- Superoxide dismutase
-
Superoxide dismutase 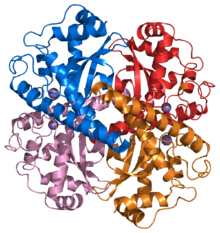
Structure of a human Mn superoxide dismutase 2 tetramer.[1] Identifiers EC number 1.15.1.1 CAS number 9054-89-1 Databases IntEnz IntEnz view BRENDA BRENDA entry ExPASy NiceZyme view KEGG KEGG entry MetaCyc metabolic pathway PRIAM profile PDB structures RCSB PDB PDBe PDBsum Gene Ontology AmiGO / EGO Search PMC articles PubMed articles Superoxide dismutases (SOD, EC 1.15.1.1) are a class of enzymes that catalyze the dismutation of superoxide into oxygen and hydrogen peroxide. As such, they are an important antioxidant defense in nearly all cells exposed to oxygen. One of the exceedingly rare exceptions is Lactobacillus plantarum and related lactobacilli, which use a different mechanism.
Contents
Reaction
The SOD-catalysed dismutation of superoxide may be written with the following half-reactions :
- M(n+1)+-SOD + O2− → Mn+-SOD + O2
- Mn+-SOD + O2− + 2H+ → M(n+1)+-SOD + H2O2.
where M = Cu (n=1) ; Mn (n=2) ; Fe (n=2) ; Ni (n=2).
In this reaction the oxidation state of the metal cation oscillates between n and n+1.
Types
General
Discovered by Irwin Fridovich and Joe McCord, SOD enzymes were previously thought to be several metalloproteins with unknown function (for example, CuZnSOD was known as erythrocuprein).[2] Several common forms of SOD exist: they are proteins cofactored with copper and zinc, or manganese, iron, or nickel. For example, Brewer (1967) identified a protein that became known as superoxide dismutase as an indophenol oxidase by protein analysis of starch gels using the phenazine-tetrazolium technique.[3]
There are three major families of superoxide dismutase, depending on the metal cofactor: Cu/Zn (which binds both copper and zinc), Fe and Mn types (which bind either iron or manganese), and the Ni type, which binds nickel.
- Copper and zinc – most commonly used by eukaryotes. The cytosols of virtually all eukaryotic cells contain an SOD enzyme with copper and zinc (Cu-Zn-SOD). For example, Cu-Zn-SOD available commercially is normally purified from the bovine erythrocytes: The Cu-Zn enzyme is a homodimer of molecular weight 32,500. The bovine Cu-Zn protein was the first SOD structure to be solved, in 1975.[5] It is an 8-stranded "Greek key" beta-barrel, with the active site held between the barrel and two surface loops. The two subunits are tightly joined back-to-back, primarily by hydrophobic and some electrostatic interactions. The ligands of the copper and zinc are six histidine and one aspartate side-chains; one histidine is shared between the two metals.[6]
- Iron or manganese – used by prokaryotes and protists, and in mitochondria
- Iron – E. coli and many other bacteria also contain a form of the enzyme with iron (Fe-SOD); some bacteria contain Fe-SOD, others Mn-SOD, and some contain both. (For the E. coli Fe-SOD: . Fe-SOD can be found in the plastids of plants. The 3D structures of the homologous Mn and Fe superoxide dismutases have the same arrangement of alpha-helices, and their active sites contain the same type and arrangement of amino acid side-chains.
