- Nitrile
-
This article is about the group of organic compounds. For the synthetic rubber product, see Nitrile rubber.
A nitrile is any organic compound that has a -C≡N functional group.[1] The prefix cyano- is used interchangeably with the term nitrile in industrial literature. Nitriles are found in many useful compounds, one example being super glue (methyl cyanoacrylate).
Inorganic compounds containing the -C≡N group are not called nitriles, but cyanides instead.[2] Though both nitriles and cyanides can be derived from cyanide salts, most nitriles are not nearly as toxic.
Contents
History
The first compound of the homolog row of nitriles, the nitrile of formic acid, hydrogen cyanide was first synthesized by C.W. Scheele in 1782.[3] In 1811 J. L. Gay-Lussac was able to prepare the very toxic and volatile pure acid. The nitrile of benzoic acids was first prepared by Friedrich Wöhler and Justus von Liebig, but due to minimal yield of the synthesis neither physical nor chemical properties were determined nor a structure suggested. Théophile-Jules Pelouze synthesized propionitrile in 1834 suggesting it to be an ether of propionic alcohol and hydrocyanic acid.[4] The synthesis of benzonitrile by Hermann Fehling in 1844, by heating ammonium benzoate, was the first method yielding enough of the substance for chemical research. He determined the structure by comparing it to the already known synthesis of hydrogen cyanide by heating ammonium formate to his results. He coined the name nitrile for the newfound substance, which became the name for the compound group.[5]
Nitriles occur naturally in a diverse set of plant and animal sources. Over 120 naturally occurring nitriles have been isolated from terrestrial and marine sources. Nitriles are commonly encountered in fruit pits, especially almonds, and during cooking of Brassica crops (such as cabbage, brussel sprouts, and cauliflower), which release nitriles being released through hydrolysis. Mandelonitrile, a cyanohydrin produced by ingesting almonds or some fruit pits, releases hydrogen cyanide and is responsible for the toxicity of cyanogenic glycosides.[6]
Over 30 nitrile-containing pharmaceuticals are currently marketed for a diverse variety of medicinal indications with more than 20 additional nitrile-containing leads in clinical development. The nitrile group is quite robust and, in most cases, is not readily metabolized but passes through the body unchanged. The types of pharmaceuticals containing nitriles is diverse, from Vildagliptin an antidiabetic drug to Anastrazole which is the gold standard in treating breast cancer. In many instances the nitrile mimics functionality present in substrates for enzymes, whereas in other cases the nitrile increases water solubility or decreases susceptibility to oxidative metabolism in the liver.[7]
Synthesis
Industrially, the main methods for producing nitriles are ammoxidation and hydrocyanation. Both routes are green in the sense that they do not generate stoichiometric amounts of salts. In ammonoxidation, a hydrocarbon is partially oxidized in the presence of ammonia. This conversion is practiced on a large scale for acrylonitrile:[8]
- CH3CH=CH2 + 3/2 O2 + NH3 → NCCH=CH2 + 3 H2O
An example of hydrocyanation is the production of adiponitrile from 1,3-butadiene.
- CH2=CH-CH=CH2 + 2 HCN → NC(CH2)4CN
Usually for more specialty applications in organic synthesis, nitriles can be prepared by a wide variety of other methods:
- Nucleophilic aliphatic substitution reactions of alkyl halides with metal cyanides in the Kolbe nitrile synthesis. Aryl nitriles are prepared in the Rosenmund-von Braun synthesis.
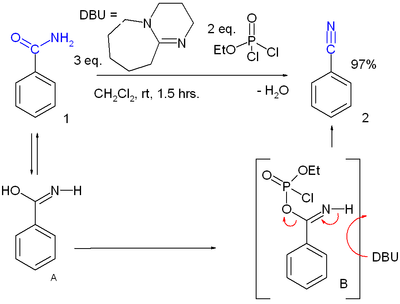
- Dehydration of primary amides. Many reagents are available, the combination of ethyl dichlorophosphate and DBU just one of them in this conversion of benzamide to benzonitrile:[9]
- dehydration of secondary amides (von Braun amide degradation)
- dehydration of aldoximes with triethylamine/sulfur dioxide, zeolites, or sulfuryl chloride
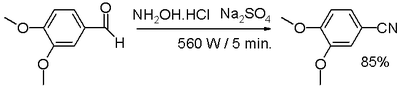
- One-pot synthesis of aldehyde with hydroxylamine and sodium sulfate.
