- Osteonecrosis of the jaw
-
Osteonecrosis of the jaws Classification and external resources
Osteonecrosis of the jaw of the upper left jaw in a patient diagnosed with chronic venous insufficiencyICD-10 M87.1 ICD-9 733.45 DiseasesDB 1174 eMedicine derm/816 Osteonecrosis of the jaw (ONJ) is a severe bone disease that affects the maxilla and the mandible. Various forms of ONJ have been described over the last 160 years, and a number of causes have been suggested in the literature. In recent years, an increased incidence of ONJ has been associated with the use of high dosages of bisphosphonates, required by some cancer treatment regimens, especially when the patient undergoes subsequent dental procedures. The possible risk from lower oral doses of bisphosphonates, taken by patients to prevent or treat osteoporosis, remains uncertain.[1]
Various treatment options have been explored, however severe cases of ONJ still require surgical removal of the affected bone.[2] A thorough history and assessement of pre-existing systemic problems and possible sites of dental infection are required to help prevent the condition, especially if bisphosphonate therapy is considered.[1]
Contents
Classification
Grade Size (diameter*) 1A Single lesion, <0.5 cm 1B Multiple lesions, largest <0.5 cm 2A Single lesion <1.0 cm 2B Multiple lesions, largest <1.0 cm 3A Single lesion, ≤2.0 cm 3B Multiple lesions, largest ≤2.0 cm 4A Single lesion >2.0 cm 4B Multiple lesions, largest >2.0 cm *Lesion size measured as the largest diameter Grade Severity 1 Asymptomatic 2 Mild 3 Moderate 4 Severe Osteonecrosis of the jaw is classified based on severity, number of lesions, and lesion size. Osteonecrosis of greater severity is given a higher grade, with asymptomatic ONJ designated as grade 1, and severe ONJ designated as grade 4.
Signs and symptoms
The definitive symptom of ONJ is the exposure of mandibular or maxillary bone through lesions in the gingiva that do not heal.[3] Pain, inflammation of the surrounding soft tissue, secondary infection or drainage may or may not be present. The development of lesions is most frequent after invasive dental procedures, such as extractions, but is also known to occur spontaneously. There may be no symptoms for weeks or months, until lesions with exposed bone appear.[4] Lesions are more common on the mandible than the maxilla.
Causes
Toxic agents
Other factors such as toxicants can adversely impact bone cells. Infections, chronic or acute, can affect blood flow by inducing platelet activation and aggregation, contributing to a localized state of excess coagulability (hypercoagulability) that may contribute to clot formation (thrombosis), a known cause of bone infarct and ischaemia. Exogenous estrogens, also called hormonal disruptors, have also been linked with an increased tendency to clot (thrombophilia) and impaired bone healing.[5]
Heavy metals such as lead and cadmium have been implicated in osteoporosis. Cadmium and lead also promotes the synthesis of plasminogen activator inhibitor-1 (PAI-1) which is the major inhibitor of fibrinolysis ( the mechanism by which the body breaks down clots ) and shown to be a cause of hypofibrinolysis.[6] Persistent blot clots can lead to congestive blood flow (hyperemia) in bone marrow, impaired blood flow and ischaemia in bone tissue resulting in lack of oxygen (hypoxia), bone cell damage and eventual cell death (apoptosis). Of significance is the fact that the average concentration of cadmium in human bones in the 20th century has increased to about 10 times above the pre-industrial level.[7]
Ethanol both from exogenous and endogenous sources and, its more toxic metabolite, acetaldehyde, have also been implicated in both osteoporosis and osteonecrosis. Acetaldehyde, a highly toxic metabolite of ethanol, can play a role in hypoxia and inhibit the osteoblastogenic potential of the bone marrow.[8] Ethanol has been shown to alter the epithelial barrier through ethanol oxidation into acetaldehyde by the colonic microflora and downstream mast cell activation. Such alterations that remain for longer periods could result in excessive endotoxin passage into the vascular network.[9] Intracolonic acetaldehyde may also be an important determinant of the blood acetaldehyde level and a possible hepatotoxin.[10] High serum antibody titers against acetaldehyde-protein adducts have been found not only in alcoholics but also in patients with nonalcoholic liver disease, suggesting a contribution of acetaldehyde derived from sources other than exogenous ethanol.[11] In a study on rats the role of intestinal bacterial overgrowth on the production and metabolism of ethanol, rats with a jejunal self-filling diverticulum (blind-loop) were compared to controls with a self-emptying diverticulum. Both endogenous ethanol and acetaldehyde were found in the blind-loop contents. Intragastric administration of sucrose produced a marked increase in acetaldehyde and acetate in the portal venous blood, with only a modest elevation of ethanol. It was concluded that the resulting high concentrations of acetaldehyde, both in the intestinal lumen and the portal blood, may have deleterious effects on the gastrointestinal(GI) mucosa and the liver.[12] Another experimental in-vitro study showed the potential of certain bacteria representing normal human colonic flora to produce acetaldehyde under various atmospheric conditions that may prevail in different parts of the GI tract. This bacterial adaptation may be an essential feature of the bacteriocolonic pathway to produce toxic and carcinogenic acetaldehyde from either endogenous or exogenous ethanol.[13] Many species of gut bacteria, yeast and fungal organisms such as Candida albicans found in the human GI tract and involved in gut dysbiosis, an imbalance in the microbial flora, have been shown to significantly increase blood ethanol levels, post-mortem, in individuals who had not consumed any alcohol before death.[14][15]
