- Monoamine oxidase B
-
Monoamine oxidase B, also known as MAOB, is a protein that in humans is encoded by the MAOB gene.
The protein encoded by this gene belongs to the flavin monoamine oxidase family. It is an enzyme located in the mitochondrial outer membrane. It catalyzes the oxidative deamination of biogenic and xenobiotic amines and plays an important role in the metabolism of neuroactive and vasoactive amines in the central nervous system and peripheral tissues. This protein preferentially degrades benzylamine and phenylethylamine.[1] Like MAOA, it also degrades dopamine.
Contents
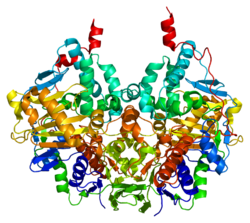
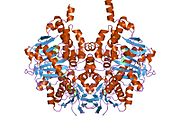
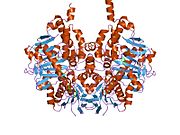
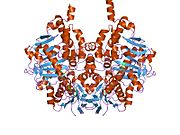
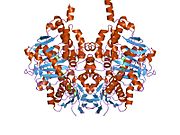
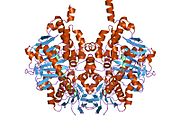
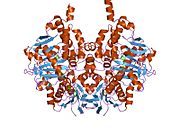
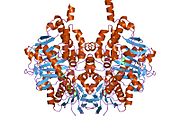
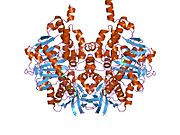
Structure
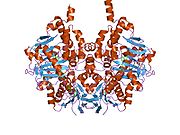
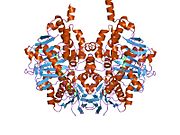
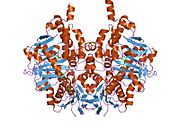
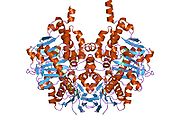
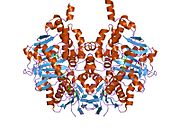
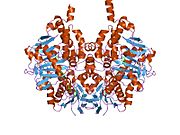
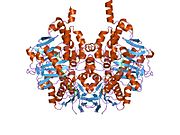
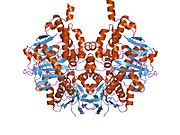
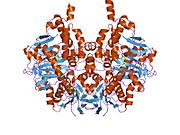
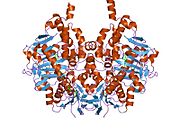
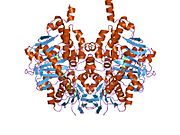
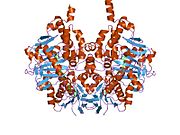
Edmondson et al. described structural features of the human enzyme: it has a hydrophobic bipartite elongated cavity that (for the "open" conformation) occupies a combined volume close to 700 Å3. hMAO-A has a single cavity that exhibits a rounder shape and is larger in volume than the "substrate cavity" of hMAO-B.[2]
The first cavity of hMAO-B has been termed the entrance cavity (290 Å3), the second substrate cavity or active site cavity (~390 Å3) – between both an isoleucine199 side-chain serves as a gate. Depending on the substrate or bound inhibitor, it can exist in either an open or a closed form, which has been shown to be important in defining the inhibitor specificity of hMAO B. At the end of the substrate cavity is the FAD coenzyme with sites for favorable amine binding about the flavin involving two nearly parallel tyrosyl (398 and 435) residues that form what has been termed an aromatic cage.[2]
Differences between MAOA and MAOB
In general, Monoamine Oxidase A (MAOA) prefers to metabolize norepinephrine (NE), serotonin (5-HT), and Dopamine (DA) (and other less clinically relevant chemicals). Monoamine Oxidase B, on the other hand, prefers to metabolize Dopamine (DA) (and other less clinically relevant chemicals).The differences between the substrate selectivity of the two enzymes are utilized clinically when treating specific disorders: Monoamine Oxidase A inhibitors have been used in the treatment of depression, and Monoamine Oxidase B inhibitors are used in the treatment of Parkinson's Disease.[citation needed]