- Manganese – Chicken liver (and nearly all other) mitochondria, and many bacteria (such as E. coli), contain a form with manganese (Mn-SOD): for example, the Mn-SOD found in human mitochondria. The ligands of the manganese ions are 3 histidine side-chains, an aspartate side-chain and a water molecule or hydroxy ligand, depending on the Mn oxidation state (respectively II and III).[7]
- nickel – prokaryotic. A hexameric structure built from right-handed 4-helix bundles, each containing N-terminal hooks that chelate a Ni ion. The Ni-hook contains the motif His-Cys-X-X-Pro-Cys-Gly-X-Tyr; it provides most of the interactions critical for metal binding and catalysis and is, therefore, a likely diagnostic of NiSODs.[8][9]
Copper/zinc superoxide dismutase 
Structure of the yeast Cu,Zn enzyme superoxide dismutase.[10] Identifiers Symbol Sod_Cu Pfam PF00080 InterPro IPR001424 PROSITE PDOC00082 SCOP 1sdy Available protein structures: Pfam structures PDB RCSB PDB; PDBe PDBsum structure summary Iron/manganese superoxide dismutases, alpha-hairpin domain 
The structure of human mitochondrial manganese superoxide dismutase, which reveals a novel tetrameric interface of two 4-helix bundles.[7] Identifiers Symbol Sod_Fe_N Pfam PF00081 InterPro IPR001189 PROSITE PDOC00083 SCOP 1n0j Available protein structures: Pfam structures PDB RCSB PDB; PDBe PDBsum structure summary Iron/manganese superoxide dismutases, C-terminal domain 
The structure of human mitochondrial manganese superoxide dismutase, which reveals a novel tetrameric interface of two 4-helix bundles.[7] Identifiers Symbol Sod_Fe_C Pfam PF02777 InterPro IPR001189 PROSITE PDOC00083 SCOP 1n0j Available protein structures: Pfam structures PDB RCSB PDB; PDBe PDBsum structure summary Nickel-containing superoxide dismutase 
Structure of nickel-containing superoxide dismutase.[9] Identifiers Symbol Sod_Ni Pfam PF09055 InterPro IPR014123 Available protein structures: Pfam structures PDB RCSB PDB; PDBe PDBsum structure summary In higher plants, SOD isozymes have been localized in different cell compartments. Mn-SOD is present in mitochondria and peroxisomes. Fe-SOD has been found mainly in chloroplasts but has also been detected in peroxisomes, and CuZn-SOD has been localized in cytosol, chloroplasts, peroxisomes, and apoplast.[11][12]
Human
In humans (as in all other mammals and most chordates), three forms of superoxide dismutase are present. SOD1 is located in the cytoplasm, SOD2 in the mitochondria, and SOD3 is extracellular. The first is a dimer (consists of two units), whereas the others are tetramers (four subunits). SOD1 and SOD3 contain copper and zinc, whereas SOD2 has manganese in its reactive centre. The genes are located on chromosomes 21, 6, and 4, respectively (21q22.1, 6q25.3 and 4p15.3-p15.1).
SOD1, soluble 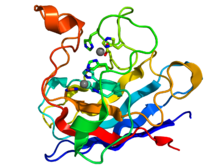
Crystallographic structure of the human SOD1 enzyme (rainbow colored N-terminus = blue, C-terminus = red) complexed with copper (blue-green sphere) and zinc (grey spheres).[13] Identifiers Symbol SOD1 Alt. symbols ALS, ALS1 Entrez 6647 HUGO 11179 OMIM 147450 RefSeq NM_000454 UniProt P00441 Other data EC number 1.15.1.1 Locus Chr. 21 q22.1 SOD2, mitochondrial 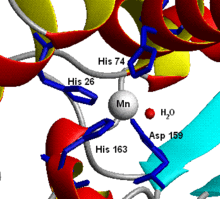
Structure of the active site of human superoxide dismutase 2.[1] Identifiers Symbol SOD2 Alt. symbols Mn-SOD; IPO-B; MVCD6 Entrez 6648 HUGO 11180 OMIM 147460 RefSeq NM_000636 UniProt P04179 Other data EC number 1.15.1.1 Locus Chr. 6 q25 SOD3, extracellular 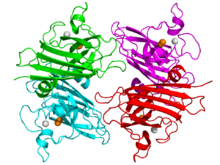
Crystallographic structure of the tetrameric human SOD3 enzyme (cartoon diagram) complexed with copper and zinc cations (orange and grey spheres respectively).[14] Identifiers Symbol SOD3 Alt. symbols EC-SOD; MGC20077 Entrez 6649 HUGO 11181 OMIM 185490 RefSeq NM_003102 UniProt P08294 Other data EC number 1.15.1.1 Locus Chr. 4 pter-q21 Plants
In higher plants, superoxide dismutase enzymes (SODs) act as antioxidants and protect cellular components from being oxidized by reactive oxygen species (ROS).[15] ROS can form as a result of drought, injury, herbicides and pesticides, ozone, plant metabolic activity, nutrient deficiencies, photoinhibition, temperature above and below ground, toxic metals, and UV or gamma rays.[16][17] Specifically, molecular O2 is reduced to O2- (an ROS called superoxide) when it absorbs an excited electron released from compounds of the electron transport chain. Superoxide is known to denature enzymes, oxidize lipids, and fragment DNA.[16] SODs catalyze the production of O2 and H2O2 from superoxide (O2-), which results in less harmful reactants.
