- DUSP3
-
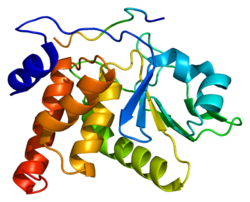
Dual specificity protein phosphatase 3 is an enzyme that in humans is encoded by the DUSP3 gene.[1][2]
The protein encoded by this gene is a member of the dual specificity protein phosphatase subfamily. These phosphatases inactivate their target kinases by dephosphorylating both the phosphoserine/threonine and phosphotyrosine residues. They negatively regulate members of the mitogen-activated protein (MAP) kinase superfamily (MAPK/ERK, SAPK/JNK, p38), which are associated with cellular proliferation and differentiation. Different members of the family of dual specificity phosphatases show distinct substrate specificities for various MAP kinases, different tissue distribution and subcellular localization, and different modes of inducibility of their expression by extracellular stimuli. This gene maps in a region that contains the BRCA1 locus which confers susceptibility to breast and ovarian cancer. Although DUSP3 is expressed in both breast and ovarian tissues, mutation screening in breast cancer pedigrees and in sporadic tumors was negative, leading to the conclusion that this gene is not BRCA1.[2]
Interactions
DUSP3 has been shown to interact with MAPK3[3] and MAPK1.[3]
References
- ^ Folander K, Douglass J, Swanson R (Feb 1995). "Confirmation of the assignment of the gene encoding Kv1.3, a voltage-gated potassium channel (KCNA3) to the proximal short arm of human chromosome 1". Genomics 23 (1): 295–6. doi:10.1006/geno.1994.1500. PMID 7829094.
- ^ a b "Entrez Gene: DUSP3 dual specificity phosphatase 3 (vaccinia virus phosphatase VH1-related)". http://www.ncbi.nlm.nih.gov/sites/entrez?Db=gene&Cmd=ShowDetailView&TermToSearch=1845.
- ^ a b Todd, J L; Tanner K G, Denu J M (May. 1999). "Extracellular regulated kinases (ERK) 1 and ERK2 are authentic substrates for the dual-specificity protein-tyrosine phosphatase VHR. A novel role in down-regulating the ERK pathway". J. Biol. Chem. (UNITED STATES) 274 (19): 13271–80. doi:10.1074/jbc.274.19.13271. ISSN 0021-9258. PMID 10224087.
Further reading
- Ishibashi T, Bottaro DP, Chan A, et al. (1993). "Expression cloning of a human dual-specificity phosphatase". Proc. Natl. Acad. Sci. U.S.A. 89 (24): 12170–4. doi:10.1073/pnas.89.24.12170. PMC 50720. PMID 1281549. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=50720.
- Kamb A, Futreal PA, Rosenthal J, et al. (1995). "Localization of the VHR phosphatase gene and its analysis as a candidate for BRCA1". Genomics 23 (1): 163–7. doi:10.1006/geno.1994.1473. PMID 7829067.
- Jones KA, Black DM, Brown MA, et al. (1995). "The detailed characterisation of a 400 kb cosmid walk in the BRCA1 region: identification and localisation of 10 genes including a dual-specificity phosphatase". Hum. Mol. Genet. 3 (11): 1927–34. doi:10.1093/hmg/3.11.1927. PMID 7874108.
- Yuvaniyama J, Denu JM, Dixon JE, Saper MA (1996). "Crystal structure of the dual specificity protein phosphatase VHR". Science 272 (5266): 1328–31. doi:10.1126/science.272.5266.1328. PMID 8650541.
- Todd JL, Tanner KG, Denu JM (1999). "Extracellular regulated kinases (ERK) 1 and ERK2 are authentic substrates for the dual-specificity protein-tyrosine phosphatase VHR. A novel role in down-regulating the ERK pathway". J. Biol. Chem. 274 (19): 13271–80. doi:10.1074/jbc.274.19.13271. PMID 10224087.
- Alonso A, Saxena M, Williams S, Mustelin T (2001). "Inhibitory role for dual specificity phosphatase VHR in T cell antigen receptor and CD28-induced Erk and Jnk activation". J. Biol. Chem. 276 (7): 4766–71. doi:10.1074/jbc.M006497200. PMID 11085983.
- Najarro P, Traktman P, Lewis JA (2001). "Vaccinia virus blocks gamma interferon signal transduction: viral VH1 phosphatase reverses Stat1 activation". J. Virol. 75 (7): 3185–96. doi:10.1128/JVI.75.7.3185-3196.2001. PMC 114112. PMID 11238845. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=114112.
- Alonso A, Rahmouni S, Williams S, et al. (2003). "Tyrosine phosphorylation of VHR phosphatase by ZAP-70". Nat. Immunol. 4 (1): 44–8. doi:10.1038/ni856. PMID 12447358.
