- Diffuse panbronchiolitis
-
Diffuse panbronchiolitis Classification and external resources ICD-10 J21.9 ICD-9 466.1 OMIM 604809 DiseasesDB 3804 Diffuse panbronchiolitis (DPB) is an inflammatory lung disease of unknown cause. It is a severe, progressive form of bronchiolitis, which is inflammation of the bronchioles, small air passages in the lungs. The term "diffuse" refers to the lesions that appear throughout both lungs, while "panbronchiolitis" refers to the inflammation found in all layers of the respiratory bronchioles (the section of the bronchioles involved in gas exchange). DPB causes severe inflammation of the respiratory bronchioles, nodule-like lesions in respiratory and terminal bronchioles, chronic sinusitis and intense coughing with large amounts of sputum production.
The disease is believed to occur when there is susceptibility, or a lack of immune system resistance to DPB-causing bacteria or a virus, caused by several genes that are found predominantly in individuals of East Asian descent. The highest incidence occurs among the Japanese, followed by Koreans. DPB occurs more often in males, and usually begins around age 40. It was recognized as a distinct new disease in the early 1960s, and was formally named "diffuse panbronchiolitis" in 1969.
If left untreated, DPB progresses to bronchiectasis, an irreversible lung condition that involves enlargement of the bronchioles, and pooling of mucus in the bronchiolar passages. The eventual result of DPB can lead to respiratory failure and heart problems. Daily treatment of DPB with macrolide antibiotics such as erythromycin eases symptoms and increases survival time, but the disease has no cure.
 A diagram of the human respiratory system
A diagram of the human respiratory system
Contents
Classification
Bronchiolitis generally describes inflammation of the bronchioles.[1] DPB is classified as a form of primary bronchiolitis, which means that the underlying cause of bronchiolitis is originating from or is confined to the bronchioles.[2][3] Along with DPB, additional forms of primary bronchiolitis include bronchiolitis obliterans, follicular bronchiolitis, respiratory bronchiolitis, mineral dust airway disease, and a number of others.[2]
Signs and symptoms
Symptoms of DPB include chronic sinusitis (inflamed paranasal sinuses), a severe cough with large amounts of sputum (coughed-up phlegm), wheezing, crackles (respiratory sounds made by obstructions such as phlegm and secretions in the lungs), and dyspnea (shortness of breath). There may be pus in the sputum, and affected individuals may have fever. Signs of DPB that include dilation (enlargement) of the bronchiolar passages and hypoxemia (low levels of oxygen in the blood) are revealed via lung X-rays and blood gasses (a blood test from an artery, used to measure the oxygen and carbon dioxide content of the blood), respectively. If left untreated, DPB progresses and signs of bronchiectasis begin to present themselves. These include advancement of the dilation and thickening of the walls of the bronchioles, inflammatory damage to respiratory and terminal bronchioles, and pooling of mucus in the lungs.[4][5] DPB is associated with progressive respiratory failure, hypercapnia (increased levels of carbon dioxide in the blood), and can eventually lead to pulmonary hypertension (high blood pressure in the pulmonary vein and artery) and cor pulmonale (dilation of the right ventricle, or "right heart failure").[6][7]
Cause
DPB is idiopathic, which means an exact physiological, environmental, or pathogenic cause of the disease is unknown. However, several known factors are involved with the pathogenesis of DPB.[4]
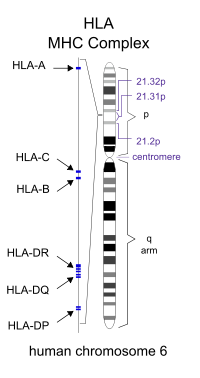
The major histocompatibility complex (MHC) is a large genomic region found in most vertebrates that is associated with the immune system. It is located on chromosome 6 in humans. A subset of MHC in humans is human leukocyte antigen (HLA), which controls the antigen-presenting system, as part of adaptive immunity against pathogens such as bacteria and viruses. When human cells are infected by a pathogen, some of them present parts of the pathogen's proteins on their surfaces; this is called antigen presentation. The infected cells then become targets for types of cytotoxic T-cells, which kill the infected cells so they can be removed from the body.[8]
