB_1iqm_EBI.jpg|1iqm: Human coagulation factor Xa in complex with M54471 |Image:PDB_1iqn_EBI.jpg|1iqn: Human coagulation factor Xa in complex with M55192 |Image:PDB_1ksn_EBI.jpg|1ksn: Crystal Structure of Human Coagulation Factor XA Complexed with FXV673 |Image:PDB_1kye_EBI.jpg|1kye: Factor Xa in complex with (R)-2-(3-adamantan-1-yl-ureido)-3-(3-carbamimidoyl-phenyl)-N-phenethyl-propionamide |Image:PDB_1lpg_EBI.jpg|1lpg: CRYSTAL STRUCTURE OF FXA IN COMPLEX WITH 79. |Image:PDB_1lpk_EBI.jpg|1lpk: CRYSTAL STRUCTURE OF FXA IN COMPLEX WITH 125. |Image:PDB_1lpz_EBI.jpg|1lpz: CRYSTAL STRUCTURE OF FXA IN COMPLEX WITH 41. |Image:PDB_1lqd_EBI.jpg|1lqd: CRYSTAL STRUCTURE OF FXA IN COMPLEX WITH 45. |Image:PDB_1mq5_EBI.jpg|1mq5: Crystal Structure of 3-chloro-N-[4-chloro-2-[[(4-chlorophenyl)amino]carbonyl]phenyl]-4-[(4-methyl-1-piperazinyl)methyl]-2-thiophenecarboxamide Complexed with Human Factor Xa |Image:PDB_1mq6_EBI.jpg|1mq6: Crystal Structure of 3-chloro-N-[4-chloro-2-[[(5-chloro-2-pyridinyl)amino]carbonyl]-6-methoxyphenyl]-4-[[(4,5-dihydro-2-oxazolyl)methylamino]methyl]-2-thiophenecarboxamide Complexed with Human Factor Xa |Image:PDB_1nfu_EBI.jpg|1nfu: CRYSTAL STRUCTURE OF HUMAN COAGULATION FACTOR XA COMPLEXED WITH RPR132747 |Image:PDB_1nfw_EBI.jpg|1nfw: CRYSTAL STRUCTURE OF HUMAN COAGULATION FACTOR XA COMPLEXED WITH RPR209685 |Image:PDB_1nfx_EBI.jpg|1nfx: CRYSTAL STRUCTURE OF HUMAN COAGULATION FACTOR XA COMPLEXED WITH RPR208944 |Image:PDB_1nfy_EBI.jpg|1nfy: CRYSTAL STRUCTURE OF HUMAN COAGULATION FACTOR XA COMPLEXED WITH RPR200095 |Image:PDB_1p0s_EBI.jpg|1p0s: Crystal Structure of Blood Coagulation Factor Xa in Complex with Ecotin M84R |Image:PDB_1v3x_EBI.jpg|1v3x: Factor Xa in complex with the inhibitor 1-[6-methyl-4,5,6,7-tetrahydrothiazolo(5,4-c)pyridin-2-yl] carbonyl-2-carbamoyl-4-(6-chloronaphth-2-ylsulphonyl)piperazine |Image:PDB_1wu1_EBI.jpg|1wu1: Factor Xa in complex with the inhibitor 4-[(5-chloroindol-2-yl)sulfonyl]-2-(2-methylpropyl)-1-[[5-(pyridin-4-yl) pyrimidin-2-yl]carbonyl]piperazine |Image:PDB_1xka_EBI.jpg|1xka: FACTOR XA COMPLEXED WITH A SYNTHETIC INHIBITOR FX-2212A,(2S)-(3'-AMIDINO-3-BIPHENYLYL)-5-(4-PYRIDYLAMINO)PENTANOIC ACID |Image:PDB_1xkb_EBI.jpg|1xkb: FACTOR XA COMPLEXED WITH A SYNTHETIC INHIBITOR FX-2212A,(2S)-(3'-AMIDINO-3-BIPHENYLYL)-5-(4-PYRIDYLAMINO)PENTANOIC ACID |Image:PDB_1z6e_EBI.jpg|1z6e: Crystal Structure of Factor Xa complexed to Razaxaban |Image:PDB_2bmg_EBI.jpg|2bmg: CRYSTAL STRUCTURE OF FACTOR XA IN COMPLEX WITH 50 |Image:PDB_2boh_EBI.jpg|2boh: CRYSTAL STRUCTURE OF FACTOR XA IN COMPLEX WITH COMPOUND ""1"" |Image:PDB_2bok_EBI.jpg|2bok: FACTOR XA - CATION |Image:PDB_2bq6_EBI.jpg|2bq6: CRYSTAL STRUCTURE OF FACTOR XA IN COMPLEX WITH 21 |Image:PDB_2bq7_EBI.jpg|2bq7: CRYSTAL STRUCTURE OF FACTOR XA IN COMPLEX WITH 43 |Image:PDB_2bqw_EBI.jpg|2bqw: CRYSTAL STRUCTURE OF FACTOR XA IN COMPLEX WITH COMPOUND 45 |Image:PDB_2cji_EBI.jpg|2cji: CRYSTAL STRUCTURE OF A HUMAN FACTOR XA INHIBITOR COMPLEX |Image:PDB_2d1j_EBI.jpg|2d1j: Factor Xa in complex with the inhibitor 2-[[4-[(5-chloroindol-2-yl)sulfonyl]piperazin-1-yl] carbonyl]thieno[3,2-b]pyridine n-oxide |Image:PDB_2fzz_EBI.jpg|2fzz: Factor Xa in complex with the inhibitor 1-(3-amino-1,2-benzisoxazol-5-yl)-6-(2'-(((3r)-3-hydroxy-1-pyrrolidinyl)methyl)-4-biphenylyl)-3-(trifluoromethyl)-1,4,5,6-tetrahydro-7h-pyrazolo[3,4-c]pyridin-7-one |Image:PDB_2g00_EBI.jpg|2g00: Factor Xa in complex with the inhibitor 3-(6-(2'-((dimethylamino)methyl)-4-biphenylyl)-7-oxo-3-(trifluoromethyl)-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridin-1-yl)benzamide |Image:PDB_2gd4_EBI.jpg|2gd4: Crystal Structure of the Antithrombin-S195A Factor Xa-Pentasaccharide Complex |Image:PDB_2h9e_EBI.jpg|2h9e: Crystal Structure of FXa/selectide/NAPC2 ternary complex |Image:PDB_2j2u_EBI.jpg|2j2u: CRYSTAL STRUCTURE OF A HUMAN FACTOR XA INHIBITOR COMPLEX |Image:PDB_2j34_EBI.jpg|2j34: CRYSTAL STRUCTURE OF A HUMAN FACTOR XA INHIBITOR COMPLEX |Image:PDB_2j38_EBI.jpg|2j38: CRYSTAL STRUCTURE OF A HUMAN FACTOR XA INHIBITOR COMPLEX |Image:PDB_2j4i_EBI.jpg|2j4i: CRYSTAL STRUCTURE OF A HUMAN FACTOR XA INHIBITOR COMPLEX |Image:PDB_2j94_EBI.jpg|2j94: CRYSTAL STRUCTURE OF A HUMAN FACTOR XA INHIBITOR COMPLEX |Image:PDB_2j95_EBI.jpg|2j95: CRYSTAL STRUCTURE OF A HUMAN FACTOR XA INHIBITOR COMPLEX |Image:PDB_2uwl_EBI.jpg|2uwl: SELECTIVE AND DUAL ACTION ORALLY ACTIVE INHIBITORS OF THROMBIN AND FACTOR XA |Image:PDB_2uwo_EBI.jpg|2uwo: SELECTIVE AND DUAL ACTION ORALLY ACTIVE INHIBITORS OF THROMBIN AND FACTOR XA |Image:PDB_2uwp_EBI.jpg|2uwp: FACTOR XA INHIBITOR COMPLEX
}}Proteins: coagulation Coagulation factors Coagulation inhibitors Thrombolysis/fibrinolysis Digestive enzymes Coagulation factors: Thrombin · Factor VIIa · Factor IXa · Factor Xa · Factor XIa · Factor XIIa · Kallikrein (PSA, KLK1, KLK2, KLK3, KLK4, KLK5, KLK6, KLK7, KLK8, KLK9, KLK10, KLK11, KLK12, KLK13, KLK14, KLK15)
fibrinolysis: Plasmin · Plasminogen activator (Tissue plasminogen activator · Urinary plasminogen activator)Complement system Other immune system Venombin Other Acrosin · Prolyl endopeptidase · Pronase · Proprotein convertases (1, 2) · Reelin · Subtilisin/Furin · Streptokinase · S1P · Cathepsin (A, G)Categories:- Human proteins
- EC 3.4.21
- Coagulation system
Wikimedia Foundation. 2010.