Selective inhibitors
Species-dependent divergences may hamper the extrapolation of inhibitor potencies.[3]
Reversible
Natural
- Geiparvarin[4]
- Desmethoxyyangonin,[5] a constituent of kava-kava extract; modest affinity
- Catechin and epicatechin
Synthetic
- Safinamide and analogs[9]
- 5H-Indeno[1,2-c]pyridazin-5-ones[3][10][6] (see 3d model)
- Substituted chalcones[11]
- 2-(N-Methyl-N-benzylaminomethyl)-1H-pyrrole[12]
- 1-(4-Arylthiazol-2-yl)-2-(3-methylcyclohexylidene)hydrazine[13]
- 2-Thiazolylhydrazone[14]
- 3,5-Diaryl pyrazole[15]
- Pyrazoline derivatives[16][17]
- Coumarins[8] and #C19*[3] (see 3d model)
- Phenylcoumarins, extremly subtype selective[7] and further analogs[18][19][20] (see 3d model)
- Chromone-3-phenylcarboxamides[21]
- Isatins[22]
- Phthalimides[23]
- 8-Benzyloxycaffeines[24][25] and CSC analogs[26]
- (E,E)-8-(4-phenylbutadien-1-yl)caffeines,[27] with A2A antagonistic component
Irreversible (covalent)
- Selegiline (Eldepryl, Zelapar, Emsam)
- Rasagiline (Azilect)
References
- ^ "Entrez Gene: MAOB monoamine oxidase B". http://www.ncbi.nlm.nih.gov/sites/entrez?Db=gene&Cmd=ShowDetailView&TermToSearch=4129.
- ^ a b Edmondson DE, Binda C, Mattevi A (2007). "STRUCTURAL INSIGHTS INTO THE MECHANISM OF AMINE OXIDATION BY MONOAMINE OXIDASES A AND B". Archives of Biochemistry and Biophysics 464 (2): 269–76. doi:10.1016/j.abb.2007.05.006. PMC 1993809. PMID 17573034. http://linkinghub.elsevier.com/retrieve/pii/S0003-9861(07)00252-4.
- ^ a b c Novaroli L, Daina A, Favre E, et al. (October 2006). "Impact of species-dependent differences on screening, design, and development of MAO B inhibitors". Journal of Medicinal Chemistry 49 (21): 6264–72. doi:10.1021/jm060441e. PMID 17034132.
- ^ Carotti A, Carrieri A, Chimichi S, Boccalini M, Cosimelli B, Gnerre C, Carotti A, Carrupt PA, Testa B (December 2002). "Natural and synthetic geiparvarins are strong and selective MAO-B inhibitors. Synthesis and SAR studies". Bioorg. Med. Chem. Lett. 12 (24): 3551–5. doi:10.1016/S0960-894X(02)00798-9. PMID 12443774.
- ^ Uebelhack R, Franke L, Schewe HJ (September 1998). "Inhibition of platelet MAO-B by kava pyrone-enriched extract from Piper methysticum Forster (kava-kava)". Pharmacopsychiatry 31 (5): 187–92. doi:10.1055/s-2007-979325. PMID 9832350.
- ^ a b compound #2d, Frédérick R, Dumont W, Ooms F, Aschenbach L, Van der Schyf CJ, Castagnoli N, Wouters J, Krief A (June 2006). "Synthesis, structural reassignment, and biological activity of type B MAO inhibitors based on the 5H-indeno[1,2-c]pyridazin-5-one core". J. Med. Chem. 49 (12): 3743–7. doi:10.1021/jm051091j. PMID 16759116.
- ^ a b compound #2, Matos MJ, Vazquez-Rodriguez S, Uriarte E, Santana L, Viña D (July 2011). "MAO inhibitory activity modulation: 3-Phenylcoumarins versus 3-benzoylcoumarins". Bioorg. Med. Chem. Lett. 21 (14): 4224–7. doi:10.1016/j.bmcl.2011.05.074. PMID 21684743.