The effects of chronic gut dysbiosis and long term exposure to low levels of endogenous acetaldehyde on bone tissue and hepatic function is not yet fully understood. However Cordts et al. suggested in 2001 that gut dysbiosis (as indicated by stool yeast) and hepatic detoxification challenge pathway exhaustion may lead to subclinical, systemic inflammation and chronic venous insufficiency (CVI). CVI is a pathological condition caused either by the congenital absence of or damage to venous valves in the superficial and communicating systems. Venous incompetence due to thrombi and formation of thrombi favoured by the Virchow triad (venous stasis, hypercoagulability, endothelial trauma) also can cause CVI.[16]
Bisphosphonates
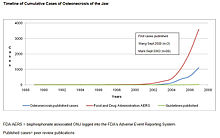
Main article: Bisphosphonate-associated osteonecrosis of the jawThe first three cases of bisphosphonate-associated osteonecrosis of the jaw were spontaneously reported to the FDA by an oral surgeon in 2002, with the toxicity being described as a potentially late toxicity of chemotherapy.[14] In 2003 and 2004, three oral surgeons independently reported to the FDA information on 104 cancer patients with bisphosphonate-associated osteonecrosis of the jaw seen in their referral practices in California, Florida, and New York.[15][16][17] These case series were published as peer-reviewed articles- two in the Journal of Oral and Maxillofacial Surgery and one in the Journal of Clinical Oncology. Subsequently, numerous instances of persons with this ADR were reported to the manufacturers and to the FDA. By December 2006, 3607 cases of persons with this ADR had been reported to the FDA and 2227 cases had been reported to the manufacturer of intravenous bisphosphonates.
The International Myeloma Foundation's web-based survey included 1203 respondents, 904 patients with myeloma and 299 with breast cancer and an estimate that after 36 months, osteonecrosis of the jaw had been diagnosed in 10% of 211 patients on zoledronate and 4% of 413 on pamidronate.[18] A population based study in Germany identified more than 300 cases of osteonecrosis of the jaw, 97% occurring in cancer patients (on high-dose intravenous bisphosphonates), and 3 cases in 780,000 patients with osteoporosis for an incidence of 0.00038%. Time to event ranged from 23–39 months and 42–46 months with high dose intravenous and oral bisphosphonates.[19] A prospective, population based study by Mavrokokki et al. estimated an incidence of osteonecrosis of the jaw of 1.15% for intravenous bisphosphonates and 0.04% for oral bisphosphonates. Most cases (73%) were precipitated by dental extractions. In contrast, safety studies sponsored by the manufacturer reported bisphosphonate-associated osteonecrosis of the jaw rates that were much lower. Although the majority of cases of ONJ have occurred in cancer patients receiving high dose intravenous bisphosphonates, almost 800 cases have been reported in oral bisphosphonate users for osteoporosis or Pagets disease. In terms of severity most cases of ONJ in oral bisphosphonate users are stage 1-2 and tend to progress to resolution with conservative measures such as oral chlorhexidine rinses.
In the past few years, thousands of cases of ONJ in patients on bisphosphonate therapy have been diagnosed usually following lack of healing after a dental extraction but also in cases of spontaneous exposure of the cortical bone tissue through the gingiva and mucosa.[20][21]
The recent increase of such cases has been linked with a major emphasis on the therapeutic use of bisphosphonates for osteoporosis, especially since hormone replacement therapy has been shown to increase the risk of breast cancer, clots and cardiovascular disease in women following the 2003 findings of the U.S. Women’s Health Initiative study.[22] Two classes of bisphosphonates are presently prescribed:
- Non-nitrogen containing bisphosphonates such as etidronate (Didronel, Procter & Gamble Pharmaceuticals)
- Nitrogen containing such as alendronate (Fosamax, Merck), pamidronate (Aredia, Novartis), zoledronate (Zometa, Novartis), risedronate (Actonel, Procter & Gamble) and ibandronate (Boniva, Roche Laboratories).
The nitrogen containing bisphosphonates are the most potent inhibitors and no case of ONJ associated with etidronate has been reported yet. The main pharmacological action of bisphosphonates is inhibition of the osteoclast driven bone resorption. This is achieved by shortening osteoclast lifespan via apoptosis and by inhibiting osteoclast activity and recruitment on the bone surface (61). When a bisphosphonate binds to bone mineral, osteoclasts resorb both bone and the bound bisphosphonate. During bone formation, if any, bisphosphonate remaining on the surface of the bone is covered and remains there until future osteoclastic bone resorption at the site. This explains why inhibition of bone resorption continues long after bisphosphonate treatment has been discontinued.[23]
This form of therapy has been shown to prevent loss of bone mineral density (BMD) as a result of a reduction in bone turnover. However bone health entails quite a bit more than just BMD. There are many other factors to consider.