Genetic predisposition for DPB susceptibility has been localized to two HLA haplotypes unique to Asians, particularly of East Asian descent. HLA-B54 is associated with DPB in Japanese individuals, while HLA-A11 is associated with the disease in Koreans.[9] Several genes within this region of class I HLA are believed to be a genetic factor responsible for DPB, by allowing increased susceptibility to the disease.[7][10] The common genetic background and similarities in the HLA profile of Japanese and Korean individuals were considered in the search for a DPB gene.[10] It was suggested that a mutation of a suspected disease-susceptibility gene located somewhere between HLA-B[11] and HLA-A[12] had occurred on an ancestral chromosome carrying both HLA-B54 and HLA-A11. Further, it is possible that a number of genetic recombination events around the disease locus (location on a chromosome) could have resulted in the disease being associated with HLA-B54 in the Japanese and HLA-A11 in Koreans. After further study of this localized area between HLA-B and HLA-A, it was concluded that a DPB susceptibility gene is located within a 200 kb (kilobase, or 1,000 base pairs) region of the 300 kb telomeric class I HLA, near the HLA-B locus at chromosome 6p21.3.[9][10]
Within this localized area of HLA, the search for a genetic cause of DPB has continued. As many genes belonging to HLA remain unidentified, positional cloning (a method used to identify a specific gene for a trait or disease, when only its location on a chromosome is known) has been used to determine that a mucin-like gene is associated with DPB. In addition, diseases known to be caused by identified HLA genes in the DPB-susceptibility region have been investigated. One of these, bare lymphocyte syndrome I (BLS I), exhibits a number of similarities with DPB in those affected, including chronic sinusitis, bronchiolar inflammation and nodules, and the presence of H. influenzae. Also like DPB, BLS I responds favorably to erythromycin therapy by showing a resolution of symptoms. The respiratory similarities between these two diseases, the corresponding success with the same mode of treatment, and the fact that the genetic mutation known to cause BLS I is located within the DPB-causing area of HLA narrows the establishment of a gene responsible for DPB.[9] Environmental factors such as inhaling toxic fumes and cigarette smoking are not believed to play a role in causing the disease.[6][7]
Cystic fibrosis (CF), a progressive multi-system lung disease, has been considered in the search for a genetic cause of DPB. This is for a number of reasons. CF, like DPB, causes severe lung inflammation, abundant mucus production, infection, and shows a genetic predominance among Caucasians of one geographic group to the rarity of others; whereas DPB dominates among East Asians, CF mainly affects individuals of European descent. While no gene has been implicated as the cause of DPB, mutation in a specific gene–much more likely to occur in Europeans–causes CF. This mutation in the CF-causing gene is not a factor in DPB, but a unique polymorphism in this gene is known to occur in many Asians not necessarily affected by either disease. It is wondered if this gene in any state of mutation could contribute to DPB.[4][9]
Pathophysiology
Inflammation is a normal part of the human immune response, whereby the immune system causes leukocytes (white blood cells) to gather and chemokines to accumulate at any location in the body where active bacterial or viral infections occur. Inflammation interferes with the activity of pathogens like bacteria and viruses, and serves to clear them from the body. In more advanced cases of DPB, bacteria such as Pseudomonas aeruginosa cause the proliferation of inflammatory cells into the bronchiolar tissues. However, when P. aeruginosa is not present with DPB, the inflammation continues for an as yet unknown reason.[4][5] In either circumstance, inflammation in DPB can be so severe that nodules containing inflammatory cells form in the walls of the bronchioles.[4][13] The presence of inflammation and infection in the airway also results in the production of excess mucus, which must be coughed up as sputum.[4][6] The combination of inflammation, nodule development, infection, mucus, and frequent cough contributes to the breathing difficulties in DPB.[4][5]
