- Complex regional pain syndrome
-
Complex regional pain syndrome/Reflex Sympathetic Dystrophy (CRPS/RSD) Classification and external resources ICD-10 M89.0, G56.4 ICD-9 337.21, 337.22, 354.4, 355.71 DiseasesDB 12635 16345 eMedicine pmr/123 MeSH D020918 Complex regional pain syndrome (CRPS) is a chronic progressive disease characterized by severe pain, swelling and changes in the skin. It often affects an arm or a leg and may spread to another part of the body.Though treatment is often unsatisfactory, early multimodal therapy can cause dramatic improvement or remission of the syndrome in some patients.[1] The International Association for the Study of Pain has proposed dividing CRPS into two types based on the presence of nerve lesion following the injury.
- Type I, formerly known as reflex sympathetic dystrophy (RSD), Sudeck's atrophy, reflex neurovascular dystrophy (RND) or algoneurodystrophy, does not have demonstrable nerve lesions.
- Type II, formerly known as causalgia, has evidence of obvious nerve damage.
The cause of this syndrome is currently unknown. Precipitating factors include injury and surgery, although there are documented cases that have no demonstrable injury to the original site.
Contents
History and nomenclature
The condition currently known as CRPS was originally described during the American Civil War by Silas Weir Mitchell, who is sometimes also credited with inventing the name "causalgia."[2] However, this term was actually coined by Mitchell's friend Robley Dunglison from the Greek words for heat and for pain.[3] Contrary to what is commonly accepted, it emerges that these causalgias were certainly major by the importance of the vasomotor and sudomotor symptoms, but stemmed from minor neurological lesions. Mitchell even thought that the CRPS etiology came from the cohabitation of the altered and unaltered cutaneous fibres on the same nerve distribution territory.[4] In the 1940s, the term reflex sympathetic dystrophy came into use to describe this condition, based on the theory that sympathetic hyperactivity was involved in the pathophysiology.[5] In 1959, Noordenbos observed in caulsalgia patients that "The damage of the nerve is always partial".[6] Misuse of the terms, as well as doubts about the underlying pathophysiology, led to calls for better nomenclature. In 1993, a special consensus workshop held in Orlando, Florida, provided the umbrella term "complex regional pain syndrome," with causalgia and RSD as subtypes.[7]
Pathophysiology
The pathophysiology of CRPS is not fully understood. “Physiological wind-up” and central nervous system (CNS) sensitization, are key neurologic processes that appear to be involved in the induction and maintenance of CRPS.[8] There is compelling evidence that the N-methyl-D-aspartate (NMDA) receptor has significant involvement in the CNS sensitization process.[9] It is also hypothesized that elevated CNS glutamate levels promote "physiological wind-up" and CNS sensitization.[8] In addition, there is experimental evidence that demonstrates NMDA receptors in peripheral nerves.[10] Because immunological functions can modulate CNS physiology, it has also been hypothesized that a variety of immune processes may contribute to the initial development and maintenance of peripheral and central sensitization.[11][12] Furthermore, trauma related cytokine release, exaggerated neurogenic inflammation, sympathetic afferent coupling, adrenoreceptor pathology, glial cell activation, cortical reorganisation,[13] and oxidative damage (e.g. by free radicals) are all concepts that have been implicated in the pathophysiology of CRPS.[14]
Susceptibility
CRPS can strike at any age, but the mean age at diagnosis is 42.[15] CRPS has been diagnosed in children as young as 2 years old.[16] It affects both men and women; however, CRPS is 3 times more frequent in females than males.[15] The number of reported CRPS cases among adolescents and young adults is increasing.[17]
Investigators estimate that 2-5 percent of those with peripheral nerve injury,[15] and 13-70 percent of those with hemiplegia (paralysis of one side of the body),[18] will suffer from CRPS. In addition, some studies have indicated that cigarette smoking was strikingly present in patients and is statistically linked to RSD. In one study, 68% of patients versus 37% of hospitalized controls were found. This may be involved in its pathology by enhancing sympathetic activity, vasoconstriction, or by some other unknown neurotransmitter-related mechanism.[19]
It is also theorized that certain people might be genetically predisposed to develop symptoms of RSD/CRPS after a significant or seemingly insignificant injury has been sustained.[20] These tests are being performed by The Reflex Sympathetic Dystrophy Syndrome Association (RSDSA), American RSD Hope, and Richard G. Boles, M.D. Research began in October 2008, but the outcome has yet to be released to the medical community.[21]
Symptoms
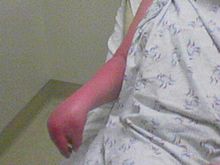
The symptoms of CRPS usually manifest near the site of an injury, which is usually minor. The most common symptoms overall are burning and electrical sensations, described to be like "shooting pain". The patient may also experience muscle spasms, local swelling, abnormally increased sweating, changes in skin temperature (usually hot but sometimes cold) and color (bright red or a reddish violet), softening and thinning of bones, joint tenderness or stiffness, and/or restricted or painful movement.
