- Sodium acetate
-
Sodium acetate 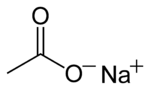


 Sodium acetateSystematic nameSodium ethanoateOther namesHot ice (Sodium acetate trihydrate)
Sodium acetateSystematic nameSodium ethanoateOther namesHot ice (Sodium acetate trihydrate)Identifiers CAS number 127-09-3  , 6131-90-4 (trihydrate)
, 6131-90-4 (trihydrate)PubChem 517045 ChemSpider 29105 
UNII NVG71ZZ7P0 
ChEBI CHEBI:32954 
ChEMBL CHEMBL1354 
RTECS number AJ4300010 (anhydrous)
AJ4580000Jmol-3D images Image 1 - [Na+].[O-]C(=O)C
Properties Molecular formula C2H3NaO2 Molar mass 82.03 g mol−1 Appearance White deliquescent powder
odorlessDensity 1.528 g/cm3
1.45 g/cm3 (trihydrate)Melting point 324 °C (anhydrous)
58 °C (trihydrate)Boiling point 881.4 °C (anhydrous)
122 °C (trihydrate)(decomposes)Solubility in water 36.2 g/100 ml (0°C)
46.4 g/100 mL (20°C)
139 g/100 mL (60°C)
170.15 g/100 mL (100°C)Solubility soluble in ethanol (5.3 g/100 mL (trihydrate) Basicity (pKb) 9.25 Refractive index (nD) 1.464 Structure Crystal structure monoclinic Hazards MSDS External MSDS Main hazards Irritant NFPA 704 Flash point 250 °C Autoignition
temperature607 °C Related compounds Other anions Sodium formate
Sodium propionateOther cations Potassium acetate
Calcium acetate (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Sodium acetate, CH3COONa, also abbreviated NaOAc,[1] also sodium ethanoate, is the sodium salt of acetic acid. This colourless salt has a wide range of uses.
Contents
Applications
Industrial
Sodium acetate is used in the textile industry to neutralize sulfuric acid waste streams, and as a photoresist while using aniline dyes. It is also a pickling agent in chrome tanning, and it helps to retard vulcanization of chloroprene in synthetic rubber production.
Concrete longevity
Sodium acetate is used to reduce the damage water can potentially do to concrete by acting as a Concrete sealant, while also being environmentally benign and cheaper than the epoxy alternative that is usually employed for sealing concrete against water permeation [2].
Food
Sodium acetate may be added to foods as a seasoning. It may be used in the form of sodium diacetate — a 1:1 complex of sodium acetate and acetic acid,[3] given the E-number E262. A frequent use of this form is in salt and vinegar chips in the United States. Many US brands, including national manufacturer Frito-Lay, sell "salt and vinegar flavoured" chips that use this chemical, with lactose and smaller percentages of other chemicals, in lieu of a real salt and vinegar preparation.[4]
Buffer solution
As the conjugate base of a weak acid, a solution of sodium acetate and acetic acid can act as a buffer to keep a relatively constant pH. This is useful especially in biochemical applications where reactions are pH dependent.
Heating pad
Sodium acetate is also used in consumer heating pads or hand warmers and is also used in hot ice. Sodium acetate trihydrate crystals melt at 54°C,[5] dissolving in their water of crystallization. When they are heated to around 100°C, and subsequently allowed to cool, the aqueous solution becomes supersaturated. This solution is capable of cooling to room temperature without forming crystals. By clicking on a metal disc in the heating pad, a nucleation centre is formed which causes the solution to crystallize into solid sodium acetate trihydrate again. The bond-forming process of crystallization is exothermic.[6][7][8] The latent heat of fusion is about 264–289 kJ/kg.[9] Unlike some other types of heat packs that depend on irreversible chemical reactions, sodium acetate heat packs can be easily recharged by boiling until all crystals are dissolved; they can be reused indefinitely.[citation needed]
 An inexpensive hand warmer containing a supersaturated solution of sodium acetate which releases heat on crystallization
An inexpensive hand warmer containing a supersaturated solution of sodium acetate which releases heat on crystallization
Preparation
For laboratory use, sodium acetate is very inexpensive, and is usually purchased instead of being synthesized. It is sometimes produced in a laboratory experiment by the reaction of acetic acid (also called ethanoic acid) with sodium carbonate, sodium bicarbonate, or sodium hydroxide. These reactions produce aqueous sodium acetate and water. Carbon dioxide is produced in the reaction with sodium carbonate and bicarbonate, and it leaves the reaction vessel as a gas (unless the reaction vessel is pressurized). This is the well-known "volcano" reaction between baking soda and vinegar.