When acclimating to increased levels of oxidative stress, SOD concentrations typically increase with the degree of stress conditions. The compartmentalization of different forms of SOD throughout the plant makes them counteract stress very effectively. There are three well-known and studied classes of SOD metallic coenzymes that exist in plants. First, Fe SODs consist of two species, one homodimer (containing 1-2 g Fe) and one tetramer (containing 2-4 g Fe). They are thought to be the most ancient SOD metalloenzymes and are found within both prokaryotes and eukaryotes. Fe SODs are most abundantly localized inside plant chloroplasts, where are they are indigenous. Second, Mn SODs consist of a homodimer and homotetramer species each containing a single Mn(III) atom per subunit. They are predominantly found in mitochondrion and peroxisomes. Third, Cu-Zn SODs have electrical properties very different from the other two classes. These are concentrated in the chloroplast, cytosol, and in some cases the extracellular space. Note that Cu-Zn SODs provide less protection than Fe SODs when localized in the chloroplast.[15][16][17]
Bacteria
Human white blood cells generate superoxide and other reactive oxygen species to kill bacteria. During infection, some bacteria (e.g., Burkholderia pseudomallei) therefore produce superoxide dismutase to protect themselves from being killed.[18]
Biochemistry
Simply stated, SOD outcompetes damaging reactions of superoxide, thus protecting the cell from superoxide toxicity. The reaction of superoxide with non-radicals is spin forbidden. In biological systems, this means its main reactions are with itself (dismutation) or with another biological radical such as nitric oxide (NO) or a metal. The superoxide anion radical (O2−) spontaneously dismutes to O2 and hydrogen peroxide (H2O2) quite rapidly (~105 M−1s−1 at pH 7). SOD is necessary because superoxide reacts with sensitive and critical cellular targets. For example, it reacts the NO radical, and makes toxic peroxynitrite. The dismutation rate is second order with respect to initial superoxide concentration. Thus, the half-life of superoxide, although very short at high concentrations (e.g., 0.05 seconds at 0.1mM) is actually quite long at low concentrations (e.g., 14 hours at 0.1 nM). In contrast, the reaction of superoxide with SOD is first order with respect to superoxide concentration. Moreover, superoxide dismutase has the largest kcat/KM (an approximation of catalytic efficiency) of any known enzyme (~7 x 109 M−1s−1),[19] this reaction being only limited by the frequency of collision between itself and superoxide. That is, the reaction rate is "diffusion limited". Even at the subnanomolar concentrations achieved by the high concentrations of SOD within cells, superoxide inactivates the citric acid cycle enzyme aconitase, can poison energy metabolism, and releases potentially toxic iron. Aconitase is one of several iron-sulfur containing (de)hydratases in metabolic pathways shown to be inactivated by superoxide.[20]
Physiology
Superoxide is one of the main reactive oxygen species in the cell, and, as such, SOD serves a key antioxidant role. The physiological importance of SODs is illustrated by the severe pathologies evident in mice genetically engineered to lack these enzymes. Mice lacking SOD2 die several days after birth, amid massive oxidative stress.[21] Mice lacking SOD1 develop a wide range of pathologies, including hepatocellular carcinoma,[22] an acceleration of age-related muscle mass loss,[23] an earlier incidence of cataracts and a reduced lifespan. Mice lacking SOD3 do not show any obvious defects and exhibit a normal lifespan, though they are more sensitive to hyperoxic injury.[24] Knockout mice of any SOD enzyme are more sensitive to the lethal effects of superoxide generating drugs, such as paraquat and diquat.