The pain of CRPS is continuous and may be heightened by emotional or physical stress. Moving or touching the limb is often intolerable. The symptoms of CRPS vary in severity and duration. There are three variants of CRPS, previously thought of as stages. It is now believed that patients with CRPS do not progress through these stages sequentially. These stages may not be time-constrained, and could possibly event-related, such as ground-level falls or re-injuries in previous areas. It is important to remember that often the parasympathetic nervous system is involved with CRPS, and a part (subset) of the parasympathetic system is the autonomic (think automatic, like blood pressure regulation or breathing or sweating) nervous system can go haywire and cause a wide variety of odd complaints that are not mental in origin. Be sure and investigate autonomic dysfunction or disorder if you think you may have one of the often distinct varieties of CRPS. Rather than a progression of CRPS from bad to worse, it is now thought, instead, patients are likely to have one of the three following types of disease progression:
- Stage one is characterized by severe, burning pain at the site of the injury. Muscle spasm, joint stiffness, restricted mobility, rapid hair and nail growth, and vasospasm. The vasospasm is that which causes the changes in the color and temperature of the skin.
- Stage two is characterized by more intense pain. Swelling spreads, hair growth diminishes, nails become cracked, brittle, grooved, and spotty, osteoporosis becomes severe and diffuse, joints thicken, and muscles atrophy.
- Stage three is characterized by irreversible changes in the skin and bones, while the pain becomes unyielding and may involve the entire limb. There is marked muscle atrophy, severely limited mobility of the affected area, and flexor tendon contractions (contractions of the muscles and tendons that flex the joints). Occasionally the limb is displaced from its normal position, and marked bone softening and thinning is more dispersed.
Diagnosis
CRPS types I and II share the common diagnostic criteria shown below. Spontaneous pain or allodynia (pain resulting from a stimulus which would not normally provoke pain, such as a light touch of the skin) is not limited to the territory of a single peripheral nerve, and is disproportionate to the inciting event.
- There is a history of edema, skin blood flow abnormality, or abnormal sweating in the region of the pain since the inciting event.
- No other conditions can account for the degree of pain and dysfunction.
The two types differ only in the nature of the inciting event. Type I CRPS develops following an initiating noxious event that may or may not have been traumatic, while type II CRPS develops after a nerve injury.
No specific test is available for CRPS, which is diagnosed primarily through observation of the symptoms. However, thermography, sweat testing, x-rays, electrodiagnostics, and sympathetic blocks can be used to build up a picture of the disorder. Diagnosis is complicated by the fact that some patients improve without treatment. A delay in diagnosis and/or treatment for this syndrome can result in severe physical and psychological problems. Early recognition and prompt treatment provide the greatest opportunity for recovery.
The International Association for the Study of Pain (IASP) lists the diagnostic criteria for complex regional pain syndrome I (CRPS I) (RSDS) as follows:
- The presence of an initiating noxious event or a cause of immobilization
- Continuing pain, allodynia (perception of pain from a nonpainful stimulus), or hyperalgesia (an exaggerated sense of pain) disproportionate to the inciting event.
- Evidence at some time of edema, changes in skin blood flow, or abnormal sudomotor activity in the area of pain
- The diagnosis is excluded by the existence of any condition that would otherwise account for the degree of pain and dysfunction.
According to the IASP, CRPS II (causalgia) is diagnosed as follows:
- The presence of continuing pain, allodynia, or hyperalgesia after a nerve injury, not necessarily limited to the distribution of the injured nerve
- Evidence at some time of edema, changes in skin blood flow, or abnormal sudomotor activity in the region of pain
- The diagnosis is excluded by the existence of any condition that would otherwise account for the degree of pain and dysfunction.