The inflammation in DPB persists with or without the presence of two bacteria typically found with the disease (P. aeruginosa and Haemophilus influenzae), and provides a means to determine several mechanisms of disease pathogenesis.[5] Leukotrienes are eicosanoids, signaling molecules made from essential fatty acids, which play a role in many lung diseases by causing the proliferation of inflammatory cells and excess mucus production in the airway.[14] In DPB and other lung diseases, the predominant mediator of neutrophil-related inflammation is leukotriene B4, which specializes in neutrophil proliferation via chemotaxis (the attraction of some types of cells to certain molecules specifically designed to attract them).[4][9]
Inflammation in DPB is also caused by the chemokine MIP-1alpha and its involvement with CD8+ T cells. The leukotrienes are an important contributor to inflammation in the respiratory tract. Beta defensins, a family of antimicrobial peptides found in the respiratory tract, are responsible for further inflammation in DPB when a pathogen such as P. aeruginosa is present. If present with DPB, the human T-lymphotropic virus, type I, a retrovirus, modifies DPB pathogenesis by infecting CD4+ T cells (T helper cells) and altering their effectiveness in recognizing the presence of known or unknown pathogens involved with DPB. Conversely, in individuals with human lymphotropic virus, onset of DPB increases the frequency of adult T-cell leukemia.[4][9]
Diagnosis
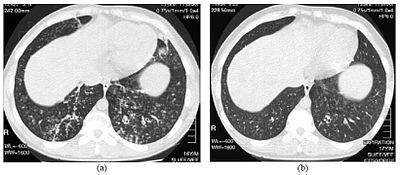 High resolution computed tomography (HRCT) images of the lower chest in a 16 year-old boy initially diagnosed with DPB (left), and 8 weeks later (right) after a 6-week course of treatment with erythromycin. The bilateral bronchiectasis and prominent centri-lobular nodules with a tree-in-bud pattern shows noticeable improvement.
High resolution computed tomography (HRCT) images of the lower chest in a 16 year-old boy initially diagnosed with DPB (left), and 8 weeks later (right) after a 6-week course of treatment with erythromycin. The bilateral bronchiectasis and prominent centri-lobular nodules with a tree-in-bud pattern shows noticeable improvement.
The diagnosis of DPB requires analysis of the lungs and bronchiolar tissues. Analysis of these tissues can require a lung biopsy, or the more preferred high resolution computed tomography (HRCT) scan of the lungs.[7] Distinguishing features of the disease that confirm its diagnosis include severe inflammation in all layers of the respiratory bronchioles, and lung tissue lesions that appear as nodules within the terminal and respiratory bronchioles in both lungs.[4] The nodules appear as opaque lumps when viewed on X-rays of the lung, and are responsible for airway obstruction in DPB, which is evaluated by a pulmonary function test, or PFT.[6] HRCT scans often show blockages of some bronchiolar passages with mucus, which is referred to as the "tree-in-bud" pattern.[7] Other findings observed with DPB include the proliferation of lymphocytes (white blood cells that fight infection), neutrophil granulocytes (white blood cells involved with inflammation), and foamy histiocytes (tissue macrophages) in the lung lumen. Bacteria such as H. influenzae and P. aeruginosa are also detectable, with the latter becoming more prominent as the disease progresses.[4][5] Elevated levels of IgG and IgA (classes of immunoglobulins) may be seen, as well as the presence of rheumatoid factor (an indicator of autoimmunity). Hemagglutination, a clumping of red blood cells in response to the presence of antibodies in the blood, may also occur. Neutrophils, beta-defensins, leukotrienes, and chemokines can also be detected in bronchoalveolar lavage (BAL) fluid taken from individuals with DPB.[4][9]
Differential diagnosis