Drosophila lacking SOD1 have a dramatically shortened lifespan, whereas flies lacking SOD2 die before birth. SOD knockdowns in C. elegans do not cause major physiological disruptions. Knockout or null mutations in SOD1 are highly detrimental to aerobic growth in the yeast Sacchormyces cerevisiae and result in a dramatic reduction in post-diauxic lifespan. SOD2 knockout or null mutations cause growth inhibition on respiratory carbon sources in addition to decreased post-diauxic lifespan.
Several prokaryotic SOD null mutants have been generated, including E. Coli. The loss of periplasmic CuZnSOD causes loss of virulence and might be an attractive target for new antibiotics.
Role in disease
Mutations in the first SOD enzyme (SOD1) can cause familial amyotrophic lateral sclerosis (ALS, a form of motor neuron disease).[25][26][27] The most common mutation in the U.S. is A4V, while the most intensely studied is G93A. The other two isoforms of SOD have not been linked to any human diseases, however, in mice inactivation of SOD2 causes perinatal lethality[21] and inactivation of SOD1 causes hepatocellular carcinoma.[22] Mutations in SOD1 can cause familial ALS, by a mechanism that is presently not understood, but not due to loss of enzymatic activity or a decrease in the conformational stability of the SOD1 protein. Overexpression of SOD1 has been linked to the neural disorders seen in Down syndrome.[28] Genetic polymorphisms in SOD enzymes and their altered expressions and activities are associated with oxidative DNA damage and subsequently the individual’s risk of cancer susceptibility.[29]
SOD has proved to be highly effective in treatment of colonic inflammation in experimental colitis. Treatment with SOD decreases reactive oxygen species generation and oxidative stress and, thus, inhibits endothelial activation and indicate that modulation of factors that govern adhesion molecule expression and leukocyte-endothelial interactions. Therefore, such as antioxidants may be important new therapies for the treatment of inflammatory bowel disease.[30]
In recent years it has become more apparent that in mice the extracellular superoxide dismutase (SOD3, ecSOD) is critical in the development of hypertension.[31][32] In other studies, diminished SOD3 activity was linked to lung diseases such as Acute Respiratory Distress Syndrome (ARDS) or Chronic obstructive pulmonary disease (COPD).[33] [34] [35]
Superoxide dismutase is also not expressed in neural crest cells in the developing fetus. Hence, high levels of free radicals can cause damage to them and induce dysraphic anomalies (neural tube defects).
Cosmetic uses
SOD is used in cosmetic products to reduce free radical damage to skin, for example to reduce fibrosis following radiation for breast cancer. Studies of this kind must be regarded as tentative, however, as there were not adequate controls in the study including a lack of randomization, double-blinding, or placebo.[36] Superoxide dismutase is known to reverse fibrosis, perhaps through reversion of myofibroblasts back to fibroblasts.[37]
See also
References
- ^ a b PDB 1VAR; Borgstahl GE, Parge HE, Hickey MJ, Johnson MJ, Boissinot M, Hallewell RA, Lepock JR, Cabelli DE, Tainer JA (April 1996). "Human mitochondrial manganese superoxide dismutase polymorphic variant Ile58Thr reduces activity by destabilizing the tetrameric interface". Biochemistry 35 (14): 4287–97. doi:10.1021/bi951892w. PMID 8605177.