The IASP criteria for CRPS I diagnosis has shown a sensitivity ranging from 98-100% and a specificity ranging from 36%-55%. Per the IASP guidelines, interobserver reliability for CRPS I diagnosis is poor. Two other criteria used for CRPS I diagnosis are Bruehl's criteria and Veldman's criteria which have moderate to good interobserver reliability. In the absence of clear evidence supporting 1 set of criteria over the others, clinicians may use IASP, Bruehl’s, or Veldman’s clinical criteria for diagnosis. While the IASP criteria are nonspecific and possibly not as reproducible as Bruehl’s or Veldman’s criteria, they are cited more widely the literature including treatment trials.[22]
Thermography
Presently, established empirical evidence suggests against thermography's efficacy as a reliable tool for diagnosing CRPS. Although CRPS may, in some cases, lead to measurably-altered blood flow throughout an affected region, many other factors can also contribute to an altered thermographic reading, including the patient's smoking habits, use of certain skin lotions, recent physical activity, and prior history of trauma to the region. Also, not all patients diagnosed with CRPS demonstrate such "vasomotor instability"—less often, still, those in the later stages of the disease.[23] Thus, thermography alone cannot be used as conclusive evidence for- or against- a diagnosis of CRPS, and must be interpreted in light of the patient's larger medical history and prior diagnostic studies.[24]
In order to minimize the confounding influence of external factors, patients undergoing infrared thermographic testing must conform to special restrictions regarding the use of certain vasoconstrictors (namely, nicotine and caffeine), skin lotions, physical therapy, and other diagnostic procedures in the days prior to testing. Patients may also be required to discontinue certain pain medications and sympathetic blockers. After a patient arrives at a thermographic laboratory, he or she is allowed to reach thermal equilibrium in a 16–20 °C, draft-free, steady-state room wearing a loose fitting cotton hospital gown for approximately 20 minutes. A technician then takes infrared images of both the patient's affected- and unaffected- limbs, as well as reference images of other parts of the patient's body including his or her face, upper-back, and lower-back. After capturing a set of baseline images, some labs further require the patient to undergo cold-water autonomic-functional-stress-testing to evaluate the function of his or her autonomic nervous system's peripheral vasoconstrictor reflex. This is performed by placing a patient's unaffected limb in a cold-water bath (approximately 20 °C) for five minutes while collecting images. In a normal-intact, functioning autonomic nervous system, a patient's affected extremity will become colder. Conversely, warming of an affected extremity may indicate a disruption of the body's normal thermoregulatory vasoconstrictor function, which may sometimes indicate underlying CRPS.[25]
Sweat testing
Abnormal sweating can be detected by several tests. A powder that changes color when exposed to sweat can be applied to the limbs; however, this method does not allow for quantification of sweating. Two quantitative tests that may be used are the resting sweat output test and the quantitative sudomotor axon reflex test. These quantitative sweat tests have been shown to correlate with clinical signs of CRPS.[26]
Radiography
Patchy osteoporosis, which may be due to disuse of the affected extremity, can be detected through X-ray imagery as early as two weeks after the onset of CRPS. A bone scan of the affected limb may detect these changes even sooner. Bone densitometry can also be used to detect changes in bone mineral density. It can also be used to monitor the results of treatment, as bone densitometry parameters improve with treatment.
Electrodiagnostic testing
EMG (Electromyography testing) should not be done for the diagnosis of CRPS I or II. It is extremely painful for patients suffering from CRPS and may even be considered unethical and cruel. This is just wrong. The EMG/NCS may be the only way to identify underlying nerve injury or alternative diagnoses.
Treatment
The general strategy in CRPS treatment is often multi-disciplinary, with the use of different types of medications combined with distinct physical therapies. Similar to the treatment principles in children and teenagers.
Physical and occupational therapy
Physical and occupational therapy are important components of the management of CRPS primarily by desensitizing the affected body part, restoring motion, and improving function. Though it should be noted some people at certain stages of the disease are incapable of participating in physical therapy due to touch intolerance. This may be where Graded Motor Imagery and Mirror Therapy (see below) are particularly helpful. People with CRPS often develop guarding behaviors where they avoid using or touching the affected limb. This inactivity exacerbates the disease and perpetuates the pain cycle. Therefore optimizing the multimodal treatment is paramount to allow for use of the involved body part. Physical therapy works best for most patients, especially goal-directed therapy, where the patient begins from an initial point, regardless of how minimal, and then endeavors to increase activity each week. Therapy is directed at facilitating the patient to engage in physical therapy, movement and stimulation of the affected areas. One difficulty with the idea of Physical Therapy, however, is that it means different things to different people. There is one systematic review of the use of physical and occupational therapy for the treatment of CRPS.[27] That review concluded: "Narrative synthesis of the results, based on effect size, found there was good to very good quality level II evidence that graded motor imagery is effective in reducing pain in adults with CRPS-1, irrespective of the outcome measure used. No evidence was found to support treatments frequently recommended in clinical guidelines, such as stress loading. CONCLUSIONS: Graded motor imagery should be used to reduce pain in adult CRPS-1 patients."
Physical therapy has been used under light general anesthesia in an attempt to remobilize the extremity. Such remobilization is used cautiously to avoid damage to atrophied tissue and bones which have become osteodystrophic.