In the differential diagnosis (finding the correct diagnosis between two or more suspected diseases that have common, or overlapping features) of some obstructive lung diseases, DPB is often considered. DPB is distinguished by the presence of lesions that appear on X-rays as nodules in the bronchioles of both lungs, inflammation in all tissue layers of the respiratory bronchioles, and its nearly exclusive prevalence among individuals with East Asian lineage.[4] A number of DPB symptoms resemble those found with three typical obstructive lung diseases: asthma, chronic bronchitis, and emphysema. While wheezing can be reminiscent of asthma, coughing with sputum production occurs with chronic bronchitis, and obstructive respiratory functional impairment occurs with emphysema.[6] Cystic fibrosis, like DPB, causes severe lung inflammation, excess mucus production, and infection; but DPB does not cause disturbances of the pancreas nor the electrolytes, as does CF, so the two diseases are thought to be entirely different and unrelated.[4][9]
DPB and bronchiolitis obliterans (or obliterative bronchiolitis, OB) are two forms of primary bronchiolitis.[2] Specific features of both diseases found to overlap include strong cough with large amounts of often pus-filled sputum, nodules viewable on lung X-rays in the lower bronchi and bronchiolar area, and chronic sinusitis. In DPB, the nodules are more restricted to the respiratory bronchioles, while in OB they are often found in the membranous bronchioles (the initial non-cartilaginous section of the bronchiole, that divides from the tertiary bronchus) up to the secondary bronchus. OB is a bronchiolar disease with worldwide prevalence, while DPB has more localized prevalence, predominantly in Japan.[2][6]
Treatment
Macrolide antibiotics, especially erythromycin, are an effective treatment for DPB when taken regularly over an extended period of time.[15][16] Long-term treatment of an individual with DPB consists of oral administration of erythromycin daily[7] for an indefinite period lasting two to three years, depending upon the success of treatment. Erythromycin therapy over an extended period has been shown to have a curative effect in some cases of DPB. This effect is considered when an individual undergoing treatment for DPB, among a number of disease-related remission criteria, has a normal neutrophil count detected in BAL fluid, and blood gas (an arterial blood test that measures the amount of oxygen and carbon dioxide in the blood) readings show that free oxygen in the blood is within the normal range. Allowing a temporary break from erythromycin therapy in these instances has been suggested, to reduce the formation of macrolide-resistant P. aeruginosa.[15] In those cases where treatment can be stopped for a time, however, DPB symptoms eventually return, and treatment would need to be resumed. Although highly effective, erythromycin may not prove successful in all individuals with the disease, particularly if macrolide-resistant P. aeruginosa is present or previously untreated DPB has progressed to the point where respiratory failure is occurring.[16][17]
The successful results of macrolides in DPB and similar lung diseases stems from managing certain symptoms through immunomodulation (adjusting the immune response),[16] which can be accomplished by taking macrolides in low doses. With erythromycin therapy in DPB, great reduction in bronchiolar inflammation and damage is achieved through suppression of not only neutrophil granulocyte proliferation, but also lymphocyte activity and obstructive mucus, water, and sputum secretions in airways.[15] The antimicrobial and antibiotic effects of macrolides are not believed to be primarily involved in their beneficial effects toward reducing inflammation in DPB.[17] This is evident, as the treatment dosage is much too low to fight infection, and in DPB cases with the occurrence of macrolide-resistant P. aeruginosa, macrolide therapy still produces substantial anti-inflammatory results.[15]
A number of factors are involved in suppression of inflammation by erythromycin or other macrolides. Macrolides are especially effective at inhibiting the proliferation and activity of neutrophils, by diminishing the ability of IL-8 and leukotriene B4 to attract them.[18] Macrolides also reduce the efficiency of adhesion molecules that allow neutrophils to stick to bronchiolar tissue linings. Mucus production in the airways is a major culprit in the morbidity and mortality of DPB and other respiratory diseases. The significant reduction of inflammation in DPB, attributed to erythromycin therapy, also greatly inhibits the production of excess mucus.[18]