- ^ McCord JM, Fridovich I (1988). "Superoxide dismutase: the first twenty years (1968-1988)". Free Radic. Biol. Med. 5 (5–6): 363–9. doi:10.1016/0891-5849(88)90109-8. PMID 2855736.
- ^ Brewer GJ (September 1967). "Achromatic regions of tetrazolium stained starch gels: inherited electrophoretic variation". Am. J. Hum. Genet. 19 (5): 674–80. PMC 1706241. PMID 4292999. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1706241.
- ^ PDB 2SOD;Tainer JA, Getzoff ED, Beem KM, Richardson JS, Richardson DC (September 1982). "Determination and analysis of the 2 A-structure of copper, zinc superoxide dismutase". J. Mol. Biol. 160 (2): 181–217. doi:10.1016/0022-2836(82)90174-7. PMID 7175933.
- ^ Richardson JS, Thomas KA, Rubin BH, Richardson DC (1975). "Crystal Structure of Bovine Cu,Zn Superoxide Dismutase at 3Å Resolution: Chain Tracing and Metal Ligands". Proc Nat Acad Sci USA 72 (4): 1349–53. doi:10.1073/pnas.72.4.1349. PMC 432531. PMID 1055410. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=432531..
- ^ Tainer JA, Getzoff ED, Richardson JS, Richardson DC (1983). "Structure and mechanism of copper, zinc superoxide dismutase". Nature 306 (5940): 284–7. doi:10.1038/306284a0. PMID 6316150..
- ^ a b c PDB 1N0J; Borgstahl GE, Parge HE, Hickey MJ, Beyer WF Jr, Hallewell RA, Tainer JA (1992). "The structure of human mitochondrial manganese superoxide dismutase reveals a novel tetrameric interface of two 4-helix bundles". Cell 71 (1): 107–18. doi:10.1016/0092-8674(92)90270-M. PMID 1394426.
- ^ Barondeau DP, Kassmann CJ, Bruns CK, Tainer JA, Getzoff ED (2004). "Nickel superoxide dismutase structure and mechanism". Biochemistry 43 (25): 8038–47. doi:10.1021/bi0496081. PMID 15209499.
- ^ a b PDB 1Q0M; Wuerges J, Lee JW, Yim YI, Yim HS, Kang SO, Djinovic Carugo K (2004). "Crystal structure of nickel-containing superoxide dismutase reveals another type of active site". Proc Natl Acad Sci 101 (23): 8569–74. doi:10.1073/pnas.0308514101. PMC 423235. PMID 15173586. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=423235.
- ^ PDB 1SDY; Djinović K, Gatti G, Coda A et al. (December 1991). "Structure solution and molecular dynamics refinement of the yeast Cu,Zn enzyme superoxide dismutase". Acta Crystallogr. B 47 (6): 918–27. doi:10.1107/S0108768191004949. PMID 1772629.
- ^ Corpas FJ, Barroso JB, del Río LA (April 2001). "Peroxisomes as a source of reactive oxygen species and nitric oxide signal molecules in plant cells". Trends Plant Sci. 6 (4): 145–50. doi:10.1016/S1360-1385(01)01898-2. PMID 11286918. http://linkinghub.elsevier.com/retrieve/pii/S1360-1385(01)01898-2.
- ^ Corpas FJ, Fernández-Ocaña A, Carreras A, Valderrama R, Luque F, Esteban FJ, Rodríguez-Serrano M, Chaki M, Pedrajas JR, Sandalio LM, del Río LA, Barroso JB (July 2006). "The expression of different superoxide dismutase forms is cell-type dependent in olive (Olea europaea L.) leaves". Plant Cell Physiol. 47 (7): 984–94. doi:10.1093/pcp/pcj071. PMID 16766574.