- ^ a b compound #41, Catto M, Nicolotti O, Leonetti F, Carotti A, Favia AD, Soto-Otero R, Méndez-Alvarez E, Carotti A (2006). "Structural insights into monoamine oxidase inhibitory potency and selectivity of 7-substituted coumarins from ligand- and target-based approaches". Journal of Medicinal Chemistry 49 (16): 4912–25. doi:10.1021/jm060183l. PMID 16884303.
- ^ Leonetti F, Capaldi C, Pisani L, Nicolotti O, Muncipinto G, Stefanachi A, Cellamare S, Caccia C, Carotti A (October 2007). "Solid-phase synthesis and insights into structure-activity relationships of safinamide analogues as potent and selective inhibitors of type B monoamine oxidase". Journal of Medicinal Chemistry 50 (20): 4909–16. doi:10.1021/jm070725e. PMID 17824599.
- ^ Carotti A, Catto M, Leonetti F, Campagna F, Soto-Otero R, Méndez-Alvarez E, Thull U, Testa B, Altomare C (November 2007). "Synthesis and monoamine oxidase inhibitory activity of new pyridazine-, pyrimidine- and 1,2,4-triazine-containing tricyclic derivatives". Journal of Medicinal Chemistry 50 (22): 5364–71. doi:10.1021/jm070728r. PMID 17910428.
- ^ Chimenti F, Fioravanti R, Bolasco A, et al. (May 2009). "Chalcones: a valid scaffold for monoamine oxidases inhibitors". J. Med. Chem. 52 (9): 2818–24. doi:10.1021/jm801590u. PMID 19378991.
- ^ compound #21, Silvestri R, La Regina G, De Martino G, Artico M, Befani O, Palumbo M, Agostinelli E, Turini P (March 2003). "Simple, potent, and selective pyrrole inhibitors of monoamine oxidase types A and B". J. Med. Chem. 46 (6): 917–20. doi:10.1021/jm0256124. PMID 12620068.
- ^ compound # (R)-8b, Chimenti F, Secci D, Bolasco A, Chimenti P, Granese A, Carradori S, Yáñez M, Orallo F, Sanna ML, Gallinella B, Cirilli R (September 2010). "Synthesis, stereochemical separation, and biological evaluation of selective inhibitors of human MAO-B: 1-(4-arylthiazol-2-yl)-2-(3-methylcyclohexylidene)hydrazines". J. Med. Chem. 53 (17): 6516–20. doi:10.1021/jm100120s. PMID 20715818.
- ^ compound #18, Chimenti F, Maccioni E, Secci D, Bolasco A, Chimenti P, Granese A, Befani O, Turini P, Alcaro S, Ortuso F, Cardia MC, Distinto S (February 2007). "Selective inhibitory activity against MAO and molecular modeling studies of 2-thiazolylhydrazone derivatives". J. Med. Chem. 50 (4): 707–12. doi:10.1021/jm060869d. PMID 17253676.
- ^ compound #3g, Chimenti F, Fioravanti R, Bolasco A, Manna F, Chimenti P, Secci D, Befani O, Turini P, Ortuso F, Alcaro S (February 2007). "Monoamine oxidase isoform-dependent tautomeric influence in the recognition of 3,5-diaryl pyrazole inhibitors". J. Med. Chem. 50 (3): 425–8. doi:10.1021/jm060868l. PMID 17266193.
- ^ compound #(S)-1, Chimenti F, Maccioni E, Secci D, Bolasco A, Chimenti P, Granese A, Befani O, Turini P, Alcaro S, Ortuso F, Cirilli R, La Torre F, Cardia MC, Distinto S (November 2005). "Synthesis, molecular modeling studies, and selective inhibitory activity against monoamine oxidase of 1-thiocarbamoyl-3,5-diaryl-4,5-dihydro-(1H)- pyrazole derivatives". J. Med. Chem. 48 (23): 7113–22. doi:10.1021/jm040903t. PMID 16279769.