Prognosis
Untreated DPB leads to bronchiectasis, respiratory failure, and death. A journal report from 1983 indicated that untreated DPB had a five-year survival rate of 62.1%, while the 10-year survival rate was 33.2%.[6] With erythromycin treatment, particularly if used early upon the initial diagnosis, individuals with DPB now have a much longer life expectancy due to increased success in management of symptoms, delay of progression, and prevention of associated infections like P. aeruginosa.[17] The 10-year survival rate for treated DPB is 90%.[4] In DPB cases where successful treatment with erythromycin has resulted in a curative effect, which sometimes happens after a treatment period lasting longer than two years, treatment has been allowed to end for a while. The prognosis for DPB when treatment has been this successful is considered to be very positive; however, individuals allowed to stop treatment during this time are closely monitored. As DPB has been proven to recur, erythromycin therapy must be promptly resumed once disease symptoms begin to reappear. In spite of the improved prognosis, DPB still has no cure.[4][9]
History
In the early 1960s, a relatively new chronic lung disease was being observed and described by physicians in Japan. In 1969,[19] the name "diffuse panbronchiolitis" was introduced to distinguish it from chronic bronchitis, emphysema, alveolitis, and several other obstructive lung disease with inflammation. Between 1978 and 1980, results of a nation-wide survey initiated by the Ministry of Health and Welfare of Japan revealed more than 1,000 probable cases of DPB, with 82 histologically confirmed. By the 1980s, it was internationally recognized as a distinct disease of the lungs.[4][6]
Before the 1980s, the prognosis or expected outcome of DPB was poor, especially in cases with superinfection (the emergence of a new viral or bacterial infection, in addition to the currently occurring infection) by P. aeruginosa.[13] DPB continued to have a very high mortality rate before generalized antibiotic treatment and oxygen therapy were beginning to be used routinely in the effort to manage symptoms. Around 1985, when long-term treatment with the macrolide antibiotic erythromycin became the standard for managing DPB, the prognosis significantly improved.[17] In 1990, the association with DPB and HLA was initially asserted.[9]
Epidemiology
DPB has its highest prevalence among the Japanese, at 11 per 100,000 population.[4] Korean,[20] Chinese,[21] and Thai[22] individuals with the disease have been reported as well. A genetic predisposition among East Asians is indicated.[23] Rarer cases of DPB, being those in individuals with non-Asian lineage, have also been noted.[24][25][26] The disease is slightly more common in males, with the male to female ratio at 1.4:2.1. The average onset of the disease is around age 40, and two-thirds of those affected with it are non-smokers.[7]
DPB has been reported in a few Asian immigrants and residents in western countries, including those of Europe and North America. Misdiagnosis has occurred in the west due to less recognition of the disease than in Asian countries. Relative to the large number of Asians living in the west, the small number of them thought to be affected by DPB suggests non-genetic factors may play some role in its cause. This rarity seen in western Asians may also be partly associated with misdiagnosis.[7][27]
References
- ^ Visscher, D. W.; Myers, J. L. (2006). "Bronchiolitis: the pathologist's perspective" (Free full text). Proceedings of the American Thoracic Society 3 (1): 41–47. doi:10.1513/pats.200512-124JH. PMID 16493150. http://pats.atsjournals.org/cgi/content/full/3/1/41.
- ^ a b c d Ryu, J. H.; Myers, J. L.; Swensen, S. J. (Dec 2003). "Bronchiolar disorders" (Free full text). American Journal of Respiratory and Critical Care Medicine 168 (11): 1277–1292. doi:10.1164/rccm.200301-053SO. PMID 14644923. http://ajrccm.atsjournals.org/cgi/content/full/168/11/1277.
- ^ Ryu, J. H. (Mar 2006). "Classification and approach to bronchiolar diseases". Current Opinion in Pulmonary Medicine 12 (2): 145–151. doi:10.1097/01.mcp.0000208455.80725.2a. PMID 16456385.