- In one study [10] an aromatic or aliphatic aldehyde is reacted with hydroxylamine and anhydrous sodium sulfate in a dry media reaction for a very small amount of time under microwave irradiation through an intermediate aldoxime.
- reaction of metal cyanides with aldehydes in the cyanohydrin reaction
- from aryl carboxylic acids (Letts nitrile synthesis)
- aromatic nitriles from diazonium compounds in the Sandmeyer reaction
- A commercial source for the cyanide group is diethylaluminum cyanide Et2AlCN which can be prepared from triethylaluminium and HCN.[11] It has been used in nucleophilic addition to ketones.[12] For an example of its use see: Kuwajima Taxol total synthesis
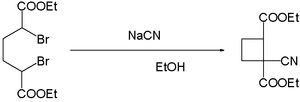
- cyanide ions facilitate the coupling of dibromides. Reaction of α,α'-dibromo adipic acid with sodium cyanide in ethanol yields the cyano cyclobutane:[13]
- In the so-called Franchimont Reaction (A. P. N. Franchimont, 1872) an α-bromocarboxylic acid is dimerized after hydrolysis of the cyanogroup and decarboxylation [14]
- Aromatic nitriles can be prepared from base hydrolysis of trichloromethyl aryl ketimines (RC(CCl3)=NH) in the Houben-Fischer synthesis [15][16]
Reactions
Nitrile groups in organic compounds can undergo various reactions when subject to certain reactants or conditions. A nitrile group can be hydrolyzed, reduced, or ejected from a molecule as a cyanide ion.
Hydrolysis
The hydrolysis of nitriles RCN proceeds in the distinct steps under acid or base treatment to achieve carboxamides RC(=O)NH2 and then carboxylic acids RCOOH. The hydrolysis of nitriles is generally considered to be one of the best methods for the preparation of carboxylic acids. However, these base or acid catalyzed reactions have certain limitations and/or disadvantages for preparation of amides. The general restriction is that the final neutralization of either base or acid leads to an extensive salt formation with inconvenient product contamination and pollution effects. Particular limitations are as follows:
- The base catalyzed reactions. The kinetic studies allowed the estimate of relative rates for the hydration at each step of the reaction and, as a typical example, the second-order rate constants for hydroxide-ion catalyzed hydrolysis of acetonitrile and acetamide are 1.6×10−6 and 7.4×10−5M−1s−1, respectively. Comparison of these two values indicates that the second step of the hydrolysis for the base-catalyzed reaction is faster than the first one, and the reaction should proceed to the final hydration product (the carboxylate salt) rather than stopping at the amide stage. This implies that amides prepared in the conventional metal-free base-catalyzed reaction should be contaminated with carboxylic acids and they can be isolated in only moderate yields.
- The acid catalyzed reactions. Application of strong acidic solutions requires a careful control of the temperature and of the ratio of reagents in order to avoid the formation of polymers, which is promoted by the exothermic character of the hydrolysis.[17]
Reduction
In organic reduction the nitrile is reduced by reacting it with hydrogen with a nickel catalyst; an amine is formed in this reaction (see nitrile reduction). Reduction to the imine followed by hydrolysis to the aldehyde takes place in the Stephen aldehyde synthesis
Nucleophiles
A nitrile is an electrophile at the carbon atom in a nucleophilic addition reactions:
- with an organozinc compound in the Blaise reaction
- and with alcohols in the Pinner reaction.
