- Pressure
-
This article is about pressure in the physical sciences. For other uses, see Pressure (disambiguation).
Pressure (the symbol: P) is the force per unit area applied in a direction perpendicular to the surface of an object. Gauge pressure is the pressure relative to the local atmospheric or ambient pressure.
Contents
Definition
Pressure is the effect of a force applied to a surface. Pressure is the amount of force acting per unit area. The symbol of pressure is P.[1][2]
Formula
Conjugate variables
of thermodynamicsPressure Volume (Stress) (Strain) Temperature Entropy Chemical potential Particle number Mathematically:
where:
- P is the pressure,
- F is the normal force,
- A is the area of the surface area on contact
Pressure is a scalar quantity. It relates the vector surface element (a vector normal to the surface) with the normal force acting on it. The pressure is the scalar proportionality constant that relates the two normal vectors:The minus sign comes from the fact that the force is considered towards the surface element, while the normal vector points outward.
It is incorrect (although rather usual) to say "the pressure is directed in such or such direction". The pressure, as a scalar, has no direction. It is the force given by the previous relationship to the quantity that has a direction, not the pressure. If we change the orientation of the surface element, the direction of the normal force changes accordingly, but the pressure remains the same.
Pressure is transmitted to solid boundaries or across arbitrary sections of fluid normal to these boundaries or sections at every point. It is a fundamental parameter in thermodynamics, and it is conjugate to volume.
Units
The SI unit for pressure is the pascal (Pa), equal to one newton per square meter (N/m2 or kg·m−1·s−2). This special name for the unit was added in 1971;[3] before that, pressure in SI was expressed simply as N/m2.
Non-SI measures such as pounds per square inch and bar are used in some parts of the world, primarily in the United States of America. The cgs unit of pressure is the barye (ba), equal to 1 dyn·cm−2 or 0.1 Pa. Pressure is sometimes expressed in grams-force/cm2, or as kg/cm2 and the like without properly identifying the force units. But using the names kilogram, gram, kilogram-force, or gram-force (or their symbols) as units of force is expressly forbidden in SI. The technical atmosphere (symbol: at) is 1 kgf/cm2 (14.223 psi).
Since a system under pressure has potential to perform work on its surroundings, pressure is a measure of potential energy stored per unit volume measured in J·m−3, related to energy density.
Some meteorologists prefer the hectopascal (hPa) for atmospheric air pressure, which is equivalent to the older unit millibar (mbar). Similar pressures are given in kilo pascals (kPa) in most other fields, where the hecto- prefix is rarely used. The inch of mercury is still used in the United States. Oceanographers usually measure underwater pressure in decibars (dbar) because an increase in pressure of 1 dbar is approximately equal to an increase in depth of 1 meter. Scuba divers often use a manometric rule of thumb: the pressure exerted by ten meters depth of water is approximately equal to one atmosphere. The increase in pressure at 34 feet of fresh water or 33 feet of sea water is one atm.
The standard atmosphere (atm) is an established constant. It is approximately equal to typical air pressure at earth mean sea level and is defined as follows:
- standard atmosphere = 101,325 Pa = 101.325 kPa = 1,013.25 hPa.
Because pressure is commonly measured by its ability to displace a column of liquid in a manometer, pressures are often expressed as a depth of a particular fluid (e.g., centimetres of water, mm or inches of mercury). The most common choices are mercury (Hg) and water; water is nontoxic and readily available, while mercury's high density allows a shorter column (and so a smaller manometer) to be used to measure a given pressure. The pressure exerted by a column of liquid of height h and density ρ is given by the hydrostatic pressure equation p = ρgh. Fluid density and local gravity can vary from one reading to another depending on local factors, so the height of a fluid column does not define pressure precisely. When millimeters of mercury or inches of mercury are quoted today, these units are not based on a physical column of mercury; rather, they have been given precise definitions that can be expressed in terms of SI units. One mmHg (millimeter of mercury) is equal to one torr. The water-based units still depend on the density of water, a measured, rather than defined, quantity. These manometric units are still encountered in many fields. Blood pressure is measured in millimeters of mercury in most of the world, and lung pressures in centimeters of water are still common.