- ^ a b c d e f g h i j k l m n o p q r Poletti, V.; Casoni, G.; Chilosi, M.; Zompatori, M. (Oct 2006). "Diffuse panbronchiolitis" (Free full text). European Respiratory Journal 28 (4): 862–871. doi:10.1183/09031936.06.00131805. PMID 17012632. http://erj.ersjournals.com/content/28/4/862.long.
- ^ a b c d e Yanagihara, K.; Kadoto, J.; Kohno, S. (2001). "Diffuse panbronchiolitis—pathophysiology and treatment mechanisms". International Journal of Antimicrobial Agents 18 Suppl 1: S83–S87. PMID 11574201.
- ^ a b c d e f g h Homma, H.; Yamanaka, A.; Tanimoto, S.; Tamura, M.; Chijimatsu, Y.; Kira, S.; Izumi, T. (Jan 1983). "Diffuse panbronchiolitis. A disease of the transitional zone of the lung" (Free full text). Chest 83 (1): 63–69. doi:10.1378/chest.83.1.63. PMID 6848335. http://chestjournal.chestpubs.org/content/83/1/63.long.
- ^ a b c d e f g h Anthony, M.; Singham, S.; Soans, B.; Tyler, G. (Oct 2009). "Diffuse panbronchiolitis: not just an Asian disease: Australian case series and review of the literature" (Free full text). Biomedical Imaging Intervention Journal 5 (4): e19. doi:10.2349/biij.5.4.e19. PMC 3097723. PMID 21610988. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=3097723.
- ^ Morris, Alan George; Hewitt, C.; Young, Stephen (1994). The major histocompatibility complex: its genes and their roles in antigen presentation. 15. Pergamon. http://books.google.com/books?id=NVG6GwAACAAJ&dq=the+major+histocompatibility+complex&hl=en&ei=JCa6TuzjHIigiQLKkOHxBA&sa=X&oi=book_result&ct=result&resnum=2&ved=0CDIQ6AEwAQ.
- ^ a b c d e f g h i j Keicho, N.; Hijikata, M. (May 2011). "Genetic predisposition to diffuse panbronchiolitis" (Free full text). Respirology 16 (4): 581–588. doi:10.1111/j.1440-1843.2011.01946.x. PMID 21303426. http://onlinelibrary.wiley.com/doi/10.1111/j.1440-1843.2011.01946.x/full.
- ^ a b c Online 'Mendelian Inheritance in Man' (OMIM) Panbronchiolitis, Diffuse -604809 Retrieved November 11, 2011.
- ^ Online 'Mendelian Inheritance in Man' (OMIM) Human Leukocyte Antigen B -142830 Retrieved September 21, 2011.
- ^ Online 'Mendelian Inheritance in Man' (OMIM) Human Leukocyte Antigen A -142800 Retrieved September 21, 2011.
- ^ a b Azuma, A.; Kudoh, S. (May 2006). "Diffuse panbronchiolitis in East Asia" (Free full text). Respirology (Carlton, Vic.) 11 (3): 249–261. doi:10.1111/j.1440-1843.2006.00845.x. PMID 16635082. http://onlinelibrary.wiley.com/doi/10.1111/j.1440-1843.2006.00845.x/full.
- ^ Busse, W. W. (Jun 1998). "Leukotrienes and inflammation" (Free full text). American Journal of Respiratory Critical Care Medicine 157 (6 pt. 1): S210–S213. PMID 9620941. http://ajrccm.atsjournals.org/cgi/content/full/157/6/S210.
- ^ a b c d Keicho, N.; Kudoh, S. (2002). "Diffuse panbronchiolitis: Role of macrolides in therapy". American Journal of Respiratory Medicine 1 (2): 119–131. PMID 14720066.