Drugs
Physicians use a variety of drugs to treat CRPS, including antidepressants, anti-inflammatories such as corticosteroids and COX-inhibitors such as piroxicam, bisphosphonates, vasodilators, GABA analogs such gabapentin and pregabalin, and alpha- or beta-adrenergic-blocking compounds, and the entire pharmacy of opioids. Although many different drugs are used, there is not much supportive evidence for most of them. This doesn't necessarily reflect evidence that they don't work, just a lack of evidence that they do.
Mirror box therapy
Recent research has utilized mirror therapy to significantly reduce pain levels in CRPS patients, where the affected limb is placed within a mirror box, such that the unaffected limb is reflected in a way as to make the patient think they are looking at the affected limb. Movement of this reflected normal limb is then performed such that it looks to the patient as though they are performing movement with the affected limb (although it will be pain free due to the fact it is a normal limb being reflected). Following this movement of the normal limb, when the affected limb is moved, levels of pain are reduced. Concepts of neural plasticity within the brain have been hypothesized as to why this effect occurs,
Mirror box therapy appears to be beneficial especially in early CRPS(e.g., McCabe et al., 2003b); however Moseley et al. (2008) concluded that the beneficial effect of mirror therapy for CRPS is still anecdotal, and should be investigated using methodologicallymore rigorous studies. Importantly, the precise neural mechanisms of action are unknown, and need to be studied using a combination of behavioural and neuroimaging approaches.[28]
Graded motor imagery
Because many studies have shown problems with the central nervous system and brain in people with CRPS, recently, treatments have been developed that target these problems. One treatment, (graded motor imagery)[29] has now been tested in three [30][31][32] randomised controlled trials and has shown to be effective at reducing pain and disability in people with chronic CRPS, or phantom limb pain after amputation or avulsion injury of the brachial plexus. The number needed to treat for a 50% reduction in pain was about 3.
Tactile discrimination training
Another approach to CRPS is based on a treatment called sensory discrimination training, which was used for phantom limb pain. A randomised controlled trial [33] demonstrated a significant drop in pain after 10 days training. For CRPS, a replicated case series [34] and a randomised repeated measures experiment [35] both demonstrated an effect of tactile discrimination training on pain, disability and sensory function, in people with CRPS of various durations. This treatment has not been tested in a randomised controlled trial.
Local anaesthetic blocks/injections
Injection of a local anesthetic such as lidocaine is often the first step in treatment. Injections are repeated as needed. The results of local anesthetic injections are short lasting and the procedure is risky. However, early intervention with non-invasive management may be preferred to repeated nerve blockade. The use of topical lidocaine patches has not been shown to be of use in the treatment of CRPS-1 and -2
Spinal cord stimulators
Neurostimulation (spinal cord stimulator) may also be surgically implanted to reduce the pain by directly stimulating the spinal cord. These devices place electrodes either in the epidural space (space above the spinal cord) or directly over nerves located outside the central nervous system. Implantable drug pumps may also be used to deliver pain medication directly to the cerebrospinal fluid which allows powerful opioids to be used in a much smaller dose than when taken orally. A recent review [36] of CRPS treatment concluded: "Some common treatments (e.g., local anesthetic blockade of sympathetic ganglia) are not supported by the aggregate of published studies and should be used less frequently. Other treatments with encouraging published results (e.g., neural stimulators) are not used often enough." A systematic review concluded: Spinal cord stimulation appears to be an effective therapy in the management of patients with CRPS type I (Level A evidence) and type II (Level D evidence).[37] Moreover, there is evidence to demonstrate that SCS is a cost-effective treatment for CRPS type I.
A randomized controlled trial performed by Kemler et al. (2000) on spinal cord stimulation (SCS) in patients with refractory RSD demonstrated that the group receiving SCS + physical therapy (n=36) had a mean reduction of 2.4 cm (using Visual analogue scale) in the intensity of pain at six months compared to a mean increase of 0.2 cm in the group assigned to receive physical therapy alone (n= 18). The intensity of pain was found to be statistically significantly different between the two groups (P < 0.001). In addition, a greater proportion of patients in the SCS + physical therapy reported a 6 (“much improved” outcome) based on a global perceived effect scale compared to physical therapy alone (39% vs. 6%, P = 0.01). However, the study did not find clinically significant improvement in functional status.[38]
Sympathectomy
Surgical, chemical, or radiofrequency sympathectomy — interruption of the affected portion of the sympathetic nervous system — can be used as a last resort in patients with impending tissue loss, edema, recurrent infection, or ischemic necrosis.[39] However, there is little evidence that these permanent interventions alter the pain symptoms of the affected patients and in addition to the normal risks of surgery, such as bleeding and infection, sympathectomy has several specific risks, such as adverse changes in how nerves function.