Gauge pressure is often given in units with 'g' appended, e.g. 'kPag' or 'psig', and units for measurements of absolute pressure are sometimes given a suffix of 'a', to avoid confusion, for example 'kPaa', 'psia'. However, the US National Institute of Standards and Technology recommends that, to avoid confusion, any modifiers be instead applied to the quantity being measured rather than the unit of measure[4] For example, "Pg = 100 psi" rather than "P = 100 psig".
Differential pressure is expressed in units with 'd' appended; this type of measurement is useful when considering sealing performance or whether a valve will open or close.
Presently or formerly popular pressure units include the following:
- atmosphere (atm)
- manometric units:
- centimeter, inch, and millimeter of mercury (torr)
- millimeter, centimeter, meter, inch, and foot of water
- customary units:
- non-SI metric units:
Pressure units Pascal Bar Technical atmosphere Standard atmosphere Torr Pound per square inch Pa bar at atm torr psi 1 Pa ≡ 1 N/m2 10−5 1.0197×10−5 9.8692×10−6 7.5006×10−3 145.04×10−6 1 bar 105 ≡ 106 dyn/cm2 1.0197 0.98692 750.06 14.5037744 1 at 0.980665 ×105 0.980665 ≡ 1 kp/cm2 0.96784 735.56 14.223 1 atm 1.01325 ×105 1.01325 1.0332 ≡ p0 760 14.696 1 Torr 133.322 1.3332×10−3 1.3595×10−3 1.3158×10−3 = 1 mmHg 19.337×10−3 1 psi 6.895×103 68.948×10−3 70.307×10−3 68.046×10−3 51.715 ≡ 1 lbF/in2 Examples
As an example of varying pressures, a finger can be pressed against a wall without making any lasting impression; however, the same finger pushing a thumbtack can easily damage the wall. Although the force applied to the surface is the same, the thumbtack applies more pressure because the point concentrates that force into a smaller area. Pressure is transmitted to solid boundaries or across arbitrary sections of fluid normal to these boundaries or sections at every point. Unlike stress, pressure is defined as a scalar quantity.
Another example is of a common knife. If we try to cut a fruit with the flat side it obviously won't cut. But if we take the thin side, it will cut smoothly. The reason is that the flat side has a greater surface area (less pressure) and so it does not cut the fruit. When we take the thin side, the surface area is reduced and so it cuts the fruit easily and quickly. This is one example of a practical application of pressure.
The gradient of pressure is called the force density. For gases, pressure is sometimes measured not as an absolute pressure, but relative to atmospheric pressure; such measurements are called gage pressure (also spelled gauge pressure).[5] An example of this is the air pressure in an automobile tire, which might be said to be "220 kPa/32psi", but is actually 220 kPa/32 psi above atmospheric pressure. Since atmospheric pressure at sea level is about 100 kPa/14.7 psi, the absolute pressure in the tire is therefore about 320 kPa/46.7 psi. In technical work, this is written "a gage pressure of 220 kPa/32 psi". Where space is limited, such as on pressure gauges, name plates, graph labels, and table headings, the use of a modifier in parentheses, such as "kPa (gage)" or "kPa (absolute)", is permitted. In non-SI technical work, a gage pressure of 32 psi is sometimes written as "32 psig" and an absolute pressure as "32 psia", though the other methods explained above that avoid attaching characters to the unit of pressure are preferred.[6]
Gauge pressure is the relevant measure of pressure wherever one is interested in the stress on storage vessels and the plumbing components of fluidics systems. However, whenever equation-of-state properties, such as densities or changes in densities, must be calculated, pressures must be expressed in terms of their absolute values. For instance, if the atmospheric pressure is 100 kPa, a gas (such as helium) at 200 kPa (gage) (300 kPa [absolute]) is 50 % denser than the same gas at 100 kPa (gage) (200 kPa [absolute]). Focusing on gage values, one might erroneously conclude the first sample had twice the density of the second one.