In healthy bone tissue there is a homeostasis between bone resorption and bone apposition. Diseased or damaged bone is resorbed through the osteoclasts mediated process while osteoblasts form new bone to replace it, thus maintaining healthy bone density. This process is commonly called remodelling.
However osteoporosis is essentially the result of a lack of new bone formation in combination with bone resorption in reactive hyperemia, related to various etiological and contributing factors, and bisphosphonates do not address these factors at all.
An individual who is already having problems with osteoporosis/ osteonecrosis of the jaws due to the effects of these etiological factors will be more susceptible to the adverse effects of bisphosphonates. In theory, by suppressing osteoclastic activity and bone resorption, any ischaemic-damaged bone will be left in situ instead of being resorbed. The damaged bone will not be repaired either if the factors already inhibiting osteoblastic activity are still present. Therefore the amount of osteonecrotic tissue should be expected to increase until it reaches a level when any trauma or insult to this necrotic bone will result in extremely poor healing, exposed necrotic bone to the oral environment, development of pain, and increased risks of microbial infection, as effectively seen in bisphosphonate-associated cases of ONJ. Recently, a proposal incorporating both the reduced bone turnover and the infectious elements of previous theories has been put forward, which cites the impaired functionality of affected macrophages as the dominant factor in the development of ONJ.[24]
In a systematic review of cases of bisphosphonate-associated ONJ up to 2006, it was concluded that the mandible is more commonly affected than the maxilla (2:1 ratio), and 60% of cases are preceded by a dental surgical procedure. According to Woo, Hellstein and Kalmar, oversuppression of bone turnover is probably the primary mechanism for the development of this form of ONJ, although there may be contributing co-morbid factors (as discussed elsewhere in this article). It is recommended that all sites of potential jaw infection should be eliminated before bisphosphonate therapy is initiated in these patients to reduce the necessity of subsequent dentoalveolar surgery. The degree of risk for osteonecrosis in patients taking oral bisphosphonates, such as alendronate (Fosamax), for osteoporosis is uncertain and warrants careful monitoring.[25] Patients taking dexamethasone and other glucocorticoids are at increased risk.[26]
Matrix metalloproteinase 2 may be a candidate gene for Bisphosphonate-associated osteonecrosis of the jaw, since it is the only gene known to be associated with bone abnormalities and atrial fibrillation, both of which are side effects of bisphosphonates.[27]
Pathophysiology
Histopathological alterations
Persons with ONJ may have either necrotic bone or bone marrow that has been slowly strangulated or nutrient-starved. Bone with chronically poor blood flow develops either a fibrous marrow since fibres can more easily live in nutrient starved areas, a greasy, dead fatty marrow (wet rot), a very dry, sometimes leathery marrow (dry rot), or a completely hollow marrow space (osteocavitation), also typical of ONJ. The blood flow impairment occurs following a bone infarct, a blood clot forming inside the smaller blood vessels of cancellous bone tissue.
Under ischaemic conditions numerous pathological changes in the bone marrow and trabeculae of oral cancellous bone have been documented. Microscopically, areas of "apparent fatty degeneration and/or necrosis, often with pooled fat from destroyed adipose cells (oil cysts) and with marrow fibrosis (reticular fatty degeneration)" are seen. These changes are present even if "most bony trabeculae appear at first glance viable, mature and otherwise normal, but closer inspection demonstrates focal loss of osteocytes and variable micro cracking (splitting along natural cleavage planes). The microscopic features are similar to those of ischaemic or aseptic osteonecrosis of long bones, corticosteroid-induced osteonecrosis, and the osteomyelitis of caisson (deep-sea diver’s) disease".[28]
In the cancellous portion of femoral head it is not uncommon to find trabeculae with apparently intact osteocytes which seem to be "alive" but are no longer synthetizing collagen. This appears to be consistent with the findings in alveolar cancellous bone.[29]
Osteonecrosis can affect any bone, but the hips, knees and jaws are most often involved. Pain can often be severe, especially if teeth and/or a branch of the trigeminal nerve is involved, but many patients do not experience pain, at least in the earlier stages. When severe facial pain is involved, the term NICO, for Neuralgia-inducing cavitational osteonecrosis, is frequently used.
