- Functional group
-
 Benzyl acetate has an ester functional group (in red), an acetyl moiety (circled with green) and a benzyloxy moiety (circled with orange). Other divisions can be made.
Benzyl acetate has an ester functional group (in red), an acetyl moiety (circled with green) and a benzyloxy moiety (circled with orange). Other divisions can be made.
In organic chemistry, functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. The same functional group will undergo the same or similar chemical reaction(s) regardless of the size of the molecule it is a part of.[1][2] However, its relative reactivity can be modified by nearby functional groups.
The word moiety (pronounced /ˈmɔɪəti/) is often used synonymously to "functional group," but, according to the IUPAC definition,[3] a moiety is a part of a molecule that may include either whole functional groups or a parts of functional groups as substructures. For example, an ester (RCOOR') has an ester functional group (COOR) and is composed of an alkoxy moiety (-OR') and an acyl moiety (RCO-), or, equivalently, it may be divided into carboxylate (RCOO-) and alkyl (-R') moieties. Each moiety may contain additional functional groups, for example, methyl para-hydroxybenzoate contains a phenol functional group within the acyl moiety.
Combining the names of functional groups with the names of the parent alkanes generates a powerful systematic nomenclature for naming organic compounds.
The atoms of functional groups are linked to each other and to the rest of the molecule by covalent bonds. When the group of covalently bound atoms bears a net charge, the group is referred to more properly as a polyatomic ion or a complex ion. Any subgroup of atoms of a compound also may be called a radical, and if a covalent bond is broken homolytically, the resulting fragment radicals are referred as free radicals.
The first carbon atom after the carbon that attaches to the functional group is called the alpha carbon; the second, beta carbon, the third, gamma carbon, etc. If there is another functional group at a carbon, it may be named with the Greek letter, e.g., the gamma-amine in gamma-aminobutanoic acid is on the third carbon of the carbon chain attached to the carboxylic acid group.
Contents
Synthetic chemistry
Organic reactions are facilitated and controlled by the functional groups of the reactants. In general, alkyls are unreactive and difficult to get to react selectively at the desired positions, with few exceptions. In contrast, unsaturated carbon functional groups, and carbon-oxygen and carbon-nitrogen functional groups have a more diverse array of reactions that are also selective. It may be necessary to create a functional group in the molecule to make it react. For example, to synthesize iso-octane (the 8-carbon ideal gasoline) from the unfunctionalized alkane isobutane (a 4-carbon gas), isobutane is first dehydrogenated into isobutene. This contains the alkene functional group and can now dimerize with another isobutene to give iso-octene, which is then catalytically hydrogenated to iso-octane using pressured hydrogen gas.
Crystallography
The International Union of Crystallography in its Crystallographic Information File dictionary defines "moiety" to represent discrete non-bonded components. Thus, Na2SO4 would contain 3 moieties (2 Na+ and one SO42-). The dictionary defines "chemical formula moiety": "Formula with each discrete bonded residue or ion shown as a separate moiety".
Functionalization
Functionalization is the addition of functional groups onto the surface of a material by chemical synthesis methods. The functional group added can be subjected to ordinary synthesis methods to attach virtually any kind of organic compound onto the surface.
Functionalization is employed for surface modification of industrial materials in order to achieve desired surface properties such as water repellent coatings for automobile windshields and non-biofouling, hydrophilic coatings for contact lenses. In addition, functional groups are used to covalently link functional molecules to the surface of chemical and biochemical devices such as microarrays and microelectromechanical systems.
Catalysts can be attached to a material that has been functionalized. For example, silica is functionalized with an alkyl silicone, wherein the alkyl contains an amine functional group. A ligand such as an EDTA fragment is synthesized onto the amine, and a metal cation is complexed into the EDTA fragment. The EDTA is not adsorbed onto the surface, but connected by a permanent chemical bond.
Functional groups are also used to covalently link molecules such as fluorescent dyes, nanoparticles, proteins, DNA, and other compounds of interest for a variety of applications such as sensing and basic chemical research.
Table of common functional groups
The following is a list of common functional groups. In the formulas, the symbols R and R' usually denote an attached hydrogen, or a hydrocarbon side chain of any length, but may sometimes refer to any group of atoms.
Hydrocarbons
Functional groups, called hydrocarbyls, that contain only carbon and hydrogen, but vary in the number and order of π bonds. Each one differs in type (and scope) of reactivity.