- likewise, the reaction of the amine sarcosine with cyanamide yields creatine [18]
- Nitriles react in Friedel-Crafts acylation in the Houben-Hoesch reaction to ketones
Alkylation
Deprotonating nitriles generates exceptionally powerful nucleophiles that alkylate a wide variety of electrophiles. Key to the exceptional nucleophilicity is the extremely small steric demand of the CN unit combined with the unusual inductive stabilization. These features make nitriles ideal for creating new carbon-carbon bonds in sterically demanding environments and is used in several syntheses of medicinal targets as well as in the synthesis of pharmaceutical medicines. Recent developments have shown that the nature of the metal counter-ion causes different coordination to either the nitrile nitrogen or the adjacent nucleophilic carbon, often with profound differences in reactivity and stereochemistry.[19]
Miscellanea
- In reductive decyanation the nitrile group is replaced by a proton.[20] An effective decyanation is by a dissolving metal reduction with HMPA and potassium metal in tert-butanol. α-Amino-nitriles can be decyanated with lithium aluminium hydride.
- Nitriles self-react in presence of base in the Thorpe reaction in a nucleophilic addition
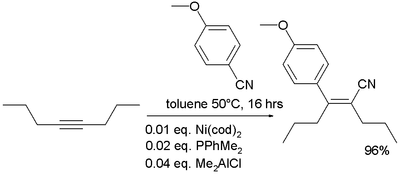
- In organometallic chemistry nitriles are known to add to alkynes in carbocyanation:[21]
Organic cyanamides
Cyanamides are N-cyano compounds with general structure R1R2N-CN and related to the inorganic parent cyanamide. For an example see: von Braun reaction.
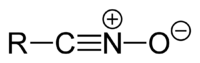
Nitrile oxides
Nitrile oxides have the general structure R-CNO.
See also
- For the polymer used to make safety gloves, see Nitrile rubber.
- Protonated nitriles: Nitrilium
- Deprotonated nitriles: Nitrile anion
- Cyanocarbon
- Nitrile ylide
References
- ^ IUPAC Gold Book nitriles
- ^ NCBI-MeSH Nitriles
- ^ David T. Mowry (1948). "The Preparation of Nitriles" (– Scholar search). Chemical Reviews 42 (2): 189–283. doi:10.1021/cr60132a001. http://pubs.acs.org/cgi-bin/abstract.cgi/chreay/1942/42/i02/f-pdf/f_cr60132a001.pdf.[dead link]
- ^ J. Pelouze (1834). "Notiz über einen neuen Cyanäther". Annalen der Chemie und Pharmacie 10 (2): 249. doi:10.1002/jlac.18340100302.
- ^ Hermann Fehling (1844). "Ueber die Zersetzung des benzoësauren Ammoniaks durch die Wärme". Annalen der Chemie und Pharmacie 49 (1): 91–97. doi:10.1002/jlac.18440490106.
- ^ Natural Product Reports Issue 5, 1999 Nitrile-containing natural products
- ^ Fleming, Fraser F.; Yao, Lihua; Ravikumar, P. C.; Funk, Lee; Shook, Brian C. (November 2010). "Nitrile-containing pharmaceuticals: efficacious roles of the nitrile pharmacophore". J Med Chem 53 (22): 7902–17. doi:10.1021/jm100762r. PMC 2988972. PMID 20804202. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2988972.
- ^ Peter Pollak, Gérard Romeder, Ferdinand Hagedorn, Heinz-Peter Gelbke "Nitriles" Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH, Weinheim. doi:10.1002/14356007.a17_363
- ^ Chun-Wei Kuo, Jia-Liang Zhu, Jen-Dar Wu, Cheng-Ming Chu, Ching-Fa Yao and Kak-Shan Shia (2007). "A convenient new procedure for converting primary amides into nitriles". Chem. Commun. 2007 (3): 301–303. doi:10.1039/b614061k. PMID 17299646.
