- Dimethylamine
-
Dimethylamine 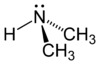
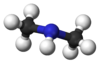 Dimethylamine[citation needed]Other namesN-Methylmethanamine[1]
Dimethylamine[citation needed]Other namesN-Methylmethanamine[1]Identifiers CAS number 124-40-3 
PubChem 674 ChemSpider 654 
UNII ARQ8157E0Q 
EC number 204-697-4 UN number 1032 KEGG C00543 
MeSH dimethylamine ChEBI CHEBI:17170 
ChEMBL CHEMBL120433 
RTECS number IP8750000 Beilstein Reference 605257 Gmelin Reference 849 3DMet B00125 Jmol-3D images Image 1 - CNC
Properties[3][4] Molecular formula C2H7N Molar mass 45.0837 g mol−1 Exact mass 45.057849229 g mol−1 Appearance Colorless gas Odor Ammonical, pungent Density 680 mg cm−3 (at 20 °C) Melting point -92 °C, 181 K, -134 °F
Boiling point 7-9 °C, 280-282 K, 44-48 °F
Solubility in water 3.540 kg dm−3 log P −0.362 Vapor pressure 170.3 pKa (at 20 °C) Acidity (pKa) 10.64[2] Basicity (pKb) 3.36 Thermochemistry Std enthalpy of
formation ΔfHo298−18.422 kJ mol−1 Hazards GHS pictograms 


GHS signal word DANGER GHS hazard statements H220, H302, H315, H318, H332, H335 GHS precautionary statements P210, P261, P280, P305+351+338, P410+403 EU Index 612-001-00-9 EU classification  F+
F+  Xn
XnR-phrases R12, R20, R37/38, R41 S-phrases (S2), S16, S26, S39 NFPA 704 Flash point −6 °C Autoignition
temperature401 °C Explosive limits 2.8–14.4% Related compounds Related amines Diethylamine
 (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Dimethylamine is an organic compound with the formula (CH3)2NH. This secondary amine is a colorless, flammable liquified gas with an ammonia-like odor. Dimethylamine is generally encountered as a solution in water at concentrations up to around 40%. In 2005, an estimated 270,000 tons were produced.[5]
Contents
Structure and properties
The molecule consists of a nitrogen atom with two methyl substituents and one proton. Dimethylamine is a base and the pKa of the ammonium salt CH3-NH2+-CH3 is 10.73, a value above methylamine (10.64) and trimethylamine (9.79).
Dimethylamine reacts with acids to form salts, such as dimethylamine hydrochloride, an odorless white solid with a melting point of 171.5 °C. Dimethylamine is produced by catalytic reaction of methanol and ammonia at elevated temperatures and high pressure:[6]
- 2 CH3OH + NH3 → (CH3)2NH + 2 H2O
Uses
Dimethylamine is a precursor to several industrially significant compounds.[5][7] It reacts with carbon disulfide to give dimethyldithiocarbamate, a precursor to a family of chemicals widely used in the vulcanization of rubber. The solvents dimethylformamide and dimethylacetamide are derived from dimethylamine. It is raw material for the production of many agrichemicals and pharmaceuticals, such as dimefox and diphenhydramine, respectively. The chemical weapon tabun is derived from dimethylamine. The surfactant lauryl dimethylamine oxide is found in soaps and cleaning compounds. Unsymmetrical dimethylhydrazine, a rocket fuel, is prepared from dimethylamine.
Biochemistry
The German cockroach utilizes dimethylamine as a pheromone for communication.[8]
Dimethylamine undergoes nitrosation under weak acid conditions to give dimethylnitrosamine. This animal carcinogen has been detected and quantified in human urine samples and it may also arise from nitrosation of dimethylamine by nitrogen oxides present in acid rain in highly industrialized countries.[citation needed]
See also
References
- ^ "dimethylamine - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 16 September 2004. Identification and Related Records. http://pubchem.ncbi.nlm.nih.gov/summary/summary.cgi?cid=674. Retrieved 21 October 2011.
- ^ Hall, H.K. (1957). J. Am. Chem. Soc. 79: 5441.
- ^ "Dimethylamine". NIST Chemistry WebBook. USA: National Institute of Standards and Technology. http://webbook.nist.gov/cgi/cbook.cgi?ID=C124403&Mask=1#Thermo-Gas. Retrieved 21 October 2011.
- ^ "Dimethylamine 38931 - ≥99.0%". Aldrich. Sigma-Aldrich Co. http://www.sigmaaldrich.com/catalog/ProductDetail.do?lang=en&N4=38931%7CALDRICH&N5=SEARCH_CONCAT_PNO. Retrieved 21 October 2011.
- ^ a b A. B. van Gysel, W. Musin "Methylamines" in Ullmann's Encyclopedia of Industrial Chemistry, 2005 Wiley-VCH Verlag, Weinheim. doi:10.1002/14356007.a16 535
- ^ Corbin D.R.; Schwarz S.; Sonnichsen G.C. (1997). "Methylamines synthesis: A review". Catalysis Today 37 (2): 71–102. doi:10.1016/S0920-5861(97)00003-5.
- ^ Ashford's Dictionary of Industrial Chemicals, 3rd edition, 2011, pages 3284-3286
- ^ Zhang AQ, Mitchell SC, Smith RL (November 1998). "Dimethylamine formation in the rat from various related amine precursors". Food Chem. Toxicol. 36 (11): 923–7. doi:10.1016/S0278-6915(98)00074-X. PMID 9771553.
External links
- International Chemical Safety Card 0260 (gas)
- International Chemical Safety Card 1485 (aqueous solution)
- NIOSH Pocket Guide to Chemical Hazards 0219
- Properties from Air Liquide
- MSDS at airliquide.com
- MSDS at physchem.ox.ac.uk
Categories:- Amines
- Pheromones
- Insect ecology
Wikimedia Foundation. 2010.