Scalar nature
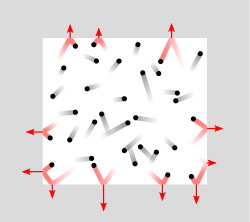
In a static gas, the gas as a whole does not appear to move. The individual molecules of the gas, however, are in constant random motion. Because we are dealing with an extremely large number of molecules and because the motion of the individual molecules is random in every direction, we do not detect any motion. If we enclose the gas within a container, we detect a pressure in the gas from the molecules colliding with the walls of our container. We can put the walls of our container anywhere inside the gas, and the force per unit area (the pressure) is the same. We can shrink the size of our "container" down to an infinitely small point, and the pressure has a single value at that point. Therefore, pressure is a scalar quantity, not a vector quantity. It has magnitude but no direction sense associated with it. Pressure acts in all directions at a point inside a gas. At the surface of a gas, the pressure force acts perpendicular (at right angle) to the surface.
A closely related quantity is the stress tensor σ, which relates the vector force F to the vector area A via
This tensor may be divided up into a scalar part (pressure) and a traceless tensor part shear. The shear tensor gives the force in directions parallel to the surface, usually due to viscous or frictional forces. The stress tensor is sometimes called the pressure tensor, but in the following, the term "pressure" will refer only to the scalar pressure.
According to the theory of general relativity pressure increases the strength of a gravitational field (see stress-energy tensor) and so adds to the mass-energy cause of gravity. This effect is unnoticeable at every-day pressures but is significant in neutron stars, although it has not been experimentally tested.[7]
Types
Fluid pressure
Fluid pressure is the pressure at some point within a fluid, such as water or air.
Fluid pressure occurs in one of two situations:
- 1. an open condition, called "open channel flow"
- a. the ocean, or
- b. swimming pool, or
- c. the atmosphere.
- 2. a closed condition, called closed conduits
- a. water line, or
- b. gas line.
Pressure in open conditions usually can be approximated as the pressure in "static" or non-moving conditions (even in the ocean where there are waves and currents), because the motions create only negligible changes in the pressure. Such conditions conform with principles of fluid statics. The pressure at any given point of a non-moving (static) fluid is called the hydrostatic pressure.
Closed bodies of fluid are either "static," when the fluid is not moving, or "dynamic," when the fluid can move as in either a pipe or by compressing an air gap in a closed container. The pressure in closed conditions conforms with the principles of fluid dynamics.
The concepts of fluid pressure are predominantly attributed to the discoveries of Blaise Pascal and Daniel Bernoulli. Bernoulli's Equation can be used in almost any situation to determine the pressure at any point in a fluid. The equation makes some assumptions about the fluid. Such as the fluid being ideal[8] and incompressible[8]. An ideal fluid is a fluid in which there is no friction, it is inviscid[8], zero viscosity[8]. The equation is written between any two points in a system that contain the same fluid.
Where:
- p = pressure of the fluid
- γ = ρg= density*acceleration of gravity = specific weight of the fluid.[8]
- v = velocity of the fluid
- g = acceleration of gravity
- z = elevation
 = pressure head
= pressure head = velocity head
= velocity head
Applications
- Artesian well
- Blood pressure
- Hydraulic head
- Plant cell turgidity
- Pythagorean cup
Explosion or deflagration pressures
Explosion or deflagration pressures are the result of the ignition of explosive gases, mists, dust/air suspensions, in unconfined and confined spaces.
Negative pressures
While pressures are, in general, positive, there are several situations in which negative pressures may be encountered:
- When dealing in relative (gauge) pressures. For instance, an absolute pressure of 80 kPa may be described as a gauge pressure of -21 kPa (i.e., 21 kPa below an atmospheric pressure of 101 kPa).
- When attractive forces (e.g., van der Waals forces) between the particles of a fluid exceed repulsive forces. Such scenarios are generally unstable since the particles will move closer together until repulsive forces balance attractive forces. Negative pressure exists in the transpiration pull of plants, and is used to suction water even higher than the ten metres that it rises in a pure vacuum.
- The Casimir effect can create a small attractive force due to interactions with vacuum energy; this force is sometimes termed 'vacuum pressure' (not to be confused with the negative gauge pressure of a vacuum).
- Depending on how the orientation of a surface is chosen, the same distribution of forces may be described either as a positive pressure along one surface normal, or as a negative pressure acting along the opposite surface normal.
- In the cosmological constant.