ONJ, even in its mild or minor forms, creates a marrow environment that is conducive to bacterial growth. Since many individuals have low-grade infections of the teeth and gums, this probably is one of the major mechanisms by which the marrow blood flow problem can worsen; any local infection / inflammation will cause increased pressures and clotting in the area involved. No other bones have this mechanism as a major risk factor for osteonecrosis. A wide variety of bacteria have been cultured from ONJ lesions. Typically, they are the same microorganisms as those found in periodontitis or devitalized teeth. However, according to special staining of biopsied tissues, bacterial elements are rarely found in large numbers. So while ONJ is not primarily an infection, many cases have a secondary, very low-level of bacterial infection and chronic non-suppurative osteomyelitis can be associated with ONJ. Fungal infections in the involved bone do not seem to be a problem, but viral infections have not been studied. Some viruses, such as the smallpox virus (no longer existent in the wild) can produce osteonecrosis.
The effects of persistent ischaemia on bone cells
Cortical bone is well vascularized by the surrounding soft tissues thus less susceptible to ischaemic damage. Cancellous bone, with its mesh like structure and spaces filled with marrow tissue is more susceptible to damage by bone infarcts, leading to Hypoxia (medical) and premature cell apoptosis.[17][18][30][31] The mean life-span of osteocytes has been estimated to be 15 years in cancellous bone,[32] and 25 years in cortical bone.[33] while the average lifespan of human osteoclasts is about 2 to 6 weeks and the average lifespan of osteoblasts is approximately 3 months.[34] In healthy bone these cells are constantly replaced by differentiation of bone marrow mesenchymal stem cells (MSC).[35] However in both non-traumatic osteonecrosis and alcohol-induced osteonecrosis of the femoral head, a decrease in the differentiation ability of mesenchymal stem into bone cells has been demonstrated,[36][37] and altered osteoblastic function plays a role in ON of the femoral head.[38] If these results are extrapolated to ONJ the altered differentiation potential of bone marrow mesenchymal stem cells (MSC) combined with the altered osteoblastic activity and premature death of existing bone cells would explain the failed attempts at repair seen in ischaemic-damaged cancellous bone tissue in ONJ.
The rapidity with which premature cell death can occur depends on the cell type and the degree and duration of the anoxia. haematopoietic cells , in bone marrow, are sensitive to anoxia and are the first to die after reduction or removal of the blood supply. In anoxic conditions they usually die within 12 hours. Experimental evidence suggests that bone cells composed of osteocytes, osteoclasts, and osteoblasts die within 12–48 hours, and marrow fat cells die within 120 hours.[19] The death of bone does not alter its radiographic opacity nor its mineral density. Necrotic bone does not undergo resorption; therefore, it appears relatively more opaque.
Attempts at repair of ischaemic-damaged bone will usual occur in 2 phases. First, when dead bone abuts live marrow, capillaries and undifferentiated mesenchymal cells grow into the dead marrow spaces, while macrophages degrade dead cellular and fat debris. Second, mesenchymal cells differentiate into osteoblasts or fibroblasts. Under favorable conditions, layers of new bone form on the surface of dead spongy trabeculae. If sufficiently thickened, these layers may decrease the radiodensity of the bone; therefore, the first radiographic evidence of previous osteonecrosis may be patchy sclerosis resulting from repair. Under unfavorable conditions repeated attempts at repair in ischaemic conditions can be seen histologically and are characterized by extensive delamination or microcracking along cement lines as well as the formation of excessive cement lines.[39] Ultimate failure of repair mechanisms due to persistent and repeated ischaemic events is manifested as trabecular fractures that occur in the dead bone under functional load. Later followed by cracks and fissures leading to structural collapse of the area involved (osteocavitation).[19]
Treatment
The treatment should be tailored to the individual patient according to the etiological factors involved and the severity of the disease process. With oral osteoporosis the emphasis should be on good nutrient absorption and metabolic wastes elimination through a healthy gastro-intestinal function, effective hepatic metabolism of toxicants such as exogenous estrogens, endogenous acetaldehyde and heavy metals, a balanced diet, healthy lifestyle, assessment of factors related to potential coagulopathies, and treatment of periodontal diseases and other oral and dental infections.
In cases of advanced oral ischaemic osteoporosis and/or ONJ that are not bisphosphonates related, clinical evidence has shown that surgically removing the damaged marrow, usually by curettage and decortication, will eliminate the problem (and the pain) in 74% of patients with jaw involvement.[2] Repeat surgeries, usually smaller procedures than the first, may be required, and almost a third of jawbone patients will need surgery in one or more other parts of the jaws because the disease so frequently present multiple lesions, i.e. multiple sites in the same or similar bones, with normal marrow in between. In the hip, at least half of all patients will get the disease in the opposite hip over time; this pattern occurs in the jaws as well. Recently, it has been found that some osteonecrosis patients respond to anticoagulation therapies alone. The earlier the diagnosis the better the prognosis. Research is ongoing on other non-surgical therapeutic modalities that could alone or in combination with surgery further improve the prognosis and reduce the morbidity of ONJ. A greater emphasis on minimizing or correcting known etiological factors is necessary while further research is conducted on chronic ischaemic bone diseases such as oral osteoporosis and ONJ.