Ketamine
Ketamine, a dissociative anesthetic, is being used in the treatment of Complex Regional Pain Syndrome with anecdotal success.[citation needed] Low dose ketamine infusion has been used successfully to treat most patients with CRPS, Post Herpatic Neuralgia and other neuropathic pain conditions.[citation needed] It is a safe and effective treatment.[citation needed] During the infusion the patient is monitored constantly, It should be done by a qualified physicians such as an Anesthesiologist only. The theory of ketamine use in CRPS/RSD is primarily advanced by neurologist Dr Robert J. Schwartzman of Drexel University College of Medicine in Philadelphia, and researchers at the University of Tübingen in Germany, but was first introduced in the United States by Doctor Ronald Harbut of Little Rock Arkansas.[citation needed] The hypothesis is that ketamine blocks NMDA receptors which might reboot aberrant brain activity.
There are two treatment modalities; the first consist of a low dose subanesethesia Ketamine infusion of between 10–90 mg per hour over several treatment days, this can be delivered as an outpatient. This is called the awake or subanesethesia technique.
One study[40] demonstrated that 83% of the patients that participated had complete relief and many others had some relief of the symptoms. Another evaluation of a 10-day infusion of intravenous ketamine (awake technique) in the CRPS patient concluded that "A four-hour ketamine infusion escalated from 40–80 mg over a 10-day period can result in a significant reduction of pain with increased mobility and a tendency to decreased autonomic dysregulation".[41] Unfortunately, these study designs are very prone to bias, which means we still need high quality randomised controlled trials of ketamine infusion for CRPS to know about its effects and side effects.
The second treatment modality consists of putting the patient into a medically-induced coma, then administering an extremely high dosage of ketamine; typically between 600 and 900 mg.[42] This version, currently not allowed in the United States, is most commonly done in Germany but some treatments are now also taking place in Monterrey, Nuevo León, Mexico.
Adjunctive treatment
EEG Biofeedback,[43] various forms of psychotherapy,[44] relaxation techniques and hypnosis [45] are adjunctive treatments which assist coping.
Prognosis
Good progress can be made in treating CRPS if treatment is begun early, ideally within 3 months of the first symptoms. If treatment is delayed, however, the disorder can quickly spread to the entire limb and changes in bone, nerve and muscle may become irreversible. The prognosis is not always good. The limb, or limbs, can experience muscle atrophy, loss of use and functionally useless parameters that require amputation. RSD/CRPS will not "burn itself out" but, if treated early, it is likely to go into remission.
Similar disorders
CRPS has characteristics similar to those of other disorders, such as shoulder-hand syndrome, which sometimes occurs after a heart attack and is marked by pain and stiffness in the arm and shoulder; Sudeck syndrome, which is prevalent in older people and women and is characterized by bone changes and muscular atrophy, but is not always associated with trauma; and Steinbrocker syndrome, which includes symptoms such as gradual stiffness, discomfort, and weakness in the shoulder and hand. Erythromelalgia also shares many components of CRPS (burning pain, redness, temperature hypersensitivity, autonomic dysfunction, vasospasm), they both involve small fiber sensory neurosympathetic components. Erythromelalgia involves a lack of sweating, whereas CRPS often involves increased sweating. Subvariations of both exist. New information lends credibility to previous positions that this is an autoimmune response disease that can be caused by injury, non injury, and can progress from the injured location throughout the entire body, to include optic nerves, ear nerves, and other facial nerves. Regarding the facial nerves, the eyes seem to be most vulnerable, with no specific pattern as to one or both. It also has the ability to affect sexual function in both the male and female anatomy, though the ability to engage in sexual activity is limited by the disease itself. There is further information that some cases may have a genetic predisposition for the disease, as with other autoimmune diseases. Myasthenia Gravis is another disease that mirrors many of the symptoms of CRPS.
Current research
The National Institute of Neurological Disorders and Stroke (NINDS), a part of the National Institutes of Health (NIH), supports and conducts research on the brain and central nervous system, including research relevant to RSDS, through grants to major medical institutions across the country. NINDS-supported scientists are working to develop effective treatments for neurological conditions and, ultimately, to find ways of preventing them. Investigators are studying new approaches to treat CRPS and intervene more aggressively after traumatic injury to lower the patient's chances of developing the disorder. In addition, NINDS-supported scientists are studying how signals of the sympathetic nervous system cause pain in CRPS patients. Using a technique called microneurography, these investigators are able to record and measure neural activity in single nerve fibers of affected patients. By testing various hypotheses, these researchers hope to discover the unique mechanism that causes the spontaneous pain of CRPS and that discovery may lead to new ways of blocking pain. Other studies to overcome chronic pain syndromes are discussed in the pamphlet "Chronic Pain: Hope Through Research," published by the NINDS.
