- Mezlocillin
-
Mezlocillin 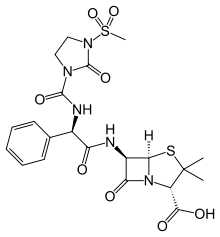
Systematic (IUPAC) name (2S,5R,6R)-3,3-dimethyl-6-[[(2R)-2-[(3-methylsulfonyl-
2-oxo-imidazolidine-1-carbonyl)amino]-2-phenyl-acetyl]
amino]-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-
carboxylic acidClinical data AHFS/Drugs.com Micromedex Detailed Consumer Information Pregnancy cat. B Legal status ? Routes Intravenous, intramuscular Pharmacokinetic data Protein binding 16–59% Metabolism Hepatic (20–30%) Half-life 1.3–4.4 hours Excretion Renal (50%) and biliary Identifiers CAS number 51481-65-3 
ATC code J01CA10 PubChem CID 656511 DrugBank APRD01113 ChemSpider 570894 
UNII OH2O403D1G 
KEGG D05021 
ChEBI CHEBI:6919 
ChEMBL CHEMBL1731 
Chemical data Formula C21H25N5O8S2 Mol. mass 539.584 g/mol SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Mezlocillin is a broad-spectrum penicillin antibiotic. It is active against both Gram-negative and some Gram-positive bacteria. Unlike most other extended spectrum penicillins, it is excreted by the liver, therefore it is useful for biliary tract infections, such as ascending colangitis.
Contents
Mechanism of action
Main article: Beta-lactam antibioticLike all other beta-lactam antibiotics, mezlocillin inhibits the third and last stage of bacterial cell wall synthesis by binding to penicillin binding proteins. This ultimately leads to cell lysis.
Susceptible organisms
Gram-negative
- Bacteroides spp., including B. fragilis
- Enterobacter spp.
- Escherichia coli
- Haemophilus influenzae
- Klebsiella species
- Morganella morganii
- Neisseria gonorrhoeae
- Proteus mirabilis
- Proteus vulgaris
- Providencia rettgeri
- Pseudomonas spp., including P. aeruginosa
- Serratia marcescens
Gram-positive
- Enterococcus faecalis
- Peptococcus spp.
- Peptostreptococcus spp.
External links
- DDB 30377
- -288358398 at GPnotebook
- Duke
- Kristof R, Clusmann H, Koehler W, Fink K, Schramm J (1998). "Treatment of accidental high dose intraventricular mezlocillin application by cerebrospinal fluid exchange.". J Neurol Neurosurg Psychiatry 64 (3): 379–81. doi:10.1136/jnnp.64.3.379. PMC 2170014. PMID 9527154. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2170014.
- McCormick P, Greenslade L, Kibbler C, Chin J, Burroughs A, McIntyre N (1997). "A prospective randomized trial of ceftazidime versus netilmicin plus mezlocillin in the empirical therapy of presumed sepsis in cirrhotic patients.". Hepatology 25 (4): 833–6. doi:10.1002/hep.510250408. PMID 9096584.
- Rohde B, Werner U, Hickstein H, Ehmcke H, Drewelow B (1997). "Pharmacokinetics of mezlocillin and sulbactam under continuous veno-venous hemodialysis (CVVHD) in intensive care patients with acute renal failure.". Eur J Clin Pharmacol 53 (2): 111–5. doi:10.1007/s002280050347. PMID 9403281.
PenemsCefazolin# • Cefacetrile • Cefadroxil • Cefalexin • Cefaloglycin • Cefalonium • Cefaloridine • Cefalotin • Cefapirin • Cefatrizine • Cefazedone • Cefazaflur • Cefradine • Cefroxadine • CeftezoleCefaclor • Cefamandole • Cefminox • Cefonicid • Ceforanide • Cefotiam • Cefprozil • Cefbuperazone • Cefuroxime • Cefuzonam • cephamycin (Cefoxitin, Cefotetan, Cefmetazole) • carbacephem (Loracarbef)Cefixime# • Ceftriaxone# • antipseudomonal (Ceftazidime# • Cefoperazone) • Cefcapene • Cefdaloxime • Cefdinir • Cefditoren • Cefetamet • Cefmenoxime • Cefodizime • Cefotaxime • Cefpimizole • Cefpiramide • Cefpodoxime • Cefsulodin • Cefteram • Ceftibuten • Ceftiolene • Ceftizoxime • oxacephem (Flomoxef, Latamoxef ‡)4th (antips-)Ceftobiprole • Ceftaroline fosamilCombinationsOther polymyxins/detergent (Colistin, Polymyxin B) • depolarizing (Daptomycin) • hydrolyze NAM-NAG (Lysozyme) • GramicidinCategories:- Antibiotic stubs
- Beta-lactam antibiotics
- Enantiopure drugs
- Imidazolidinones
Wikimedia Foundation. 2010.