- ^ a b c Lopez-Boado, Y. S.; Rubin, B. K. (Jun 2008). "Macrolides as immunomodulatory medications for the therapy of chronic lung diseases". Current Opinion in Pharmacology 8 (3): 286–291. doi:10.1016/j.coph.2008.01.010. PMID 18339582.
- ^ a b c d Schultz, M. J. (Jul 2004). "Macrolide activities beyond their antimicrobial effects: macrolides in diffuse panbronchiolitis and cystic fibrosis" (Free full text). Journal of Antimicrobial Chemotherapy 54 (1): 21–28. doi:10.1093/jac/dkh309. PMID 15190022. http://jac.oxfordjournals.org/content/54/1/21.long.
- ^ a b Tamaoki, J.; Kadota, J.; Takizawa, H. (Nov 2004). "Clinical implications of the immunomodulatory effects of macrolides". The American Journal of Medicine 117 Suppl 9A: 5S–11S. PMID 15586558.
- ^ Yamanaka, A.; Saiki, S.; Tamura, S.; Saito, K. (Mar 1969). "Problems in chronic obstructive bronchial diseases, with special reference to diffuse panbronchiolitis" (in Japanese). Naika 23 (3): 442–451. PMID 5783341.
- ^ Kim, Y. W.; Han, S. K.; Shim, Y. S.; Kim, K. Y.; Han, Y. C.; Seo, J. W.; Im, J. G. (1992). "The first report of diffuse panbronchiolitis in Korea: five case reports". Internal Medicine (Tokyo, Japan) 31 (5): 695–701. doi:10.2169/internalmedicine.31.695. PMID 1504438.
- ^ Chen, Y.; Kang, J.; Li, S. (Jan 2005). "Diffuse panbronchiolitis in China". Respirology 10 (1): 70–75. doi:10.1111/j.1440-1843.2005.00622.x. PMID 15691241.
- ^ Chantarotorn, S.; Palwatwichai, A.; Vattanathum, A.; Tantamacharik, D. (Aug 1999). "Diffuse panbronchiolitis, the first case reports in Thailand". Journal of the Medical Association of Thailand 82 (8): 833–838. PMID 10511794.
- ^ Hoiby, N. (Jun 1994). "Diffuse panbronchiolitis and cystic fibrosis: East meets West" (Free full text). Thorax 49 (6): 531–532. doi:10.1136/thx.49.6.531. PMC 474936. PMID 8016786. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=474936.
- ^ Fitzgerald, J. E.; King Jr, T. E.; Lynch, D. A.; Tuder, R. M.; Schwarz, M. I. (Aug 1996). "Diffuse panbronchiolitis in the United States". American Journal of Respiratory and Critical Care Medicine 154 (2 Pt 1): 497–503. PMID 8756828.
- ^ Martinez, J. A.; Guimarães, S. M.; Ferreira, R. G.; Pereira, C. A. (Mar 2000). "Diffuse panbronchiolitis in Latin America". The American Journal of the Medical Sciences 319 (3): 183–185. doi:10.1097/00000441-200003000-00008. PMID 10746829.
- ^ Sandrini, A.; Balter, M. S.; Chapman, K. R. (Nov-Dec 2003). "Diffuse panbronchiolitis in a Caucasian man in Canada". Canadian Respiratory Journal : journal of the Canadian Thoracic Society 10 (8): 449–451. PMID 14679410.
- ^ Brugiere, O.; Milleron, B.; Antoine, M.; Carette, M. F.; Philippe, C.; Mayaud, C. (Oct 1996). "Diffuse panbronchiolitis in an Asian immigrant" (Free full text). Thorax 51 (10): 1065–1067. doi:10.1136/thx.51.10.1065. PMC 472677. PMID 8977613. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC472677/pdf/thorax00329-0111.pdf.
Categories:- Genetic disorders with OMIM but no gene
- Respiratory diseases
- Rare diseases
Wikimedia Foundation. 2010.