Research into treating the condition with Mirror Visual Feedback is being undertaken at the Royal National Hospital for Rheumatic Disease in Bath. Patients are taught how to desensitize in the most effective way then progress on to using mirrors to rewrite the faulty signals in the brain that appear responsible for this condition.
The Netherlands currently has the most comprehensive program of research into CRPS, as part of a multi-million Euro initiative called TREND.[46] German and Australian research teams are also pursuing better understanding and treatments for CRPS.
In animals
CRPS has also been described in animals.[47]
References
- ^ Neuropathic pain. The Merck Manuals: The Merck Manual for Healthcare Professionals.
- ^ Mitchell, S.W. (1872) Injuries of Nerves and their Consequences. Philadelphia: JB Lippincott Co
- ^ Richards RL (1967). "The Term 'Causalgia'". Medical History 11 (1): 86–90. PMC 1033670. PMID 5341038. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1033670.
- ^ Spicher, C.J. (2006) Handbook for Somatosensory Rehabilitation. Montpellier, Paris: Sauramps Medical[1]
- ^ Evans JA (1946). "Reflex sympathetic dystrophy". Surg Clin North America 26 (3): 780–90. PMID 20288177.
- ^ Noordenbos, W. (1959). PAIN Problems pertaining to the transmission of nerve impulses which give rise to pain. Amsterdam : Elsevier
- ^ Stanton-Hicks M, Janig W, Hassenbusch S, Haddox JD, Boas R, Wilson P (1995). "Reflex sympathetic dystrophy: changing concepts and taxonomy". Pain 63 (1): 127–33. doi:10.1016/0304-3959(95)00110-E. PMID 8577483.
- ^ a b Correll GE, Maleki J, Gracely EJ, Muir JJ, Harbut RE (2004). "Subanesthetic ketamine infusion therapy: a retrospective analysis of a novel therapeutic approach to complex regional pain syndrome". Pain Med 5 (3): 263–75. doi:10.1111/j.1526-4637.2004.04043.x. PMID 15367304.
- ^ Kiefer RT, Rohr P, Ploppa A, et al (2008). "A pilot open-label study of the efficacy of subanesthetic isomeric S(+)-ketamine in refractory CRPS patients". Pain Med 9 (1): 44–54. doi:10.1111/j.1526-4637.2006.00223.x. PMID 18254766.
- ^ Pöyhiä R, Vainio A (2006). "Topically administered ketamine reduces capsaicin-evoked mechanical hyperalgesia". Clin J Pain 22 (1): 32–6. doi:10.1097/01.ajp.0000149800.39240.95. PMID 16340591. http://meta.wkhealth.com/pt/pt-core/template-journal/lwwgateway/media/landingpage.htm?an=00002508-200601000-00005.
- ^ Watkins LR, Maier SF (2005). "Immune regulation of central nervous system functions: from sickness responses to pathological pain". J. Intern. Med. 257 (2): 139–55. doi:10.1111/j.1365-2796.2004.01443.x. PMID 15656873.
- ^ Koffler SP, Hampstead BM, Irani F, et al (2007). "The neurocognitive effects of 5 day anesthetic ketamine for the treatment of refractory complex regional pain syndrome". Arch Clin Neuropsychol 22 (6): 719–29. doi:10.1016/j.acn.2007.05.005. PMID 17611073.
- ^ Birklein F (2005). "Complex regional pain syndrome". J. Neurol. 252 (2): 131–8. doi:10.1007/s00415-005-0737-8. PMID 15729516.
- ^ Zollinger PE, Tuinebreijer WE, Breederveld RS, Kreis RW (2007). "Can Vitamin C Prevent Complex Regional Pain Syndrome in Patients with Wrist Fractures? A Randomized, Controlled, Multicenter Dose-Response Study". J Bone Joint Surg Am 89 (7): 1424–1431. doi:10.2106/JBJS.F.01147. PMID 17606778.
- ^ a b c Veldman PH, Reynen HM, Arntz IE, Goris RJ (1993). "Signs and symptoms of reflex sympathetic dystrophy: prospective study of 829 patients". Lancet 342 (8878): 1012–6. doi:10.1016/0140-6736(93)92877-V. PMID 8105263.
- ^ Güler-Uysal F, Başaran S, Geertzen JH, Göncü K (2003). "A 2½-year-old girl with reflex sympathetic dystrophy syndrome (CRPS type I): case report". Clin Rehabil 17 (2): 224–7. doi:10.1191/0269215503cr589oa. PMID 12625665. http://cre.sagepub.com/cgi/pmidlookup?view=long&pmid=12625665.