Chemical class Group Formula Structural Formula Prefix Suffix Example Alkane Alkyl RH 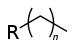
alkyl- -ane 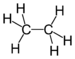
EthaneAlkene Alkenyl R2C=CR2 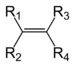
alkenyl- -ene 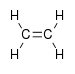
Ethylene
(Ethene)Alkyne Alkynyl RC≡CR' 
alkynyl- -yne 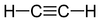
Acetylene
(Ethyne)Benzene derivative Phenyl RC6H5
RPh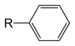
phenyl- -benzene 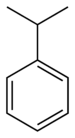
Cumene
(2-phenylpropane)Toluene derivative Benzyl RCH2C6H5
RBn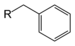
benzyl- 1-(substituent)toluene 
Benzyl bromide
(α-Bromotoluene)There are also a large number of branched or ring alkanes that have specific names, e.g., tert-butyl, bornyl, cyclohexyl, etc.
Hydrocarbons may form charged structures: positively charged carbocations or negative carbanions. Carbocations are often named -um. Examples are tropylium and triphenylmethyl cations and the cyclopentadienyl anion.
Groups containing halogens
Haloalkanes are a class of molecule that is defined by a carbon-halogen bond. This bond can be relatively weak (in the case of an iodoalkane) or quite stable (as in the case of a fluoroalkane). In general, with the exception of fluorinated compounds, haloalkanes readily undergo nucleophilic substitution reactions or elimination reactions. The substitution on the carbon, the acidity of an adjacent proton, the solvent conditions, etc. all can influence the outcome of the reactivity.
Chemical class Group Formula Structural Formula Prefix Suffix Example haloalkane halo RX 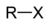
halo- alkyl halide 
Chloroethane
(Ethyl chloride)fluoroalkane fluoro RF 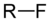
fluoro- alkyl fluoride 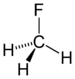
Fluoromethane
(Methyl fluoride)chloroalkane chloro RCl 
chloro- alkyl chloride 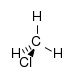
Chloromethane
(Methyl chloride)bromoalkane bromo RBr 
bromo- alkyl bromide 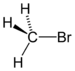
Bromomethane
(Methyl bromide)iodoalkane iodo RI 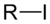
iodo- alkyl iodide 
Iodomethane
(Methyl iodide)Groups containing oxygen
Compounds that contain C-O bonds each possess differing reactivity based upon the location and hybridization of the C-O bond, owing to the electron-withdrawing effect of sp hybridized oxygen (carbonyl groups) and the donating effects of sp2 hybridized oxygen (alcohol groups).
Chemical class Group Formula Structural Formula Prefix Suffix Example Alcohol Hydroxyl ROH 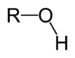
hydroxy- -ol 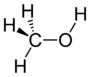
MethanolKetone Carbonyl RCOR' 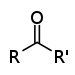
-oyl- (-COR')
or
oxo- (=O)-one 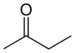
Butanone
(Methyl ethyl ketone)Aldehyde Aldehyde RCHO 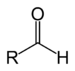
formyl- (-COH)
or
oxo- (=O)-al 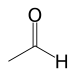
Ethanal
(Acetaldehyde)Acyl halide Haloformyl RCOX 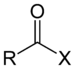
carbonofluoridoyl-
carbonochloridoyl-
carbonobromidoyl-
carbonoiodidoyl--oyl halide 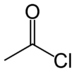
Acetyl chloride
(Ethanoyl chloride)Carbonate Carbonate ester ROCOOR 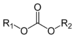
(alkoxycarbonyl)oxy- alkyl carbonate 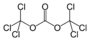
Triphosgene
(Di(trichloromethyl) carbonate)Carboxylate Carboxylate RCOO− 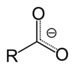
carboxy- -oate 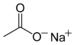
Sodium acetate
(Sodium ethanoate)Carboxylic acid Carboxyl RCOOH 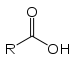
carboxy- -oic acid 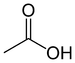
Acetic acid
(Ethanoic acid)Ester Ester RCOOR' 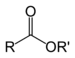
alkanoyloxy-
or
alkoxycarbonylalkyl alkanoate 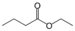
Ethyl butyrate
(Ethyl butanoate)Hydroperoxide Hydroperoxy ROOH 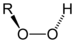
hydroperoxy- alkyl hydroperoxide 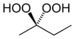
Methyl ethyl ketone peroxidePeroxide Peroxy ROOR 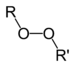
peroxy- alkyl peroxide 
Di-tert-butyl peroxideEther Ether ROR' 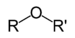
alkoxy- alkyl ether 
Diethyl ether
(Ethoxyethane)Hemiacetal Hemiacetal RCH(OR')(OH) 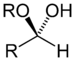
alkoxy -ol -al alkyl hemiacetal Hemiketal Hemiketal RC(ORʺ)(OH)R' 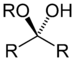
alkoxy -ol -one alkyl hemiketal Acetal Acetal RCH(OR')(OR") 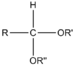
dialkoxy- -al dialkyl acetal Ketal (or Acetal) Ketal (or Acetal) RC(ORʺ)(OR‴)R' 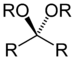
dialkoxy- -one dialkyl ketal Orthoester Orthoester RC(OR')(ORʺ)(OR‴) 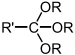
trialkoxy- Orthocarbonate ester Orthocarbonate ester C(OR)(OR')(ORʺ)(OR″) tetralkoxy- tetraalkyl orthocarbonate Groups containing nitrogen
Compounds that contain nitrogen in this category may contain C-O bonds, such as in the case of amides.