In patients with bisphosphonates-associated ONJ, the response to surgical treatment is usually poor.[40] Conservative debridement of necrotic bone, pain control, infection management, use of antimicrobial oral rinses, and withdrawal of bisphosphonates are preferable to aggressive surgical measures for treating this form of ONJ.[41] Although an effective treatment for bisphosphonate-associated bone lesions has not yet been established,[42] and this is unlikely to occur until this form of ONJ is better understood, there as been clinical reports of some improvement after 6 months or more of complete cessation of bisphosphonate therapy.[43]
History
ONJ is not a new disease, around 1850 various forms of "chemical osteomyelitis" resulting from environmental pollutants, such as lead and the white phosphorus used in early (non-safety) matches (Phossy jaw), as well as from popular medications containing mercury, arsenic or bismuth, were reported in the literature.[44][45][46][47][48][49][50] This disease apparently did not often occur in individuals with good gingival health, and usually targeted the mandible first.[45] It was associated with localized or generalized deep ache or pain, often of multiple jawbone sites. The teeth often appeared sound and suppuration was not present. Even so, the dentist often began extracting one tooth after another in the region of pain, often with temporary relief but usually to no real effect.[46]
Today a growing body of scientific evidence indicate that this disease process, in the cancellous bone and bone marrow, is caused by bone infarcts mediated by a range of local and systemic factors. Bone infarcts as well as damage to the deeper portion of the cancellous bone is an insidious process. It is certainly not visible clinically and routine imaging techniques such as radiographs are not effective for that sort of damage. "An important and often incompletely understood principle of radiography is the amount of bone destruction that goes undetected by routine x-rays procedures; this has been demonstrated by numerous investigators. Destruction confined to the cancellous portion of the bone cannot be detected radiographically, ad radiolucencies appear only when there is internal or external erosion or destruction of the bone cortex."[51] In fact no radiographic findings are specific for bone infarction / osteonecrosis. A variety of pathologies may mimic bone infarction, including stress fractures, infections, inflammations, and metabolic and neoplastic processes. The limitations apply to all imaging modalities, including plain radiography, radionuclide studies, CT scans, and magnetic resonance imaging (MRI). Through-transmission alveolar ultrasound, based on quantitative ultrasound (QUS) in combination with panoramic dental radiography (orthopantomography) is helpful in assessing changes in jawbone density.[52][53] When practitioners have an up to date understanding of the disease process and a good anamnesis is combined with detailed clinical findings and course of events, the diagnosis, with the help of various imaging modality, can be achieved earlier, in most patients.
In the modern dental profession, it is only recently when severe cases associated with bisphosphonates came to light, that the issue of ONJ has been brought to the attention of a majority of dentists. At present, the focus is mostly on bisphosphonates associated cases, and is sometimes referred to colloquially as "phossy jaw", a similar, earlier occupational disease.[54][55] However, the pharmaceutical manufacturers of bisphosphonates drugs such as Merck and Novartis have stated that ONJ in patients on this class of drug, can be related to a pre-existing condition, coagulopathy, anemia, infection, use of corticosteroids, alcoholism and other conditions already known to be associated with ONJ in absence of bisphosphonate therapy. The implication is that bisphosphonates may not be the initiating cause of ONJ and that other pre-existing or concurrent systemic and/or local dental factors are involved.[56][57]
Since ONJ has been diagnosed in many patients who did not take bisphosphonates, it is thus logical to assume that bisphosphonates are not the only factor in ONJ. While the oversuppression of bone turnover seems to play a major role in aggravating the disease process, other factors can and do initiate the pathophysiological mechanisms responsible for ONJ. In non-bisphosphonate cases of ONJ, it is mainly the cancellous portion of the bone and it’s marrow content that are involved in the disease process. The first stage is an oedema of the bone marrow initiated by a bone infarct, which is itself modulated by numerous etiological factors, leading to myelofibrosis as a result of hypoxia and gradual loss of mineral bone density characteristic of ischaemic osteoporosis. Further deterioration can be triggered by additional bone infarcts leading to anoxia and a localized areas of osteonecrosis within the osteoporotic cancellous bone. Secondary events such as dental infection, injection of local anaesthetics with vasoconstrictors, such as epinephrine, and trauma can add further complications to the disease process and chronic non-pus forming bone infection osteomyelitis can also be associated with ONJ.[58][59][60]
However, in patients on bisphosphonates, the cortical bone is also frequently involved as well. Spontaneous exposure of necrotic bone tissue through the oral soft tissues or following non-healing bone exposure after routine dental surgery, characteristics of this form of ONJ, may be the result of late diagnosis of a disease process that has been masked by the oversuppression of osteoclastic activity, allowing pre-existing etiological factors to further aggravate bone damage.
References
- ^ a b Woo S, Hellstein J, Kalmar J (2006). "Narrative [corrected] review: bisphosphonates and osteonecrosis of the jaws.". Ann Intern Med 144 (10): 753–61. PMID 16702591.