- ^ "RSDSA :: Reflex Sympathetic Dystrophy Syndrome Association". Rsds.org. 2010-01-21. http://rsds.org/2/fact_fiction/index.html. Retrieved 2010-04-10.
- ^ Yu D (August 2004). "Shoulder pain in hemiplegia". Phys Med Rehabil Clin N Am 15 (3): vi–vii, 683–97. doi:10.1016/S1047-9651(03)00130-X. PMID 15219895.
- ^ Pawelka, S.; Fialka, V.; Ernst, E. (1993). "Reflex sympathetic dystrophy and cigarette smoking". Hand Surgery 18 (1): 168. doi:10.1016/j.0363-5023.1993.90273.6. PMID 8423309.
- ^ "RSDSA: research study". Rsds.org. http://www.rsds.org/study.html. Retrieved 2010-04-10.
- ^ Information sheet for individuals with RSD
- ^ Quisel A, Gill JM, Witherell P (2005). "Complex regional pain syndrome underdiagnosed". J Fam Pract 54 (6): 524–32. PMID 15939004. http://www.jfponline.com/Pages.asp?AID=1947.
- ^ 3.Birklein F, Kunzel W, Sieweke N, Despite clinical similarities there are significant differences between acute limb trauma and complex regional pain syndrome I (CRPS-I). Pain 2001; 93:165-171.
- ^ 5.Wasner G, Schattschneider J, Baron R. Skin temperature side differences: a diagnostic tool for CRPS?
- ^ 4.Gulevich SJ, Conwell TD, Lane J, et al. Stress Infrared Telethermography is useful in the diagnosis of complex regional pain syndrome, type I (formerly reflex sympathetic dystrophy). Clin J Pain 1997; 13:50-59
- ^ Sandroni P, Low PA, Ferrer T, Opfer-Gehrking TL, Willner CL, Wilson PR (1998). "Complex regional pain syndrome I (CRPS I): prospective study and laboratory evaluation". Clin J Pain 14 (4): 282–9. doi:10.1097/00002508-199812000-00003. PMID 9874005.
- ^ Daly, Anne E.; Bialocerkowski, Andrea E. (2009). "Does evidence support physiotherapy management of adult Complex Regional Pain Syndrome Type One? A systematic review". European Journal of Pain 13 (4): 339. doi:10.1016/j.ejpain.2008.05.003. PMID 18619873.
- ^ Swart, Stins, Beek, Cortical changes in complex regional pain synrome (CRPS) European Journal of Pain (2008)[2]
- ^ noi - graded motor imagery
- ^ Lorimer (2009-10-28). "Graded motor imagery is effective for long-standing complex regional pain syndrome". Bodyinmind.com.au. http://www.bodyinmind.com.au/graded-motor-imagery-is-effective-for-long-standing-complex-regional-pain-syndrome/. Retrieved 2010-04-10.
- ^ Lorimer (2009-10-21). "Is successful rehabilitation of complex regional pain syndrome due to sustained attention to the affected limb?". Bodyinmind.com.au. http://www.bodyinmind.com.au/is-successful-rehabilitation-of-complex-regional-pain-syndrome-due-to-sustained-attention-to-the-affected-limb/. Retrieved 2010-04-10.
- ^ admin (2009-10-07). "Graded motor imagery for chronic pain". Bodyinmind.com.au. http://www.bodyinmind.com.au/graded-motor-imagery-for-pathologic-pain/. Retrieved 2010-04-10.
- ^ Flor, H; Denke, C; Schaefer, M; Grüsser, S (2001). "Effect of sensory discrimination training on cortical reorganisation and phantom limb pain.". Lancet 357 (9270): 1763–4. doi:10.1016/S0140-6736(00)04890-X. PMID 11403816.
- ^ Moseley, G; Zalucki, N; Wiech, K (2008). "Tactile discrimination, but not tactile stimulation alone, reduces chronic limb pain". Pain 137 (3): 600. doi:10.1016/j.pain.2007.10.021. PMID 18054437.
- ^ admin (2009-11-25). "Training the Brain 3 – Brain changes, S1 reorganisation". Bodyinmind.com.au. http://www.bodyinmind.com.au/training-the-brain-3-brain-changes-s1-reorganisation/. Retrieved 2010-04-10.
- ^ Hord, E. Daniela; Oaklander, Anne Louise (2003). "Complex regional pain syndrome: A review of evidence-supported treatment options". Current Pain and Headache Reports 7 (3): 188. doi:10.1007/s11916-003-0072-7. PMID 12720598.