Chemical class Group Formula Structural Formula Prefix Suffix Example Amide Carboxamide RCONR2 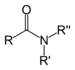
carboxamido-
or
carbamoyl--amide 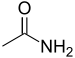
Acetamide
(Ethanamide)Amines Primary amine RNH2 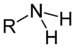
amino- -amine 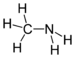
Methylamine
(Methanamine)Secondary amine R2NH 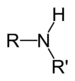
amino- -amine 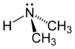
DimethylamineTertiary amine R3N 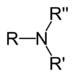
amino- -amine 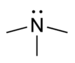
Trimethylamine4° ammonium ion R4N+ 
ammonio- -ammonium 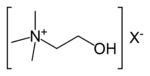
CholineImine Primary ketimine RC(=NH)R' 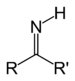
imino- -imine Secondary ketimine RC(=NR)R' 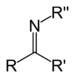
imino- -imine Primary aldimine RC(=NH)H 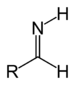
imino- -imine Secondary aldimine RC(=NR')H 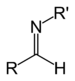
imino- -imine Imide Imide (RCO)2NR' 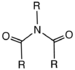
imido- -imide Azide Azide RN3 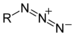
azido- alkyl azide 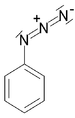
Phenyl azide
(Azidobenzene)Azo compound Azo
(Diimide)RN2R' 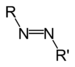
azo- -diazene 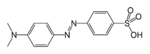
Methyl orange
(p-dimethylamino-azobenzenesulfonic acid)Cyanates Cyanate ROCN 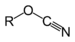
cyanato- alkyl cyanate 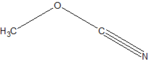
Methyl cyanateIsocyanate RNCO 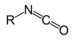
isocyanato- alkyl isocyanate 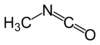
Methyl isocyanateNitrate Nitrate RONO2 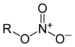
nitrooxy-, nitroxy- alkyl nitrate
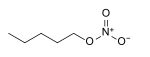
Amyl nitrate
(1-nitrooxypentane)Nitrile Nitrile RCN 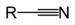
cyano- alkanenitrile
alkyl cyanide
Benzonitrile
(Phenyl cyanide)Isonitrile RNC 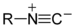
isocyano- alkaneisonitrile
alkyl isocyanide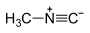
Methyl isocyanideNitrite Nitrosooxy RONO 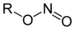
nitrosooxy- alkyl nitrite
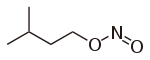
Isoamyl nitrite
(3-methyl-1-nitrosooxybutane)Nitro compound Nitro RNO2 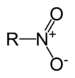
nitro- 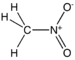
NitromethaneNitroso compound Nitroso RNO 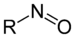
nitroso- 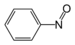
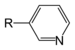
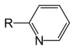
NitrosobenzenePyridine derivative Pyridyl RC5H4N 4-pyridyl
(pyridin-4-yl)3-pyridyl
(pyridin-3-yl)2-pyridyl
(pyridin-2-yl)-pyridine 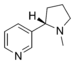
NicotineGroups containing sulphur
Compounds that contain sulfur exhibit unique chemistry due to their ability to form more bonds than oxygen, their lighter analogue on the periodic table. Substitutive nomenclature (marked as prefix in table) is preferred over functional class nomenclature (marked as suffix in table) for sulfides, disulfides, sulfoxides and sulfones.