- ^ Mishra N, Sasmal D (April 2011). "Development of selective and reversible pyrazoline based MAO-B inhibitors: virtual screening, synthesis and biological evaluation". Bioorg. Med. Chem. Lett. 21 (7): 1969–73. doi:10.1016/j.bmcl.2011.02.030. PMID 21377879.
- ^ Matos MJ, Viña D, Janeiro P, Borges F, Santana L, Uriarte E (September 2010). "New halogenated 3-phenylcoumarins as potent and selective MAO-B inhibitors". Bioorg. Med. Chem. Lett. 20 (17): 5157–60. doi:10.1016/j.bmcl.2010.07.013. PMID 20659799.
- ^ Matos MJ, Viña D, Picciau C, Orallo F, Santana L, Uriarte E (September 2009). "Synthesis and evaluation of 6-methyl-3-phenylcoumarins as potent and selective MAO-B inhibitors". Bioorg. Med. Chem. Lett. 19 (17): 5053–5. doi:10.1016/j.bmcl.2009.07.039. PMID 19628387.
- ^ Matos MJ, Viña D, Quezada E, Picciau C, Delogu G, Orallo F, Santana L, Uriarte E (June 2009). "A new series of 3-phenylcoumarins as potent and selective MAO-B inhibitors". Bioorg. Med. Chem. Lett. 19 (12): 3268–70. doi:10.1016/j.bmcl.2009.04.085. PMID 19423346.
- ^ compound #9, #12, Gaspar A, Reis J, Fonseca A, Milhazes N, Viña D, Uriarte E, Borges F (January 2011). "Chromone 3-phenylcarboxamides as potent and selective MAO-B inhibitors". Bioorg. Med. Chem. Lett. 21 (2): 707–9. doi:10.1016/j.bmcl.2010.11.128. PMID 21194943.
- ^ compound #9i, Manley-King CI, Bergh JJ, Petzer JP (January 2011). "Inhibition of monoamine oxidase by selected C5- and C6-substituted isatin analogues". Bioorg. Med. Chem. 19 (1): 261–74. doi:10.1016/j.bmc.2010.11.028. PMID 21134756.
- ^ compound #5c, Manley-King CI, Bergh JJ, Petzer JP (August 2011). "Inhibition of monoamine oxidase by C5-substituted phthalimide analogues". Bioorg. Med. Chem. 19 (16): 4829–40. doi:10.1016/j.bmc.2011.06.070. PMID 21778064.
- ^ Strydom B, Bergh JJ, Petzer JP (August 2011). "8-Aryl- and alkyloxycaffeine analogues as inhibitors of monoamine oxidase". Eur J Med Chem 46 (8): 3474–85. doi:10.1016/j.ejmech.2011.05.014. PMID 21621312.
- ^ Strydom B, Malan SF, Castagnoli N, Bergh JJ, Petzer JP (February 2010). "Inhibition of monoamine oxidase by 8-benzyloxycaffeine analogues". Bioorg. Med. Chem. 18 (3): 1018–28. doi:10.1016/j.bmc.2009.12.064. PMID 20093036.
- ^ Vlok N, Malan SF, Castagnoli N, Bergh JJ, Petzer JP (May 2006). "Inhibition of monoamine oxidase B by analogues of the adenosine A2A receptor antagonist (E)-8-(3-chlorostyryl)caffeine (CSC)". Bioorg. Med. Chem. 14 (10): 3512–21. doi:10.1016/j.bmc.2006.01.011. PMID 16442801.
- ^ Pretorius J, Malan SF, Castagnoli N, Bergh JJ, Petzer JP (September 2008). "Dual inhibition of monoamine oxidase B and antagonism of the adenosine A(2A) receptor by (E,E)-8-(4-phenylbutadien-1-yl)caffeine analogues". Bioorganic & Medicinal Chemistry 16 (18): 8676–84. doi:10.1016/j.bmc.2008.07.088. PMID 18723354.
