- Pivmecillinam
-
Pivmecillinam 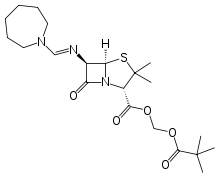
Systematic (IUPAC) name 2,2-dimethylpropanoyloxymethyl (2S,5R,6R)-6-[(azepan-1-ylmethylene)amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate Clinical data AHFS/Drugs.com Micromedex Detailed Consumer Information Pregnancy cat. B(US)
Appears safe in pregnancy[1]Legal status ℞ Prescription only Routes Oral Pharmacokinetic data Bioavailability Low Protein binding 5 to 10% (as mecillinam) Metabolism Pivmecillinam is hydrolyzed to mecillinam Half-life 1 to 3 hours Excretion Renal and biliary, mostly as mecillinam Identifiers CAS number 32886-97-8 
ATC code J01CA08 PubChem CID 36272 ChemSpider 33356 
UNII 1WAM1OQ30B 
KEGG D02889 
Chemical data Formula C21H33N3O5S Mol. mass 439.569 g/mol SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Pivmecillinam (INN) or amdinocillin pivoxil (USAN, trade names Selexid, Penomax and Coactabs) is an orally-active prodrug of mecillinam, an extended-spectrum penicillin antibiotic. Pivmecillinam is the pivaloyloxymethyl ester of mecillinam. Neither drug is available in the United States.[2]
Pivmecillinam is only considered to be active against Gram-negative bacteria, and is used primarily in the treatment of lower urinary tract infections. In the Nordic countries, pivmecillinam has been widely used in that indication since the 1970s. It has been proposed as the first-line drug of choice for empirical treatment of acute cystitis.[1][3] It has also been used to treat paratyphoid fever.[4]
Activity
See also: MecillinamAdverse effects
See also: Beta-lactam antibiotic: Adverse effectsThe adverse effect profile of pivmecillinam is similar to that of other penicillins. The most common side effects of mecillinam use are rash and gastrointestinal upset, including nausea and vomiting.[1][5]
Prodrugs that release pivalic acid when broken down by the body — such as pivmecillinam, pivampicillin and cefditoren pivoxil — have long been known to deplete levels of carnitine.[6][7] This is not due to the drug itself, but to pivalate, which is mostly removed from the body by forming a conjugate with carnitine. Although short-term use of these drugs can cause a marked decrease in blood levels of carnitine,[8] it is unlikely to be of clinical significance;[7] long-term use, however, appears problematic and is not recommended.[7][9][10]
References
- ^ a b c Nicolle LE (August 2000). "Pivmecillinam in the treatment of urinary tract infections". J Antimicrob Chemother. 46 Suppl A (90001): 35–39. doi:10.1093/jac/46.suppl_1.35. PMID 10969050. http://jac.oxfordjournals.org/cgi/pmidlookup?view=long&pmid=10969050.
- ^ Pham P, Bartlett JG (August 28, 2008). "Amdinocillin (Mecillinam)". Point-of-Care Information Technology ABX Guide. Johns Hopkins University. http://prod.hopkins-abxguide.org/antibiotics/antibacterial/pcn_others/amdinocillin__mecillinam_.html. Retrieved on August 31, 2008. Freely available with registration.
- ^ Graninger W (October 2003). "Pivmecillinam—therapy of choice for urinary tract infection". Int J Antimicrob Agents. 22 Suppl 2: 73–8. doi:10.1016/S0924-8579(03)00235-8. PMID 14527775. http://linkinghub.elsevier.com/retrieve/pii/S0924857903002358.
- ^ Tanphaichitra D, Srimuang S, Chiaprasittigul P, Menday P, Christensen OE (1984). "The combination of pivmecillinam and pivampicillin in the treatment of enteric fever". Infection 12 (6): 381–3. doi:10.1007/BF01645219. PMID 6569851.
- ^ "Selexid Tablets". electronic Medicines Compendium. June 5, 2008. http://emc.medicines.org.uk/emc/assets/c/html/DisplayDoc.asp?DocumentID=2566. Retrieved on August 31, 2008.
- ^ Holme E, Greter J, Jacobson CE, et al. (August 1989). "Carnitine deficiency induced by pivampicillin and pivmecillinam therapy". Lancet 2 (8661): 469–73. doi:10.1016/S0140-6736(89)92086-2. PMID 2570185. http://linkinghub.elsevier.com/retrieve/pii/S0140-6736(89)92086-2.
- ^ a b c Brass EP (December 2002). "Pivalate-generating prodrugs and carnitine homeostasis in man". Pharmacol Rev 54 (4): 589–98. doi:10.1124/pr.54.4.589. PMID 12429869. http://pharmrev.aspetjournals.org/cgi/pmidlookup?view=long&pmid=12429869.
- ^ Abrahamsson K, Holme E, Jodal U, Lindstedt S, Nordin I (June 1995). "Effect of short-term treatment with pivalic acid containing antibiotics on serum carnitine concentration—a risk irrespective of age". Biochem. Mol. Med. 55 (1): 77–9. doi:10.1006/bmme.1995.1036. PMID 7551831. http://linkinghub.elsevier.com/retrieve/pii/S1077315085710368.
- ^ Holme E, Jodal U, Linstedt S, Nordin I (September 1992). "Effects of pivalic acid-containing prodrugs on carnitine homeostasis and on response to fasting in children". Scand J Clin Lab Invest 52 (5): 361–72. doi:10.3109/00365519209088371. PMID 1514015.
- ^ Makino Y, Sugiura T, Ito T, Sugiyama N, Koyama N (September 2007). "Carnitine-associated encephalopathy caused by long-term treatment with an antibiotic containing pivalic acid". Pediatrics 120 (3): e739–41. doi:10.1542/peds.2007-0339. PMID 17724113.
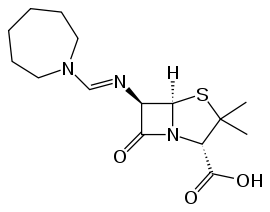 Skeletal formula of mecillinam, the active moiety of pivmecillinam
Skeletal formula of mecillinam, the active moiety of pivmecillinam Categories:
Categories:- Beta-lactam antibiotics
- Azepanes
- Prodrugs
Wikimedia Foundation. 2010.