- ^ Taylor, R; Buyten, J; Buchser, E (2006). "Spinal cord stimulation for complex regional pain syndrome: A systematic review of the clinical and cost-effectiveness literature and assessment of prognostic factors". European Journal of Pain 10 (2): 91. doi:10.1016/j.ejpain.2005.02.004. PMID 16310712.
- ^ Kemler MA, de Vet HC, Barendse GA, van den Wildenberg FA, van Kleef M (February 2008). "Effect of spinal cord stimulation for chronic complex regional pain syndrome Type I: five-year final follow-up of patients in a randomized controlled trial". J. Neurosurg. 108 (2): 292–8. doi:10.3171/JNS/2008/108/2/0292. PMID 18240925.
- ^ Stanton-Hicks M, Baron R, Boas R, Gordh T, Harden N, Hendler N, Koltzenburg M, Raj P, Wilder R (1998). "Complex Regional Pain Syndromes: guidelines for therapy". Clin J Pain 14 (2): 155–66. doi:10.1097/00002508-199806000-00012. PMID 9647459.
- ^ Correll, Graeme E.; Maleki, Jahangir; Gracely, Edward J.; Muir, Jesse J.; Harbut, Ronald E. (2004). "Subanesthetic Ketamine Infusion Therapy: A Retrospective Analysis of a Novel Therapeutic Approach to Complex Regional Pain Syndrome". Pain Medicine 5 (3): 263. doi:10.1111/j.1526-4637.2004.04043.x. PMID 15367304.
- ^ Goldberg ME, Domsky R, Scaringe D, et al (2005). "Multi-day low dose ketamine infusion for the treatment of complex regional pain syndrome". Pain Physician 8 (2): 175–9. PMID 16850072. http://www.painphysicianjournal.com/linkout_vw.php?issn=1533-3159&vol=8&page=175.
- ^ CNN report on Ketamine therapy for CRPS/RSD, September 1, 2006
- ^ Grunert BK, Devine CA, Sanger JR, Matloub HS, Green D (1990). "Thermal self-regulation for pain control in reflex sympathetic dystrophy syndrome". J Hand Surg [Am] 15 (4): 615–8. doi:10.1016/S0363-5023(09)90024-7. PMID 2199569. http://www.rsds.org/5/news/2005/biofeedback.htm.
- ^ The Psychologist's Role in the Chronic Pain of Reflex Sympathetic Dystrophy. Rosemarie Scolaro Moser, Ph.D. Printed in New Jersey Psychologist, Spring 1999, pp. 24–25.
- ^ Gainer MJ (1992). "Hypnotherapy for reflex sympathetic dystrophy". Am J Clin Hypn 34 (4): 227–32. PMID 1349789.
- ^ TREND homepage.
- ^ Bergadano A, Moens Y, Schatzmann U (2006). "Continuous extradural analgesia in a cow with complex regional pain syndrome". Vet Anaesth Analg 33 (3): 189–92. doi:10.1111/j.1467-2995.2005.00245.x. PMID 16634945.
External links
- Complex regional pain syndrome at the Open Directory Project
- Reflex sympathetic dystrophy at the Open Directory Project
- CRPS explained for children and teenagers
- Reflex Sympathetic Dystrophy
Nervous system pathology, PNS, somatic (G50–G64, 350–357) Nerve, nerve root, plexus V (Trigeminal neuralgia, Anesthesia dolorosa) · VII (Facial nerve paralysis, Bell's palsy, Melkersson–Rosenthal syndrome, Parry–Romberg syndrome, Central seven) · XI (Accessory nerve disorder)Lower limbGeneralPolyneuropathies/Polyradiculoneuropathy OtherOsteochondropathy (M80–M94, 730–733) Osteopathies endocrine bone disease: Osteitis fibrosa cystica (Brown tumor)infectious bone disease: Osteomyelitis (Sequestrum, Involucrum) · Sesamoiditis · Brodie abscess · PeriostitisBone density
and structureDensity / metabolic bone diseaseContinuity of boneOtherFibrous dysplasia (Monostotic, Polyostotic) · Skeletal fluorosis · bone cyst (Aneurysmal bone cyst) · Hyperostosis (Infantile cortical hyperostosis) · Osteosclerosis (Melorheostosis)OtherPaget's disease of bone · Algoneurodystrophy · Hypertrophic pulmonary osteoarthropathy · Nonossifying fibromaChondropathies OtherBoth lower limb: hip (Legg–Calvé–Perthes syndrome) · tibia (Osgood-Schlatter disease, Blount's disease) · foot (Köhler disease, Sever's disease)Categories:- Ailments of unknown etiology
- Neurological disorders
- Neurocutaneous conditions
- Pain
- Syndromes
- Osteopathies
Wikimedia Foundation. 2010.