- ^ Sharwan K, Dewan, Ravinder Singh, and Anil Kumar (2006). "One pot synthesis of nitriles from aldehydes and hydroxylamine hydrochloride using sodium sulfate (anhyd) and sodium bicarbonate in dry media under microwave irradiation" (open access). Arkivoc: (ii) 41–44. http://www.arkat-usa.org/ark/journal/2006/I02_General/1646/05-1646D%20as%20published%20mainmanuscript.pdf.
- ^ W. Nagata and M. Yoshioka (1988), "Diethylaluminum cyanide", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv6p0436; Coll. Vol. 6: 436
- ^ W. Nagata, M. Yoshioka, and M. Murakami (1988), "PREPARATION OF CYANO COMPOUNDS USING ALKYLALUMINUM INTERMEDIATES: 1-CYANO-6-METHOXY-3,4-DIHYDRONAPHTHALENE", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv6p0307; Coll. Vol. 6: 307
- ^ Reynold C. Fuson, Oscar R. Kreimeier, and Gilbert L. Nimmo (1930). "Ring Closures In The Cyclobutane Series. Ii. Cyclization Of Α,Α′-Dibromo-Adipic Esters". J. Am. Chem. Soc. 52 (10): 4074–4076. doi:10.1021/ja01373a046.
- ^ Franchimont Reaction
- ^ Über eine neue Methode zur Darstellung cyclischer Nitrile durch katalytischen Abbau (I. Mitteil.) (p 2464-2472) J. Houben, Walter Fischer Berichte der deutschen chemischen Gesellschaft (A and B Series) Volume 63, Issue 9 , Pages 2464 - 2472 doi:10.1002/cber.19300630920
- ^ http://www.drugfuture.com/OrganicNameReactions/ONR197.htm Merck & Co., Inc., Whitehouse Station
- ^ V. Yu. Kukushkin, A. J. L. Pombeiro, Metal-mediated and metal-catalyzed hydrolysis of nitriles (a review), Inorg. Chim. Acta, 358 (2005) 1–21
- ^ Smith, Andri L.; Tan, Paula (2006). "Creatine Synthesis: An Undergraduate Organic Chemistry Laboratory Experiment". J. Chem. Educ. 83: 1654. doi:10.1021/ed083p1654. http://jchemed.chem.wisc.edu/Journal/Issues/2006/Nov/abs1654.html.
- ^ Tetrahedron Volume 61, Issue 4, 24 January 2005, Pages 747-789 doi:10.1016/j.tet.2004.11.012
- ^ The reductive decyanation reaction: chemical methods and synthetic applications Jean-Marc Mattalia, Caroline Marchi-Delapierre, Hassan Hazimeh, and Michel Chanon Arkivoc (AL-1755FR) pp 90-118 2006 Article
- ^ Yoshiaki Nakao, Akira Yada, Shiro Ebata, and Tamejiro Hiyama (2007). "A Dramatic Effect of Lewis-Acid Catalysts on Nickel-Catalyzed Carbocyanation of Alkynes" (Communication). J. Am. Chem. Soc. 129 (9): 2428–2429. doi:10.1021/ja067364x. PMID 17295484.
- External links:
- IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "nitrile".
- IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "cyanide".
Functional groups Acetyl · Acetoxy · Acryloyl · Acyl · Alcohol · Aldehyde · Alkane · Alkene · Alkyne · Alkoxy group · Amide · Amine · Azo compound · Benzene derivative · Carboxylic acid · Cyanate · Disulfide · Ester · Ether · Epoxide · Haloalkane · Hydrazone · Hydroxyl · Imine · Isocyanate · Isonitrile · Isothiocyanate · Ketone · Methine · Nitrile · Nitro compound · Nitroso compound · Organophosphorus · Oxime · Peroxide · Phosphonous and Phosphonic acid · Pyridine derivative · Sulfone · Sulfonic acid · Sulfoxide · Thiocyanate · Thioester · Thioether · Thiol · Urea
See also Chemical classification Categories:- Nitriles
Wikimedia Foundation. 2010.