- ^ PDB 3CQQ; Cao X, Antonyuk SV, Seetharaman SV, Whitson LJ, Taylor AB, Holloway SP, Strange RW, Doucette PA, Valentine JS, Tiwari A, Hayward LJ, Padua S, Cohlberg JA, Hasnain SS, Hart PJ (June 2008). "Structures of the G85R variant of SOD1 in familial amyotrophic lateral sclerosis". J. Biol. Chem. 283 (23): 16169–77. doi:10.1074/jbc.M801522200. PMC 2414278. PMID 18378676. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2414278.
- ^ PDB 2JLP; Antonyuk SV, Strange RW, Marklund SL, Hasnain SS (May 2009). "The structure of human extracellular copper-zinc superoxide dismutase at 1.7 A resolution: insights into heparin and collagen binding". J. Mol. Biol. 388 (2): 310–26. doi:10.1016/j.jmb.2009.03.026. PMID 19289127.
- ^ a b Alscher RG, Erturk N, Heath LS (May 2002). "Role of superoxide dismutases (SODs) in controlling oxidative stress in plants". J. Exp. Bot. 53 (372): 1331–41. doi:10.1093/jexbot/53.372.1331. PMID 11997379.
- ^ a b c Smirnoff, Nicholas (1993). "Tansley Review No. 52 The role of active oxygen in the response of plants to water deficit and desiccation". Plant Phytology 125.
- ^ a b Raychaudhuri, Sarmistha; Xing Deng. (2008). "The Role of Superoxide Dismutase in Combating Oxidative Stress in Higher Plants". The Botanical Review 66 (1): 89–98.
- ^ Vanaporn M, Wand M, Michell SL et al. (2011). "Superoxide dismutase C is required for intracellular survival and virulence of Burkholderia pseudomallei". Microbiology 157 (Pt 8): 2392–400. doi:10.1099/mic.0.050823-0. PMID 21659326.
- ^ Heinrich, Peter; Georg Löffler; Petro E. Petrides (2006). Biochemie und Pathobiochemie (Springer-Lehrbuch) (German Edition). Berlin: Springer. pp. 123. ISBN 3-540-32680-4.
- ^ Gardner PR, Raineri I, Epstein LB, White CW (June 1995). "Superoxide radical and iron modulate aconitase activity in mammalian cells". J. Biol. Chem. 270 (22): 13399–405. doi:10.1074/jbc.270.22.13399. PMID 7768942.
- ^ a b Li Y, Huang TT, Carlson EJ, Melov S, Ursell PC, Olson JL, Noble LJ, Yoshimura MP, Berger C, Chan PH, Wallace DC, Epstein CJ (December 1995). "Dilated cardiomyopathy and neonatal lethality in mutant mice lacking manganese superoxide dismutase". Nat. Genet. 11 (4): 376–81. doi:10.1038/ng1295-376. PMID 7493016.
- ^ a b Elchuri S, Oberley TD, Qi W, Eisenstein RS, Jackson Roberts L, Van Remmen H, Epstein CJ, Huang TT (January 2005). "CuZnSOD deficiency leads to persistent and widespread oxidative damage and hepatocarcinogenesis later in life". Oncogene 24 (3): 367–80. doi:10.1038/sj.onc.1208207. PMID 15531919.
- ^ Muller FL, Song W, Liu Y, Chaudhuri A, Pieke-Dahl S, Strong R, Huang TT, Epstein CJ, Roberts LJ, Csete M, Faulkner JA, Van Remmen H (June 2006). "Absence of CuZn superoxide dismutase leads to elevated oxidative stress and acceleration of age-dependent skeletal muscle atrophy". Free Radic. Biol. Med. 40 (11): 1993–2004. doi:10.1016/j.freeradbiomed.2006.01.036. PMID 16716900.