Chemical class Group Formula Structural Formula Prefix Suffix Example Thiol Sulfhydryl RSH 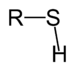
sulfanyl-
(-SH)-thiol 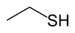
EthanethiolSulfide
(Thioether)Sulfide RSR' 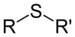
substituent sulfanyl-
(-SR')di(substituent) sulfide
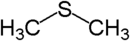
(Methylsulfanyl)methane (prefix) or
Dimethyl sulfide (suffix)Disulfide Disulfide RSSR' 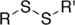
substituent disulfanyl-
(-SSR')di(substituent) disulfide
(Methyldisulfanyl)methane (prefix) or
Dimethyl disulfide (suffix)Sulfoxide Sulfinyl RSOR' 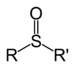
-sulfinyl-
(-SOR')di(substituent) sulfoxide 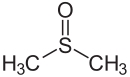
(Methanesulfinyl)methane (prefix) or
Dimethyl sulfoxide (suffix)Sulfone Sulfonyl RSO2R' 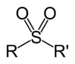
-sulfonyl-
(-SO2R')di(substituent) sulfone 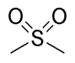
(Methanesulfonyl)methane (prefix) or
Dimethyl sulfone (suffix)Sulfinic acid Sulfino RSO2H 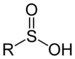
sulfino-
(-SO2H)-sulfinic acid 
2-Aminoethanesulfinic acidSulfonic acid Sulfo RSO3H 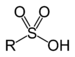
sulfo-
(-SO3H)-sulfonic acid 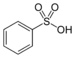
Benzenesulfonic acidThiocyanate Thiocyanate RSCN 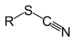
thiocyanato-
(-SCN)substituent thiocyanate 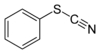
Phenyl thiocyanateIsothiocyanate RNCS 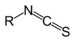
isothiocyanato-
(-NCS)substituent isothiocyanate 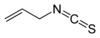
Allyl isothiocyanateThione Carbonothioyl RCSR' 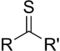
-thioyl-
(-CSR')
or
sulfanylidene-
(=S)-thione 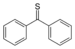
Diphenylmethanethione
(Thiobenzophenone)Thial Carbonothioyl RCSH 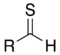
methanethioyl-
(-CSH)
or
sulfanylidene-
(=S)-thial Groups containing phosphorus
Compounds that contain phosphorus exhibit unique chemistry due to their ability to form more bonds than nitrogen, their lighter analogues on the periodic table.
Chemical class Group Formula Structural Formula Prefix Suffix Example Phosphine
(Phosphane)Phosphino R3P 
phosphanyl- -phosphane 
MethylpropylphosphanePhosphonic acid Phosphono RP(=O)(OH)2 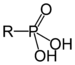
phosphono- substituent phosphonic acid 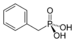
Benzylphosphonic acidPhosphate Phosphate ROP(=O)(OH)2 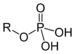
phosphonooxy-
or
O-phosphono- (phospho-)substituent phosphate 
Glyceraldehyde 3-phosphate (suffix)
O-Phosphonocholine (prefix)
(Phosphocholine)Phosphodiester Phosphate HOPO(OR)2 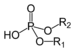
[(alkoxy)hydroxyphosphoryl]oxy-
or
O-[(alkoxy)hydroxyphosphoryl]-di(substituent) hydrogen phosphate
or
phosphoric acid di(substituent) esterDNA O‑[(2‑Guanidinoethoxy)hydroxyphosphoryl]‑L‑serine (prefix)
(Lombricine)See also
- For a list of all functional groups: Category:Functional groups
References
- ^ Compendium of Chemical Terminology (IUPAC "Gold Book") http://goldbook.iupac.org/F02555.html
- ^ March, Jerry (1985), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (3rd ed.), New York: Wiley, ISBN 0-471-85472-7
- ^ IUPAC. Compendium of Chemical Terminology, 2nd ed. (the "Gold Book"). Compiled by A. D. McNaught and A. Wilkinson. Blackwell Scientific Publications, Oxford (1997). XML on-line corrected version: http://goldbook.iupac.org (2006-) created by M. Nic, J. Jirat, B. Kosata; updates compiled by A. Jenkins. ISBN 0-9678550-9-8 doi:10.1351/goldbook.M03968 http://goldbook.iupac.org/M03968.html
External links
- IUPAC Blue Book (organic nomenclature)
- IUPAC ligand abbreviations (pdf)
- _chemical_formula_moiety in CIF dictionary
- Functional group synthesis from organic-reaction.com
Functional groups Acetyl · Acetoxy · Acryloyl · Acyl · Alcohol · Aldehyde · Alkane · Alkene · Alkyne · Alkoxy group · Amide · Amine · Azo compound · Benzene derivative · Carboxylic acid · Cyanate · Disulfide · Ester · Ether · Epoxide · Haloalkane · Hydrazone · Hydroxyl · Imine · Isocyanate · Isonitrile · Isothiocyanate · Ketone · Methine · Nitrile · Nitro compound · Nitroso compound · Organophosphorus · Oxime · Peroxide · Phosphonous and Phosphonic acid · Pyridine derivative · Sulfone · Sulfonic acid · Sulfoxide · Thiocyanate · Thioester · Thioether · Thiol · Urea
Concepts in organic chemistry Aromaticity · Covalent bonding · Functional groups · Nomenclature · Organic compounds · Organic reactions · Organic synthesis · Publications · Spectroscopy · Stereochemistry · List of organic compoundsCategories:- Functional groups
- Organic chemistry
Wikimedia Foundation. 2010.