- ^ a b Bouquot JE, Christian J (1995). "Long-term effects of jawbone curettage on the pain of facial neuralgia". J. Oral Maxillofac. Surg. 53 (4): 387–97; discussion 397–9. doi:10.1016/0278-2391(95)90708-4. PMID 7699492.
- ^ JOURNAL OF BONE AND MINERAL RESEARCH, 2007, Bisphosphonate-Associated Osteonecrosis of the Jaw: Report of a Task Force of the American Society for Bone and Mineral Research
- ^ American Dental Association, Osteonecrosis of the Jaw
- ^ Glueck C, McMahon R, Bouquot J, Triplett D (1998). "Exogenous estrogen may exacerbate thrombophilia, impair bone healing and contribute to development of chronic facial pain.". Cranio 16 (3): 143–53. PMID 9852807.
- ^ Yamamoto C (May 2000). "[Toxicity of cadmium and lead on vascular cells that regulate fibrinolysis]". Yakugaku Zasshi 120 (5): 463–73. PMID 10825810.
- ^ Jaworowski Z, Barbalat F, Blain C, Peyre E (1985). "Heavy metals in human and animal bones from ancient and contemporary France". Sci. Total Environ. 43 (1–2): 103–26. doi:10.1016/0048-9697(85)90034-8. PMID 4012292.
- ^ Giuliani N, Girasole G, Vescovi P, Passeri G, Pedrazzoni M (February 1999). "Ethanol and acetaldehyde inhibit the formation of early osteoblast progenitors in murine and human bone marrow cultures". Alcohol Clin Exp Res 23 (2): 381–5. doi:10.1111/j.1530-0277.1999.tb04126.x. PMID 10069572.
- ^ Ferrier L, Bérard F, Debrauwer L, et al. (2006). "Impairment of the Intestinal Barrier by Ethanol Involves Enteric Microflora and Mast Cell Activation in Rodents". Am. J. Pathol. 168 (4): 1148–54. doi:10.2353/ajpath.2006.050617. PMC 1606551. PMID 16565490. http://ajp.amjpathol.org/cgi/content/full/168/4/1148.
- ^ Salaspuro M (1996). "Bacteriocolonic pathway for ethanol oxidation: characteristics and implications". Ann. Med. 28 (3): 195–200. doi:10.3109/07853899609033120. PMID 8811162.
- ^ Ma XL, Baraona E, Hernández-Muñoz R, Lieber CS (1989). "High levels of acetaldehyde in nonalcoholic liver injury after threonine or ethanol administration". Hepatology 10 (6): 933–40. doi:10.1002/hep.1840100607. PMID 2511135.
- ^ Baraona E, Julkunen R, Tannenbaum L, Lieber CS (1986). "Role of intestinal bacterial overgrowth in ethanol production and metabolism in rats". Gastroenterology 90 (1): 103–10. PMID 3940238.
- ^ Salaspuro V, Nyfors S, Heine R, Siitonen A, Salaspuro M, Jousimies-Somer H (1999). "Ethanol oxidation and acetaldehyde production in vitro by human intestinal strains of Escherichia coli under aerobic, microaerobic, and anaerobic conditions". Scand. J. Gastroenterol. 34 (10): 967–73. doi:10.1080/003655299750025057. PMID 10563665.
- ^ a b Lewis RJ, Johnson RD, Angier MK, Vu NT (2004). "Ethanol formation in unadulterated postmortem tissues". Forensic Sci. Int. 146 (1): 17–24. doi:10.1016/j.forsciint.2004.03.015. PMID 15485717.
- ^ a b Yajima D, Motani H, Kamei K, Sato Y, Hayakawa M, Iwase H (2006). "Ethanol production by Candida albicans in postmortem human blood samples: effects of blood glucose level and dilution". Forensic Sci. Int. 164 (2–3): 116–21. doi:10.1016/j.forsciint.2005.12.009. PMID 16427751.
- ^ a b Cordts PR, Kaminski MV, Raju S, Clark MR, Woo KM (2001). "Could gut-liver function derangements cause chronic venous insufficiency?". Vasc Surg 35 (2): 107–14. PMID 11668378.
- ^ a b Marcus R, Feldman D, Kelsey J. (1996). Osteoporosis. San Diego: Academic Press. ISBN 0124708633.
- ^ a b Bullough PG (1997). Orthopaedic pathology (3rd ed.). Baltimore: Wolfe-Mosby.
- ^ a b c Bone infarct at eMedicine - accessed 2006-05-21
- ^ Fosamax does more harm then good. Accessed 21 May 2006.
- ^ Hay KD, Bishop PA (2006). "Association of osteonecrosis of the jaws and bisphosphonate pharmacotherapy: dental implications". N Z Dent J 102 (1): 4–9. PMID 16568881.
- ^ Wassertheil-Smoller S, Hendrix SL, Limacher M, et al. (2003). "Effect of estrogen plus progestin on stroke in postmenopausal women: the Women's Health Initiative: a randomized trial". JAMA 289 (20): 2673–84. doi:10.1001/jama.289.20.2673. PMID 12771114.