Further reading
- Edmondson DE, Binda C, Mattevi A (2004). "The FAD binding sites of human monoamine oxidases A and B". Neurotoxicology 25 (1–2): 63–72. doi:10.1016/S0161-813X(03)00114-1. PMID 14697881.
- Kumar MJ, Andersen JK (2004). "Perspectives on MAO-B in aging and neurological disease: where do we go from here?". Mol. Neurobiol. 30 (1): 77–89. doi:10.1385/MN:30:1:077. PMID 15247489.
- Ghozlan A, Munnich A (2004). "[MAOB: a modifier gene in phenylketonuria?] [MAOB: a modifier gene in phenylketonuria?]" (in French). Med Sci (Paris) 20 (10): 929–32. doi:10.1051/medsci/20042010929. PMID 15461973.
- Zhu QS, Grimsby J, Chen K, Shih JC (1992). "Promoter organization and activity of human monoamine oxidase (MAO) A and B genes". J. Neurosci. 12 (11): 4437–46. PMID 1432104.
- Grimsby J, Chen K, Wang LJ, et al. (1991). "Human monoamine oxidase A and B genes exhibit identical exon-intron organization". Proc. Natl. Acad. Sci. U.S.A. 88 (9): 3637–41. doi:10.1073/pnas.88.9.3637. PMC 51507. PMID 2023912. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=51507.
- Bach AW, Lan NC, Johnson DL, et al. (1988). "cDNA cloning of human liver monoamine oxidase A and B: molecular basis of differences in enzymatic properties". Proc. Natl. Acad. Sci. U.S.A. 85 (13): 4934–8. doi:10.1073/pnas.85.13.4934. PMC 280552. PMID 3387449. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=280552.
- Kochersperger LM, Parker EL, Siciliano M, et al. (1987). "Assignment of genes for human monoamine oxidases A and B to the X chromosome". J. Neurosci. Res. 16 (4): 601–16. doi:10.1002/jnr.490160403. PMID 3540317.
- Wyatt RJ, Murphy DL, Belmaker R, et al. (1973). "Reduced monoamine oxidase activity in platelets: a possible genetic marker for vulnerability to schizophrenia". Science 179 (4076): 916–8. doi:10.1126/science.179.4076.916. PMID 4687789.
- Goldin LR, Gershon ES, Lake CR, et al. (1982). "Segregation and linkage studies of plasma dopamine-beta-hydroxylase (DBH), erythrocyte catechol-O-methyltransferase (COMT), and platelet monoamine oxidase (MAO): possible linkage between the ABO locus and a gene controlling DBH activity". Am. J. Hum. Genet. 34 (2): 250–62. PMC 1685293. PMID 6951409. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1685293.
- Denney RM, Fritz RR, Patel NT, Abell CW (1982). "Human liver MAO-A and MAO-B separated by immunoaffinity chromatography with MAO-B-specific monoclonal antibody". Science 215 (4538): 1400–3. doi:10.1126/science.7063850. PMID 7063850.
- Raddatz R, Parini A, Lanier SM (1996). "Imidazoline/guanidinium binding domains on monoamine oxidases. Relationship to subtypes of imidazoline-binding proteins and tissue-specific interaction of imidazoline ligands with monoamine oxidase B". J. Biol. Chem. 270 (46): 27961–8. doi:10.1074/jbc.270.46.27961. PMID 7499273.
- Woo JC, Silverman RB (1994). "Observation of two different chromophores in the resting state of monoamine oxidase B by fluorescence spectroscopy". Biochem. Biophys. Res. Commun. 202 (3): 1574–8. doi:10.1006/bbrc.1994.2111. PMID 8060341.
- Wu HF, Chen K, Shih JC (1993). "Site-directed mutagenesis of monoamine oxidase A and B: role of cysteines". Mol. Pharmacol. 43 (6): 888–93. PMID 8316221.
- Chen K, Wu HF, Shih JC (1993). "The deduced amino acid sequences of human platelet and frontal cortex monoamine oxidase B are identical". J. Neurochem. 61 (1): 187–90. doi:10.1111/j.1471-4159.1993.tb03554.x. PMID 8515265.