- ^ Sentman ML, Granström M, Jakobson H, Reaume A, Basu S, Marklund SL (March 2006). "Phenotypes of mice lacking extracellular superoxide dismutase and copper- and zinc-containing superoxide dismutase". J. Biol. Chem. 281 (11): 6904–9. doi:10.1074/jbc.M510764200. PMID 16377630.
- ^ Deng HX, Hentati A, Tainer JA, Iqbal Z, Cayabyab A, Hung WY, Getzoff ED, Hu P et al. (August 1993). "Amyotrophic lateral sclerosis and structural defects in Cu,Zn superoxide dismutase". Science. 261 (5124): 1047–51. doi:10.1126/science.8351519. PMID 8351519.
- ^ Conwit RA (December 2006). "Preventing familial ALS: a clinical trial may be feasible but is an efficacy trial warranted?". J. Neurol. Sci. 251 (1–2): 1–2. doi:10.1016/j.jns.2006.07.009. PMID 17070848.
- ^ Al-Chalabi A, Leigh PN (August 2000). "Recent advances in amyotrophic lateral sclerosis". Curr. Opin. Neurol. 13 (4): 397–405. doi:10.1097/00019052-200008000-00006. PMID 10970056. http://meta.wkhealth.com/pt/pt-core/template-journal/lwwgateway/media/landingpage.htm?issn=1350-7540&volume=13&issue=4&spage=397.
- ^ Groner Y, Elroy-Stein O, Avraham KB, Schickler M, Knobler H, Minc-Golomb D, Bar-Peled O, Yarom R, Rotshenker S (1994). "Cell damage by excess CuZnSOD and Down syndrome". Biomed. Pharmacother. 48 (5–6): 231–40. doi:10.1016/0753-3322(94)90138-4. PMID 7999984.
- ^ Khan MA, Tania M, Zhang D, Chen H (2010). "Antioxidant enzymes and cancer". Chin J Cancer Res 22 (2): 87–92. doi:10.1007/s11670-010-0087-7.
- ^ Seguí J, Gironella M, Sans M, Granell S, Gil F, Gimeno M, Coronel P, Piqué JM, Panés J (September 2004). "Superoxide dismutase ameliorates TNBS-induced colitis by reducing oxidative stress, adhesion molecule expression, and leukocyte recruitment into the inflamed intestine". J. Leukoc. Biol. 76 (3): 537–44. doi:10.1189/jlb.0304196. PMID 15197232.
- ^ Gongora, Maria C.; Qin Z, Laude K, Kim HW, McCann L, Folz JR, Dikalov S, Fukai T, Harrison DG. (September 2006). "Role of extracellular superoxide dismutase in hypertension". Hypertension 48 (3): 473–81. doi:10.1161/01.HYP.0000235682.47673.ab. PMID 16864745.
- ^ Lob, HE; Marvar, PJ; Guzik, TJ; Sharma, S; McCann, LA; Weyand, C; Gordon, FJ; Harrison, DG (2010). "Induction of hypertension and peripheral inflammation by reduction of extracellular superoxide dismutase in the central nervous system". Hypertension 55 (2): 277–83, 6p following 283. doi:10.1161/HYPERTENSIONAHA.109.142646. PMC 2813894. PMID 20008675. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2813894.
- ^ Young, RP; Hopkins, R; Black, PN; Eddy, C; Wu, L; Gamble, GD; Mills, GD; Garrett, JE et al. (2006). "Functional variants of antioxidant genes in smokers with COPD and in those with normal lung function". Thorax 61 (5): 394–9. doi:10.1136/thx.2005.048512. PMC 2111196. PMID 16467073. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2111196.
- ^ Ganguly, K; Depner, M; Fattman, C; Bein, K; Oury, TD; Wesselkamper, SC; Borchers, MT; Schreiber, M et al. (2009). "Superoxide dismutase 3, extracellular (SOD3) variants and lung function". Physiological genomics 37 (3): 260–7. doi:10.1152/physiolgenomics.90363.2008. PMC 2685504. PMID 19318538. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2685504.