- ^ Rodan GA, Reszka AA (2003). "Osteoporosis and bisphosphonates". J Bone Joint Surg Am 85-A Suppl 3: 8–12. PMID 12925603.
- ^ Pazianas M. Osteonecrosis of the jaw and the role of macrophages. J Natl Cancer Inst. 2011 Feb 2;103(3):232-40. Epub 2010 Dec 28.
- ^ Woo SB, Hellstein JW, Kalmar JR (2006). "Narrative [corrected] review: bisphosphonates and osteonecrosis of the jaws". Ann. Intern. Med. 144 (10): 753–61. PMID 16702591.
- ^ Marx RE, Sawatari Y, Fortin M, Broumand V (November 2005). "Bisphosphonate-induced exposed bone (osteonecrosis/osteopetrosis) of the jaws: risk factors, recognition, prevention, and treatment". J. Oral Maxillofac. Surg. 63 (11): 1567–75. doi:10.1016/j.joms.2005.07.010. PMID 16243172. http://linkinghub.elsevier.com/retrieve/pii/S0278-2391(05)01187-0.
- ^ Lehrer S, Montazem A, Ramanathan L, et al. (January 2009). "Bisphosphonate-induced osteonecrosis of the jaws, bone markers, and a hypothesized candidate gene". J. Oral Maxillofac. Surg. 67 (1): 159–61. doi:10.1016/j.joms.2008.09.015. PMID 19070762. http://linkinghub.elsevier.com/retrieve/pii/S0278-2391(08)01493-6.
- ^ Neville BW, Damn D, Allen C, Bouquot JE. (1995). "Facial pain and neuromuscular diseases.". Oral and maxillofacial pathology. W.B Saunders Co. pp. 631–632.
- ^ Arlet J, Durroux R, Fauchier C, Thiechart M.. "Topographic and evolutive aspects". In Arlet J, Ficat PR, Hungerford DS. (eds). Histophatology of the nontraumatic necrosis of the femoral head.
- ^ Kanus JA. (1996). Textbook of osteoporosis.. Osford: Blackwell Science Ltd..
- ^ Vigorita VJ. (1999). Orthopaedic pathology. Philadelphia: Lippincott Williams & Wilkins.
- ^ Parfitt AM (1994). "Osteonal and hemi-osteonal remodeling: the spatial and temporal framework for signal traffic in adult human bone". J. Cell. Biochem. 55 (3): 273–86. doi:10.1002/jcb.240550303. PMID 7962158.
- ^ Parfitt, A M.; Kleerekoper, M.; Villanueva, A R. (1987). "Increased bone age: mechanisms and consequences". Osteoporosis. Copenhagen: Osteopress. pp. 301–308.
- ^ Frost, H M. Charles C. Thomas; Springfield, IL (1963). Bone Remodeling Dynamics.
- ^ Owen M, Friedenstein AJ (1988). "Stromal stem cells: marrow- derived osteogenic precursors". Ciba Found Symp 136: 42–60. PMID 3068016.
- ^ Lee JS, Lee JS, Roh HL, Kim CH, Jung JS, Suh KT (2006). "Alterations in the differentiation ability of mesenchymal stem cells in patients with nontraumatic osteonecrosis of the femoral head: comparative analysis according to the risk factor". J. Orthop. Res. 24 (4): 604–9. doi:10.1002/jor.20078. PMID 16514658.
- ^ Suh KT, Kim SW, Roh HL, Youn MS, Jung JS (2005). "Decreased osteogenic differentiation of mesenchymal stem cells in alcohol-induced osteonecrosis". Clin. Orthop. Relat. Res. &NA; (431): 220–5. doi:10.1097/01.blo.0000150568.16133.3c. PMID 15685079.
- ^ Gangji V, Hauzeur JP, Schoutens A, Hinsenkamp M, Appelboom T, Egrise D (2003). "Abnormalities in the replicative capacity of osteoblastic cells in the proximal femur of patients with osteonecrosis of the femoral head". J. Rheumatol. 30 (2): 348–51. PMID 12563694.
- ^ Adams WR, Spolnik KJ, Bouquot JE (1999). "Maxillofacial osteonecrosis in a patient with multiple "idiopathic" facial pains". J. Oral Pathol. Med. 28 (9): 423–32. PMID 10535367.
- ^ Zarychanski R, Elphee E, Walton P, Johnston J (2006). "Osteonecrosis of the jaw associated with pamidronate therapy". Am. J. Hematol. 81 (1): 73–5. doi:10.1002/ajh.20481. PMID 16369966.
- ^ Abu-Id MH, Açil Y, Gottschalk J, Kreusch T (2006). "[Bisphosphonate-associated osteonecrosis of the jaw]" (in German). Mund Kiefer Gesichtschir 10 (2): 73–81. doi:10.1007/s10006-005-0670-0. PMID 16456688.