Stagnation pressure
Stagnation pressure is the pressure a fluid exerts when it is forced to stop moving. Consequently, although a fluid moving at higher speed will have a lower static pressure, it may have a higher stagnation pressure when forced to a standstill. Static pressure and stagnation pressure are related by the Mach number of the fluid. In addition, there can be differences in pressure due to differences in the elevation (height) of the fluid. See Bernoulli's equation (note: Bernoulli's equation only applies for incompressible, inviscid flow).
The pressure of a moving fluid can be measured using a Pitot tube, or one of its variations such as a Kiel probe or Cobra probe, connected to a manometer. Depending on where the inlet holes are located on the probe, it can measure static pressure or stagnation pressures.
Surface pressure
There is a two-dimensional analog of pressure – the lateral force per unit length applied on a line perpendicular to the force.
Surface pressure is denoted by π and shares many similar properties with three-dimensional pressure. Properties of surface chemicals can be investigated by measuring pressure/area isotherms, as the two-dimensional analog of Boyle's law, πA = k, at constant temperature.
Pressure of an ideal gas
Main article: Ideal gas lawIn an ideal gas, molecules have no volume and do not interact. Pressure varies linearly with temperature, volume, and quantity according to the ideal gas law,
where:
- P is the absolute pressure of the gas
- n is the amount of substance
- T is the absolute temperature
- V is the volume
- R is the ideal gas constant.
Real gases exhibit a more complex dependence on the variables of state.[10]
Vapor pressure
Main article: Vapor pressureVapor pressure is the pressure of a vapor in thermodynamic equilibrium with its condensed phases in a closed system. All liquids and solids have a tendency to evaporate into a gaseous form, and all gases have a tendency to condense back to their liquid or solid form.
The atmospheric pressure boiling point of a liquid (also known as the normal boiling point) is the temperature at which the vapor pressure equals the ambient atmospheric pressure. With any incremental increase in that temperature, the vapor pressure becomes sufficient to overcome atmospheric pressure and lift the liquid to form vapor bubbles inside the bulk of the substance. Bubble formation deeper in the liquid requires a higher pressure, and therefore higher temperature, because the fluid pressure increases above the atmospheric pressure as the depth increases.
The vapor pressure that a single component in a mixture contributes to the total pressure in the system is called partial vapor pressure.
Liquid pressure or pressure at depth
Used with liquid columns of constant density or at a depth within a substance (example: pressure at 20 km depth in the Earth).
- P = ρgh
Where:
- P is Pressure
- g is gravity at the surface of overlaying material
- ρ is density of liquid or overlaying material
- h is height of liquid or depth within a substance
See also
References
- ^ Giancoli, Douglas G. (2004). Physics: principles with applications. Upper Saddle River, N.J.: Pearson Education. ISBN 0-13-060620-0.
- ^ Note the upper case P is also used for power.
- ^ 14th Conference of the International Bureau of Weights and Measures
- ^ "Rules and Style Conventions for Expressing Values of Quantities". NIST. http://physics.nist.gov/Pubs/SP811/sec07.html#7.4. Retrieved 2009-07-07.
- ^ The preferred spelling varies by country and even by industry. Further, both spellings are often used within a particular industry or country. Industries in British English-speaking countries typically use the "gauge" spelling. Many of the largest American manufacturers of pressure transducers and instrumentation use the spelling "gage pressure" in their most formal documentation (Honeywell-Sensotec’s FAQ page and Fluke Corporation’s product search page).
- ^ NIST, Rules and Style Conventions for Expressing Values of Quantities, Sect. 7.4.
- ^ Einstein’s gravity under pressure
- ^ a b c d e Finnemore, John, E. and Joseph B. Franzini (2002). Fluid Mechanics: With Engineering Applications. New York, NY.: McGraw Hill, Inc.. pp. 14–29. ISBN 978-0-07-243202-2.
- ^ NCEES (2011). Fundamentals of Engineering: Supplied Reference Handbook. Clemson, SC: NCEES. pp. 64. ISBN 978-1-932613-59-9.
- ^ P. Atkins, J. de Paula Elements of Physical Chemistry, 4th Ed, W.H. Freeman, 2006. ISBN 0-7167-7329-5.
External links
Categories:- Atmospheric thermodynamics
- Underwater diving
- Fundamental physics concepts
- Fluid dynamics
- Fluid mechanics
- Hydraulics
- Pressure
- Thermodynamics
- State functions
Wikimedia Foundation. 2010.