- Fowler JS, Volkow ND, Wang GJ, et al. (1996). "Inhibition of monoamine oxidase B in the brains of smokers". Nature 379 (6567): 733–6. doi:10.1038/379733a0. PMID 8602220.
- Lenders JW, Eisenhofer G, Abeling NG, et al. (1996). "Specific genetic deficiencies of the A and B isoenzymes of monoamine oxidase are characterized by distinct neurochemical and clinical phenotypes". J. Clin. Invest. 97 (4): 1010–9. doi:10.1172/JCI118492. PMC 507147. PMID 8613523. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=507147.
- Cesura AM, Gottowik J, Lahm HW, et al. (1996). "Investigation on the structure of the active site of monoamine oxidase-B by affinity labeling with the selective inhibitor lazabemide and by site-directed mutagenesis". Eur. J. Biochem. 236 (3): 996–1002. doi:10.1111/j.1432-1033.1996.00996.x. PMID 8665924.
- Bonaldo MF, Lennon G, Soares MB (1997). "Normalization and subtraction: two approaches to facilitate gene discovery". Genome Res. 6 (9): 791–806. doi:10.1101/gr.6.9.791. PMID 8889548.
- Saura J, Bleuel Z, Ulrich J, et al. (1997). "Molecular neuroanatomy of human monoamine oxidases A and B revealed by quantitative enzyme radioautography and in situ hybridization histochemistry". Neuroscience 70 (3): 755–74. doi:10.1016/S0306-4522(96)83013-2. PMID 9045087.
- Checkoway H, Franklin GM, Costa-Mallen P, et al. (1998). "A genetic polymorphism of MAO-B modifies the association of cigarette smoking and Parkinson's disease". Neurology 50 (5): 1458–61. PMID 9596006.
PDB gallery 1gos: HUMAN MONOAMINE OXIDASE B1oj9: HUMAN MONOAMINE OXIDASE B IN COMPLEX WITH 1,4-DIPHENYL-2-BUTENE1oja: HUMAN MONOAMINE OXIDASE B IN COMPLEX WITH ISATIN1ojb: HUMAN MONOAMINE OXIDASE B IN COMPLEX WITH TRANYLCYPROMINE1ojc: HUMAN MONOAMINE OXIDASE B IN COMPLEX WITH N-(2-AMINOETHYL)-P-CHLOROBENZAMIDE1ojd: HUMAN MONOAMINE OXIDASE B IN COMPLEX WITH LAURYLDIMETHYLAMINE-N-OXIDE (LDAO)1s2q: Crystal structure of MAOB in complex with N-propargyl-1(R)-aminoindan (Rasagiline)1s2y: Crystal structure of MAOB in complex with N-propargyl-1(S)-aminoindan1s3b: Crystal structure of MAOB in complex with N-methyl-N-propargyl-1(R)-aminoindan1s3e: Crystal structure of MAOB in complex with 6-hydroxy-N-propargyl-1(R)-aminoindan2bk3: HUMAN MONOAMINE OXIDASE B IN COMPLEX WITH FARNESOL2bk4: HUMAN MONOAMINE OXIDASE B: I199F MUTANT IN COMPLEX WITH RASAGILINE2bk5: HUMAN MONOAMINE OXIDASE B: I199F MUTANT IN COMPLEX WITH ISATIN2byb: HUMAN MONOAMINE OXIDASE B IN COMPLEX WITH DEPRENYL2c64: MAO INHIBITION BY RASAGILINE ANALOGUES2c65: MAO INHIBITION BY RASAGILINE ANALOGUES2c66: MAO INHIBITION BY RASAGILINE ANALOGUES2c67: MAO INHIBITION BY RASAGILINE ANALOGUES2c70: FUNCTIONAL ROLE OF THE