- Tumor necrosis factors
-
TNF(Tumour Necrosis Factor) family [[Image: |220px]] Model of TNF-alpha, produced by M. musculus, based on PDB structure 2TNF (1.4Å Resolution). Different colors represent different monomers. Baeyens, KJ et al. (1999). [1] Generated in FirstGlance Jmol.
Model of TNF-alpha, produced by M. musculus, based on PDB structure 2TNF (1.4Å Resolution). Different colors represent different monomers. Baeyens, KJ et al. (1999). [1] Generated in FirstGlance Jmol.
Identifiers Symbol TNF Pfam PF00229 InterPro IPR006052 PROSITE PDOC00224 SCOP 1tnf Available protein structures: Pfam structures PDB RCSB PDB; PDBe PDBsum structure summary Tumor necrosis factors (or the TNF-family) refers to a group of cytokines family that can cause cell death (apoptosis). The first two members of the family to be identified were:
- Tumor necrosis factor-alpha (TNF-α) is the best-known member of this class, and sometimes referred to when the term "tumor necrosis factor" is used. TNF-α is a monocyte-derived cytotoxin that has been implicated in tumour regression, septic shock and cachexia.[2][3] The protein is synthesised as a prohormone with an unusually long and atypical signal sequence, which is absent from the mature secreted cytokine.[4] A short hydrophobic stretch of amino acids serves to anchor the prohormone in lipid bilayers.[5] Both the mature protein and a partially-processed form of the hormone can be secreted after cleavage of the propeptide.[5]
- Tumor necrosis factor-beta (TNF-β), also known as lymphotoxin is a cytokine that is inhibited by interleukin 10[6]
Contents
Family members
Nineteen cytokines have been identified as part of the TNF family on the basis of sequence, functional, and structural similarities.[7] They include:[8][9][10]
- Tumor Necrosis Factor (TNF) (also known as cachectin or TNF-alpha)[11][12] is a cytokine that has a wide variety of functions. It can cause cytolysis of certain tumor cell lines; it is involved in the induction of cachexia; it is a potent pyrogen, causing fever by direct action or by stimulation of interleukin-1 secretion; it can stimulate cell proliferation and induce cell differentiation under certain conditions.
- Lymphotoxin-alpha (LT-alpha) and lymphotoxin-beta (LT-beta), two related cytokines produced by lymphocytes that are cytotoxic for a wide range of tumor cells in vitro and in vivo.[13]
- T cell antigen gp39 (CD40L), a cytokine that seems to be important in B-cell development and activation.
- CD27L, a cytokine that plays a role in T-cell activation. It induces the proliferation of costimulated T cells and enhances the generation of cytolytic T cells.
- CD30L, a cytokine that induces proliferation of T cells.
- FASL, a cytokine involved in cell death.[14]
- 4-1BBL, a inducible T cell surface molecule that contributes to T-cell stimulation.
- OX40L, a cytokine that co-stimulates T cell proliferation and cytokine production.[15]
- TNF-related apoptosis inducing ligand (TRAIL), a cytokine that induces apoptosis.[16]
All these cytokines seem to form homotrimeric (or heterotrimeric in the case of LT-alpha/beta) complexes that are recognized by their specific receptors. Strong hydrogen bonds between the monomers stabilizes the tertiary structure. One such example is the Asn34-Arg82 hydrogen bond in the M. musculus TNF alpha[17].
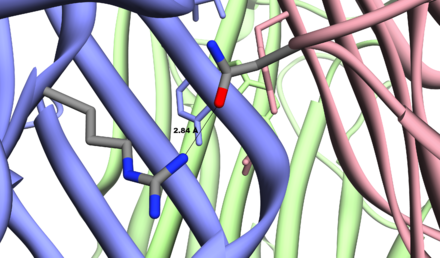 Model of hydrogen bond between Asn34 of subunit A and Arg82 of subunit C, produced by M. musculus, based on PDB structure 2TNF. The residues participating the hydrogen bond are shown in stick. The short bond length, 2.84Å, highly suggests a strong hydrogen bond that supports the tertiary structure. Baeyens, KJ et al. (1999)[18]. Generated in Chimera.
Model of hydrogen bond between Asn34 of subunit A and Arg82 of subunit C, produced by M. musculus, based on PDB structure 2TNF. The residues participating the hydrogen bond are shown in stick. The short bond length, 2.84Å, highly suggests a strong hydrogen bond that supports the tertiary structure. Baeyens, KJ et al. (1999)[18]. Generated in Chimera.
The PROSITE pattern for this family is located in a beta-strand in the central section of the protein that is conserved across all members.
All members of the TNF family, with the exception of the secreted lymphotoxin and a proliferation-inducing ligand (APRIL), are type II transmembrane proteins which protrude from immune cells. Such membrane-bound TNF ligands frequently signal back to the immune cells when they contact and bind their cognate receptors on other cells.[7]
Examples
Human proteins containing this domain include:
- CD40LG; CD70; EDA; FASLG; LTA; LTB;
- TNFSF4; TNFSF5; TNFSF7; TNFSF8; TNFSF9; TNFSF10; TNFSF11; TNFSF12; TNFSF13; TNFSF13B; TNFSF14; TNFSF15; TNFSF18;
- TWEAK;
References
- ^ Baeyens, KJ et al. (1999). "The structure of mouse tumour-necrosis factor at 1.4Å resolution towards modulation of its selectivity and trimerization". Acta Crystallography Section D: Biological Crystallographyl 55 (Pt4): 772-8. PMID 10089307.