- ^ Merigo E, Manfredi M, Meleti M, Corradi D, Vescovi P (2005). "Jaw bone necrosis without previous dental extractions associated with the use of bisphosphonates (pamidronate and zoledronate): a four-case report". J. Oral Pathol. Med. 34 (10): 613–7. doi:10.1111/j.1600-0714.2005.00351.x. PMID 16202082.
- ^ Gibbs SD, O'Grady J, Seymour JF, Prince HM (2005). "Bisphosphonate-induced osteonecrosis of the jaw requires early detection and intervention". Med. J. Aust. 183 (10): 549–50. PMID 16296983.
- ^ Bond TE Jr. (1848). A practical treatise on dental medicine. Philadelphia: Lindsay & Blakiston.
- ^ a b "Necrosis of the lower jaw in makers of Lucifer matches". Am J Dent Science 1 (series 3): 96–7. 1867.
- ^ a b Bouquot J.E. The history of maxillofacial osteonecrosis. Maxillofacial Center for Diagnostics and Research. Accessed 22 May 2006.
- ^ Ferguson W (1868). "New treatment of necrosis". Am J Dent Science 1 (series 3): 189.
- ^ Noel HR (1868). "A lecture on caries and necrosis of bone". Am J Dent Science 1 (series 3): 425, 482.
- ^ Barrett WC (1898). Oral pathology and practice. Philadelphia: S.S. White Dental Mfg Co.
- ^ Black GV (1915). A work on special dental pathology (2nd ed.). Chicago: Medico_Dental Publ Co.
- ^ Burns, Richard; Cohen, Stephen P. (1980). Pathways of the pulp (2nd ed.). St. Louis: Mosby. pp. 55–7. ISBN 0-8016-1009-5.
- ^ Imbeau J (2005). "Introduction to through-transmission alveolar ultrasonography (TAU) in dental medicine". Cranio 23 (2): 100–12. PMID 15898566. http://www.integrativedentalmedicine.com/PDF/Introduction%20to%20Through-transmission%20Ultrasonography%20in%20Dental%20Medicine%20-%20Dr.%20Jacques%20Imbeau.pdf.[dead link]
- ^ Bouquot J, Margolis M, Shankland WE, Imbeau J (April 2002). "Through-transmission alveolar sonography (TTAS) – a new technology for evaluation of medullary diseases, correlation with histopathology of 285 scanned jaw sites". 56th annual meeting of the American Academy of Oral and Maxillofacial Pathology.
- ^ Purcell PM, Boyd IW (2005). "Bisphosphonates and osteonecrosis of the jaw". Med. J. Aust. 182 (8): 417–8. PMID 15850440. http://www.mja.com.au/public/issues/182_08_180405/pur10144_fm.html.
- ^ J Carreyrou, Fosamax Drug Could Become Next Merck Woe, Dow Jones (THE WALL STREET JOURNAL), Apr. 12, 2006
- ^ Statement by Merck on Fosamax and rare cases of osteonecrosis of the jaws.- Accessed 21 May 2006.
- ^ Information on osteonecrosis of the jaws on the Novartis web site.- Accessed 21 May 2006.
- ^ Bouquot J, Wrobleski G, Fenton S (2000). "The most common osteonecrosis? Prevalence of maxillofacial osteonecrosis (MFO)". J Oral Pathol Med 29: 345.
- ^ Glueck CJ, McMahon RE, Bouquot J, et al. (1996). "Thrombophilia, hypofibrinolysis, and alveolar osteonecrosis of the jaws". Oral Surg Oral Med Oral Pathol Oral Radiol Endod 81 (5): 557–66. doi:10.1016/S1079-2104(96)80047-3. PMID 8734702.
- ^ Gruppo R, Glueck C, McMahon R, Bouquot J, Rabinovich B, Becker A, Tracy T, Wang P (1996). "The pathophysiology of alveolar osteonecrosis of the jaw: anticardiolipin antibodies, thrombophilia, and hypofibrinolysis". J Lab Clin Med 127 (5): 481–8. doi:10.1016/S0022-2143(96)90065-7. PMID 8621985.
External links
Osteochondropathy (M80–M94, 730–733) Osteopathies endocrine bone disease: Osteitis fibrosa cystica (Brown tumor)infectious bone disease: Osteomyelitis (Sequestrum, Involucrum) · Sesamoiditis · Brodie abscess · PeriostitisBone density
and structureDensity / metabolic bone diseaseContinuity of boneOtherFibrous dysplasia (Monostotic, Polyostotic) · Skeletal fluorosis · bone cyst (Aneurysmal bone cyst) · Hyperostosis (Infantile cortical hyperostosis) · Osteosclerosis (Melorheostosis)Avascular necrosis (Osteonecrosis of the jaw)OtherChondropathies OtherBoth lower limb: hip (Legg–Calvé–Perthes syndrome) · tibia (Osgood-Schlatter disease, Blount's disease) · foot (Köhler disease, Sever's disease)Categories:- Skeletal disorders
- Jaw disorders
Wikimedia Foundation. 2010.