AROMATIC CAGE IN HUMAN MONOAMINE OXIDASE B: STRUCTURES AND CATALYTIC PROPERTIES OF TYR435 MUTANT PROTEINS2c72: FUNCTIONAL ROLE OF THE AROMATIC CAGE IN HUMAN MONOAMINE OXIDASE B: STRUCTURES AND CATALYTIC PROPERTIES OF TYR435 MUTANT PROTEINS2c73: FUNCTIONAL ROLE OF THE AROMATIC CAGE IN HUMAN MONOAMINE OXIDASE B: STRUCTURES AND CATALYTIC PROPERTIES OF TYR435 MUTANT PROTEINS2c75: FUNCTIONAL ROLE OF THE AROMATIC CAGE IN HUMAN MONOAMINE OXIDASE B: STRUCTURES AND CATALYTIC PROPERTIES OF TYR435 MUTANT PROTEINS2c76: FUNCTIONAL ROLE OF THE AROMATIC CAGE IN HUMAN MONOAMINE OXIDASE B: STRUCTURES AND CATALYTIC PROPERTIES OF TYR435 MUTANT PROTEINSCH-NH2 oxidoreductases (EC 1.4) - primarily amino acid oxidoreductases 1.4.1: NAD/NADP acceptor 1.4.3: oxygen acceptor 1.4.4: disulfide acceptor 1.4.99: other acceptors B enzm: 1.1/2/3/4/5/6/7/8/10/11/13/14/15-18, 2.1/2/3/4/5/6/7/8, 2.7.10, 2.7.11-12, 3.1/2/3/4/5/6/7, 3.1.3.48, 3.4.21/22/23/24, 4.1/2/3/4/5/6, 5.1/2/3/4/99, 6.1-3/4/5-6 Mitochondrial proteins Outer membrane Intermembrane space Inner membrane oxidative phosphorylation (Coenzyme Q - cytochrome c reductase, Cytochrome c, NADH dehydrogenase, Succinate dehydrogenase)
pyrimidine metabolism (Dihydroorotate dehydrogenase)
mitochondrial shuttle (Malate-aspartate shuttle, Glycerol phosphate shuttle)
other (Glutamate aspartate transporter, Glycerol-3-phosphate dehydrogenase, ATP synthase, Carnitine palmitoyltransferase II, Uncoupling protein)Matrix citric acid cycle (Citrate synthase, Aconitase, Isocitrate dehydrogenase, Oxoglutarate dehydrogenase, Succinyl coenzyme A synthetase, Fumarase, Malate dehydrogenase)
anaplerotic reactions (Aspartate transaminase, Glutamate dehydrogenase, Pyruvate dehydrogenase complex)
urea cycle (Carbamoyl phosphate synthetase I, Ornithine transcarbamylase, N-Acetylglutamate synthase)
alcohol metabolism (ALDH2)
PMPCBOther/to be sorted Mitochondrial DNA Complex I (MT-ND1, MT-ND2, MT-ND3, MT-ND4, MT-ND4L, MT-ND5, MT-ND6) - Complex III (MT-CYB) - Complex IV (MT-CO1, MT-CO2, MT-CO3)
ATP synthase (MT-ATP6, MT-ATP8)
tRNA (MT-TA, MT-TC, MT-TD, MT-TE, MT-TF, MT-TG, MT-TH, MT-TI, MT-TK, MT-TL1, MT-TL2, MT-TM, MT-TN, MT-TP, MT-TQ, MT-TR, MT-TS1, MT-TS2, MT-TT, MT-TV, MT-TW, MT-TY)see also mitochondrial diseases
B strc: edmb (perx), skel (ctrs), epit, cili, mito, nucl (chro)monoamine glutamate→GABAanabolism: Glutamate decarboxylase
catabolism: 4-aminobutyrate aminotransferase · 4-aminobutyrate transaminasearginine→NO choline→Acetylcholine anabolism: Choline acetyltransferase
catabolism: Cholinesterase (Acetylcholinesterase, Butyrylcholinesterase)Categories:- Human proteins
- Chromosome X gene stubs
Wikimedia Foundation. 2010.