- ^ Fransen L, Müller R, Marmenout A, Tavernier J, Van der Heyden J, Kawashima E, Chollet A, Tizard R, Van Heuverswyn H, Van Vliet A (June 1985). "Molecular cloning of mouse tumour necrosis factor cDNA and its eukaryotic expression". Nucleic Acids Res. 13 (12): 4417–29. doi:10.1093/nar/13.12.4417. PMC 321797. PMID 2989794. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=321797.
- ^ Kriegler M, Perez C, DeFay K, Albert I, Lu SD (April 1988). "A novel form of TNF/cachectin is a cell surface cytotoxic transmembrane protein: ramifications for the complex physiology of TNF". Cell 53 (1): 45–53. doi:10.1016/0092-8674(88)90486-2. PMID 3349526.
- ^ Sherry B, Jue DM, Zentella A, Cerami A (December 1990). "Characterization of high molecular weight glycosylated forms of murine tumor necrosis factor". Biochem. Biophys. Res. Commun. 173 (3): 1072–8. doi:10.1016/S0006-291X(05)80895-2. PMID 2268312.
- ^ a b Cseh K, Beutler B (September 1989). "Alternative cleavage of the cachectin/tumor necrosis factor propeptide results in a larger, inactive form of secreted protein". J. Biol. Chem. 264 (27): 16256–60. PMID 2777790.
- ^ Waltenbaugh C, Doan T, Melvold R, Viselli S (2008). Immunology. Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins. p. 68. ISBN 0-7817-9543-5.
- ^ a b Sun M, Fink PJ (2007). "A new class of reverse signaling costimulators belongs to the TNF family". J Immunol. 179 (7): 4307–12. PMID 17878324.
- ^ Peitsch MC, Jongeneel CV (February 1993). "A 3-D model for the CD40 ligand predicts that it is a compact trimer similar to the tumor necrosis factors". Int. Immunol. 5 (2): 233–8. doi:10.1093/intimm/5.2.233. PMID 8095800.
- ^ Farrah T, Smith CA (July 1992). "Emerging cytokine family". Nature 358 (6381): 26. doi:10.1038/358026b0. PMID 1377364.
- ^ Bazan JF (September 1993). "Emerging families of cytokines and receptors". Curr. Biol. 3 (9): 603–6. doi:10.1016/0960-9822(93)90009-D. PMID 15335677.
- ^ Beutler B, Cerami A (October 1988). "The history, properties, and biological effects of cachectin". Biochemistry 27 (20): 7575–82. doi:10.1021/bi00420a001. PMID 3061461.
- ^ Vilcek J, Lee TH (April 1991). "Tumor necrosis factor. New insights into the molecular mechanisms of its multiple actions". J. Biol. Chem. 266 (12): 7313–6. PMID 1850405.
- ^ Browning JL, Ngam-ek A, Lawton P, DeMarinis J, Tizard R, Chow EP, Hession C, O'Brine-Greco B, Foley SF, Ware CF (March 1993). "Lymphotoxin beta, a novel member of the TNF family that forms a heteromeric complex with lymphotoxin on the cell surface". Cell 72 (6): 847–56. doi:10.1016/0092-8674(93)90574-A. PMID 7916655.
- ^ Suda T, Takahashi T, Golstein P, Nagata S (December 1993). "Molecular cloning and expression of the Fas ligand, a novel member of the tumor necrosis factor family". Cell 75 (6): 1169–78. doi:10.1016/0092-8674(93)90326-L. PMID 7505205.
- ^ Baum PR, Gayle RB, Ramsdell F, Srinivasan S, Sorensen RA, Watson ML, Seldin MF, Baker E, Sutherland GR, Clifford KN (September 1994). "Molecular characterization of murine and human OX40/OX40 ligand systems: identification of a human OX40 ligand as the HTLV-1-regulated protein gp34". EMBO J. 13 (17): 3992–4001. PMC 395319. PMID 8076595. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=395319.
- ^ Wiley SR, Schooley K, Smolak PJ, Din WS, Huang CP, Nicholl JK, Sutherland GR, Smith TD, Rauch C, Smith CA (December 1995). "Identification and characterization of a new member of the TNF family that induces apoptosis". Immunity 3 (6): 673–82. doi:10.1016/1074-7613(95)90057-8. PMID 8777713.
- ^ Baeyens, KJ et al. (1999). "The structure of mouse tumour-necrosis factor at 1.4Å resolution towards modulation of its selectivity and trimerization". Acta Crystallography Section D: Biological Crystallographyl 55 (Pt4): 772-8. PMID 10089307.
- ^ Baeyens, KJ et al. (1999). "The structure of mouse tumour-necrosis factor at 1.4Å resolution towards modulation of its selectivity and trimerization". Acta Crystallography Section D: Biological Crystallographyl 55 (Pt4): 772-8. PMID 10089307.
External links
Cell signaling: cytokines By family CCLCXCLCX3CLXCLTNFIL6 like/gp130IL-12 family/IL12RB1OtherIL-10 familyIL-17 familyOtherBy function/
cellB trdu: iter (nrpl/grfl/cytl/horl), csrc (lgic, enzr, gprc, igsr, intg, nrpr/grfr/cytr), itra (adap, gbpr, mapk), calc, lipd; path (hedp, wntp, tgfp+mapp, notp, jakp, fsap, hipp, tlrp) This article includes text from the public domain Pfam and InterPro IPR006052
Categories:- Cytokines
Wikimedia Foundation. 2010.
