- Cefazolin
-
Cefazolin 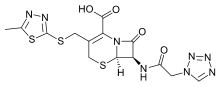
Systematic (IUPAC) name (6R,7R)-3-{[(5-methyl-1,3,4-thiadiazol-2-yl)thio]methyl}-8-oxo-7-[(1H-tetrazol-1-ylacetyl)amino]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid Clinical data Trade names Ancef, Kefzol AHFS/Drugs.com monograph Pregnancy cat. B1(AU) B(US) Legal status ℞ Prescription only Routes Intravenous, intramuscular Pharmacokinetic data Bioavailability NA Metabolism ? Half-life 1.8 hours (given IV)
2 hours (given IM)Excretion Renal, unchanged Identifiers CAS number 25953-19-9 ATC code J01DB04 QJ51DA04 PubChem CID 33255 DrugBank DB01327 ChemSpider 30723 
UNII IHS69L0Y4T 
KEGG D02299 
ChEBI CHEBI:474053 
ChEMBL CHEMBL1435 
Chemical data Formula C14H14N8O4S3 Mol. mass 454.51 g/mol SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Cefazolin (INN), also known as cefazoline or cephazolin, is a first-generation cephalosporin antibiotic.
The drug is usually administrated by either intramuscular injection (injection into a large muscle) or intravenous infusion (intravenous fluid into a vein).
Contents
Indications
Cefazolin is mainly used to treat bacterial infections of the skin. It can also be used to treat moderately severe bacterial infections involving the lung, bone, joint, stomach, blood, heart valve, and urinary tract. It is clinically effective against infections caused by staphylococci and streptococci of Gram-positive bacteria. These organisms are common on normal human skin. Resistance to cefazolin is seen in several species of bacteria.
Adverse effects
Adverse drug reactions from cefazolin are not common. Possible side effects include diarrhea, stomach pain or upset stomach, vomiting, and rash.
Like that of several other cephalosporins, the chemical structure of cefazolin contains an N-methylthiotetrazole (NMTT or 1-MTT) side-chain. As the antibiotic is broken down in the body, it releases free NMTT, which can cause hypoprothrombinemia (likely due to inhibition of the enzyme vitamin K epoxide reductase) and a reaction with ethanol similar to that produced by disulfiram (Antabuse), due to inhibition of aldehyde dehydrogenase.[1]
Brands
Cefazolin is marketed under the following brand names: Ancef, Cefacidal, Cefamezin, Cefrina, Elzogram, Faxilen, Gramaxin, Kefazol, Kefol, Kefzol, Kefzolan, Kezolin, Novaporin, Reflin,Zinol and Zolicef.
References
- ^ Stork CM (2006). "Antibiotics, antifungals, and antivirals". In Nelson LH, Flomenbaum N, Goldfrank LR, Hoffman RL, Howland MD, Lewin NA (eds.). Goldfrank's toxicologic emergencies. New York: McGraw-Hill. pp. 847. ISBN 0-07-143763-0. http://books.google.com/books?id=cvJuLqBxGUcC&pg=PA847. Retrieved 2009-07-03.
External links
Categories:- Cephalosporin antibiotics
- World Health Organization essential medicines
- Tetrazoles
- Thiadiazoles
Wikimedia Foundation. 2010.