- CH3COOH + NaHCO3 → CH3COONa + H2O + CO2
Industrially, sodium acetate is prepared from glacial acetic acid and sodium hydroxide.
- CH3COOH + NaOH → CH3COONa + H2O
Reactions
Sodium acetate can be used to form an ester with an alkyl halide such as bromoethane:
- CH3COONa + BrCH2CH3 → CH3COOCH2CH3 + NaBr
Caesium salts catalyze this reaction.
References
- ^ Clayden, Jonathan; Greeves, Nick; Warren, Stuart; Wothers, Peter (2001). Organic Chemistry (1st ed.). Oxford University Press. ISBN 978-0-19-850346-0.
- ^ http://www.sciencedaily.com/releases/2007/08/070806101941.htm
- ^ http://www.jungbunzlauer.com/products-applications/products/specialties/sodium-diacetate/general-information.html
- ^ http://www.fritolay.com/our-snacks/lays-salt-vinegar.html
- ^ [Courty JM, Kierlik E, Les chaufferettes chimiques, Pour la Science, décembre 2008, p 108-110]
- ^ "Crystallization of Supersaturated Sodium Acetate". Journal of Chemical Education. http://jchemed.chem.wisc.edu/JCESoft/CCA/CCA3/MAIN/ACETATE/PAGE1.HTM.
- ^ Fake latent heat and supersaturation
- ^ "How do sodium acetate heat pads work?". HowStuffWorks. http://www.howstuffworks.com/question290.htm. Retrieved 2007-09-03.
- ^ Ibrahim Dincer and Marc A. Rosen. Thermal Energy Storage: Systems and Applications, page 155
External links
Sodium compounds NaAlO2 · NaBH3(CN) · NaBH4 · NaBr · NaBrO3 · NaCH3COO · NaCN · NaC6H5CO2 · NaC6H4(OH)CO2 · NaCl · NaClO · NaClO2 · NaClO3 · NaClO4 · NaF · NaH · NaHCO3 · NaHSO3 · NaHSO4 · NaI · NaIO3 · NaIO4 · NaMnO4 · NaNH2 · NaNO2 · NaNO3 · NaN3 · NaOH · NaO2 · NaPO2H2 · NaReO4 · NaSCN · NaSH · NaTcO4 · NaVO3 · Na2CO3 · Na2C2O4 · Na2CrO4 · Na2Cr2O7 · Na2MnO4 · Na2MoO4 · Na2O · Na2O2 · Na2O(UO3)2 · Na2S · Na2SO3 · Na2SO4 · Na2S2O3 · Na2S2O4 · Na2S2O5 · Na2S2O6 · Na2S2O7 · Na2S2O8 · Na2Se · Na2SeO3 · Na2SeO4 · Na2SiO3 · Na2Te · Na2TeO3 · Na2Ti3O7 · Na2U2O7 · NaWO4 · Na2Zn(OH)4 · Na3N · Na3P · Na3VO4 · Na4Fe(CN)6 · Na5P3O10 · NaBiO3
Categories:- Acetates
- Sodium compounds
- Food additives
- Photographic chemicals
Wikimedia Foundation. 2010.