- Strausberg RL, Feingold EA, Grouse LH, et al. (2003). "Generation and initial analysis of more than 15,000 full-length human and mouse cDNA sequences". Proc. Natl. Acad. Sci. U.S.A. 99 (26): 16899–903. doi:10.1073/pnas.242603899. PMC 139241. PMID 12477932. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=139241.
- Kim HS, Song MC, Kwak IH, et al. (2003). "Constitutive induction of p-Erk1/2 accompanied by reduced activities of protein phosphatases 1 and 2A and MKP3 due to reactive oxygen species during cellular senescence". J. Biol. Chem. 278 (39): 37497–510. doi:10.1074/jbc.M211739200. PMID 12840032.
- Ota T, Suzuki Y, Nishikawa T, et al. (2004). "Complete sequencing and characterization of 21,243 full-length human cDNAs". Nat. Genet. 36 (1): 40–5. doi:10.1038/ng1285. PMID 14702039.
- Gerhard DS, Wagner L, Feingold EA, et al. (2004). "The status, quality, and expansion of the NIH full-length cDNA project: the Mammalian Gene Collection (MGC)". Genome Res. 14 (10B): 2121–7. doi:10.1101/gr.2596504. PMC 528928. PMID 15489334. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=528928.
- Rual JF, Venkatesan K, Hao T, et al. (2005). "Towards a proteome-scale map of the human protein-protein interaction network". Nature 437 (7062): 1173–8. doi:10.1038/nature04209. PMID 16189514.
- Rahmouni S, Cerignoli F, Alonso A, et al. (2006). "Loss of the VHR dual-specific phosphatase causes cell-cycle arrest and senescence". Nat. Cell Biol. 8 (5): 524–31. doi:10.1038/ncb1398. PMID 16604064.
- Hao L, ElShamy WM (2007). "BRCA1-IRIS activates cyclin D1 expression in breast cancer cells by downregulating the JNK phosphatase DUSP3/VHR". Int. J. Cancer 121 (1): 39–46. doi:10.1002/ijc.22597. PMID 17278098.
- Hoyt R, Zhu W, Cerignoli F, et al. (2007). "Cutting edge: selective tyrosine dephosphorylation of interferon-activated nuclear STAT5 by the VHR phosphatase". J. Immunol. 179 (6): 3402–6. PMC 2770724. PMID 17785772. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2770724.
PDB gallery Esterase: protein tyrosine phosphatases (EC 3.1.3.48) Class I Classical PTPsReceptor type PTPs (PTPRA, PTPRB, PTPRC, PTPRD, PTPRE, PTPRF, PTPRG, PTPRH, PTPRJ, PTPRK, PTPRM, PTPRN, PTPRN2, PTPRO, PTPRQ, PTPRR, PTPRS, PTPRT, PTPRU, PTPRZ)
Non receptor type PTPs (PTPN1, PTPN2, PTPN3, PTPN4, PTPN5, PTPN6, PTPN7, PTPN9, PTPN11, PTPN12, PTPN13, PTPN14, PTPN18, PTPN20, PTPN21, PTPN22, PTPN23MAPK phosphatases (MKPs) (DUSP1, DUSP2, DUSP4, DUSP5, DUSP6, DUSP7, DUSP8, DUSP9, DUSP10, DUSP16, MK-STYX)
CDC14s (CDC14A, CDC14B, CDKN3, PTP9Q22)
Atypical DSPs (DUSP3, DUSP11, DUSP12, DUSP13A, DUSP13B, DUSP14, DUSP15, DUSP18, DUSP19, DUSP21, DUSP22, DUSP23, DUSP24, DUSP25, DUSP26, DUSP27, EMP2A, RNGTT, STYX)
Phosphatase and tensin homologs (PTENs) (PTEN, TPIP, TPTE, TNS, TENC1)
Myotubularins (MTM1, MTMR2, MTMR3, MTMR4, MTMR5, MTMR6, MTMR7, MTMR8, MTMR9, MTMR10, MTMR11, MTMR12, MTMR13, MTMR14, MTMR15)Class II Class III Class IV B enzm: 1.1/2/3/4/5/6/7/8/10/11/13/14/15-18, 2.1/2/3/4/5/6/7/8, 2.7.10, 2.7.11-12, 3.1/2/3/4/5/6/7, 3.1.3.48, 3.4.21/22/23/24, 4.1/2/3/4/5/6, 5.1/2/3/4/99, 6.1-3/4/5-6 Categories:- Human proteins
- Chromosome 17 gene stubs
Wikimedia Foundation. 2010.