- ^ Gongora, MC; Lob, HE; Landmesser, U; Guzik, TJ; Martin, WD; Ozumi, K; Wall, SM; Wilson, DS et al. (2008). "Loss of extracellular superoxide dismutase leads to acute lung damage in the presence of ambient air: A potential mechanism underlying adult respiratory distress syndrome". The American journal of pathology 173 (4): 915–26. doi:10.2353/ajpath.2008.080119. PMC 2543061. PMID 18787098. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2543061.
- ^ Campana F, Zervoudis S, Perdereau B, Gez E, Fourquet A, Badiu C, Tsakiris G, Koulaloglou S (2004). "Topical superoxide dismutase reduces post-irradiation breast cancer fibrosis". J. Cell. Mol. Med. 8 (1): 109–16. doi:10.1111/j.1582-4934.2004.tb00265.x. PMID 15090266.
- ^ Vozenin-Brotons MC, Sivan V, Gault N, Renard C, Geffrotin C, Delanian S, Lefaix JL, Martin M (January 2001). "Antifibrotic action of Cu/Zn SOD is mediated by TGF-beta1 repression and phenotypic reversion of myofibroblasts". Free Radic. Biol. Med. 30 (1): 30–42. doi:10.1016/S0891-5849(00)00431-7. PMID 11134893.
External links
- Online 'Mendelian Inheritance in Man' (OMIM) 105400 (ALS)
- The ALS Online Database
- A short but substantive overview of SOD and its literature.
- Damage-Based Theories of Aging Includes a discussion of the roles of SOD1 and SOD2 in aging.
- Physicians' Comm. For Responsible Med.
- SOD and Oxidative Stress Pathway Image
- Historical information on SOD research"The evolution of Free Radical Biology & Medicine: A 20-year history" and "Free Radical Biology & Medicine The last 20 years: The most highly cited papers"
- JM McCord discusses the discovery of SOD
Other oxidoreductases (EC 1.15-1.18) 1.15: Acting on superoxide as acceptor 1.16: Oxidizing metal ions 1.17: Acting on CH or CH2 groups 1.18: Acting on iron-sulfur proteins as donors 1.19: Acting on reduced flavodoxin as donor 1.20: Acting on phosphorus or arsenic in donors 1.21: Acting on X-H and Y-H to form an X-Y bond Anti-inflammatory products (M01A) Pyrazolidine/Butylpyrazolidines Ampyrone • Clofezone • Kebuzone • Metamizole • Mofebutazone • Oxyphenbutazone • Phenazone • Phenylbutazone • Sulfinpyrazone • Feprazone •Acetic acid derivatives
and related substancesAceclofenac • Acemetacin • Alclofenac • Bromfenac • Bumadizone • Bufexamac • Diclofenac • Difenpiramide • Etodolac • Fentiazac • Indometacin • Ketorolac • Lonazolac • Oxametacin • Proglumetacin • Sulindac • Tolmetin • Zomepirac • AmfenacOxicams Propionic acid derivatives Alminoprofen • Benoxaprofen • Dexibuprofen • Dexketoprofen • Fenbufen • Fenoprofen • Flunoxaprofen • Flurbiprofen • Ibuprofen • Ibuproxam • Indoprofen • Ketoprofen • Naproxen • Oxaprozin • Pirprofen • Suprofen • Tiaprofenic acidFenamates Coxibs Other Nabumetone • Glucosamine • Benzydamine • Glycosaminoglycan • Magnesium salicylate • Proquazone • Superoxide dismutase/Orgotein • Nimesulide • Diacerein • Tenidap • Oxaceprol • Chondroitin sulfateCategories:- Genes on chromosome 21
- Genes on chromosome 6
- Genes on chromosome 4
- Antioxidants
- Metalloproteins
- Oxidoreductases
- EC 1.15.1
- Copper enzymes
Wikimedia Foundation. 2010.

