- Vacuum
-
This article is about empty physical space or the absence of matter. For other uses, see Vacuum (disambiguation)."Free space" redirects here. For other uses, see Free space (disambiguation).
In everyday usage, vacuum is a volume of space that is essentially empty of matter, such that its gaseous pressure is much less than atmospheric pressure.[1] The word comes from the Latin term for "empty". A perfect vacuum would be one with no particles in it at all, which is impossible to achieve in practice. Physicists often discuss ideal test results that would occur in a perfect vacuum, which they simply call "vacuum" or "free space", and use the term partial vacuum to refer to an actual imperfect vacuum as one might have in a laboratory or in space. The Latin term in vacuo is also used to describe an object as being in what would otherwise be a vacuum.
The quality of a vacuum refers to how closely it approaches a perfect vacuum. Other things equal, lower gas pressure means higher-quality vacuum. For example, a typical vacuum cleaner produces enough suction to reduce air pressure by around 20%.[2] Much higher-quality vacuums are possible. Ultra-high vacuum chambers, common in chemistry, physics, and engineering, operate below one trillionth (10−12) of atmospheric pressure (100 nPa), and can reach around 100 particles/cm3.[3] Outer space is an even higher-quality vacuum, with the equivalent of just a few hydrogen atoms per cubic meter on average.[4] However, even if every single atom and particle could be removed from a volume, it would still not be "empty" due to vacuum fluctuations, dark energy, and other phenomena in quantum physics. In modern Particle Physics, the vacuum is considered as the ground state of matter.
Vacuum has been a frequent topic of philosophical debate since ancient Greek times, but was not studied empirically until the 17th century. Evangelista Torricelli produced the first laboratory vacuum in 1643, and other experimental techniques were developed as a result of his theories of atmospheric pressure. A torricellian vacuum is created by filling with mercury a tall glass container closed at one end and then inverting the container into a bowl to contain the mercury.[5]
Vacuum became a valuable industrial tool in the 20th century with the introduction of incandescent light bulbs and vacuum tubes, and a wide array of vacuum technology has since become available. The recent development of human spaceflight has raised interest in the impact of vacuum on human health, and on life forms in general.
 A large vacuum chamber
A large vacuum chamber
Contents
Etymology
From Latin vacuum (an empty space, void) noun use of neuter of vacuus (empty) related to vacare (be empty).
"Vacuum" is one of the few words in the English language that contains two consecutive 'u's[6].
Uses
 Light bulbs contain a partial vacuum, usually backfilled with argon, which protects the tungsten filament
Light bulbs contain a partial vacuum, usually backfilled with argon, which protects the tungsten filament
Vacuum is useful in a variety of processes and devices. Its first widespread use was in the incandescent light bulb to protect the filament from chemical degradation. The chemical inertness produced by a vacuum is also useful for electron beam welding, cold welding, vacuum packing and vacuum frying. Ultra-high vacuum is used in the study of atomically clean substrates, as only a very good vacuum preserves atomic-scale clean surfaces for a reasonably long time (on the order of minutes to days). High to ultra-high vacuum removes the obstruction of air, allowing particle beams to deposit or remove materials without contamination. This is the principle behind chemical vapor deposition, physical vapor deposition, and dry etching which are essential to the fabrication of semiconductors and optical coatings, and to surface science. The reduction of convection provides the thermal insulation of thermos bottles. Deep vacuum lowers the boiling point of liquids and promotes low temperature outgassing which is used in freeze drying, adhesive preparation, distillation, metallurgy, and process purging. The electrical properties of vacuum make electron microscopes and vacuum tubes possible, including cathode ray tubes. The elimination of air friction is useful for flywheel energy storage and ultracentrifuges.
Vacuum driven machines
Vacuums are commonly used to produce suction, which has an even wider variety of applications. The Newcomen steam engine used vacuum instead of pressure to drive a piston. In the 19th century, vacuum was used for traction on Isambard Kingdom Brunel's experimental atmospheric railway. Vacuum brakes were once widely used on trains in the UK but, except on heritage railways, they have been replaced by air brakes.
Manifold vacuum can be used to drive accessories on automobiles. The best-known application is the vacuum servo, used to provide power assistance for the brakes. Obsolete applications include vacuum-driven windscreen wipers and fuel pumps.
Outer space
Main article: Outer space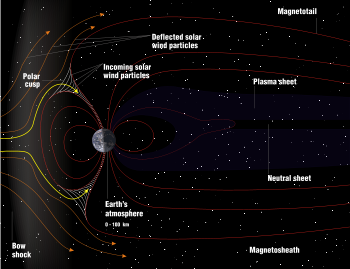 Outer space is not a perfect vacuum, but a tenuous plasma awash with charged particles, electromagnetic fields, and the occasional star.
Outer space is not a perfect vacuum, but a tenuous plasma awash with charged particles, electromagnetic fields, and the occasional star.
Outer space has very low density and pressure, and is the closest physical approximation of a perfect vacuum. It has effectively no friction, allowing stars, planets and moons to move freely along ideal gravitational trajectories. But no vacuum is truly perfect, not even in interstellar space, where there are still a few hydrogen atoms per cubic centimeter.[4]
Stars, planets and moons keep their atmospheres by gravitational attraction, and as such, atmospheres have no clearly delineated boundary: the density of atmospheric gas simply decreases with distance from the object. The Earth's atmospheric pressure drops to about 3.2 × 10−2 Pa at 100 kilometres (62 mi) of altitude,[7] the Kármán line, which is a common definition of the boundary with outer space. Beyond this line, isotropic gas pressure rapidly becomes insignificant when compared to radiation pressure from the sun and the dynamic pressure of the solar wind, so the definition of pressure becomes difficult to interpret. The thermosphere in this range has large gradients of pressure, temperature and composition, and varies greatly due to space weather. Astrophysicists prefer to use number density to describe these environments, in units of particles per cubic centimetre.
But although it meets the definition of outer space, the atmospheric density within the first few hundred kilometers above the Kármán line is still sufficient to produce significant drag on satellites. Most artificial satellites operate in this region called low earth orbit and must fire their engines every few days to maintain orbit.[citation needed] The drag here is low enough that it could theoretically be overcome by radiation pressure on solar sails, a proposed propulsion system for interplanetary travel. Planets are too massive for their trajectories to be significantly affected by these forces, although their atmospheres are eroded by the solar winds.
All of the observable universe is filled with large numbers of photons, the so-called cosmic background radiation, and quite likely a correspondingly large number of neutrinos. The current temperature of this radiation is about 3 K, or -270 degrees Celsius or -454 degrees Fahrenheit.
Effects on humans and animals
See also: Human adaptation to space This painting, An Experiment on a Bird in the Air Pump by Joseph Wright of Derby, 1768, depicts an experiment performed by Robert Boyle in 1660.
This painting, An Experiment on a Bird in the Air Pump by Joseph Wright of Derby, 1768, depicts an experiment performed by Robert Boyle in 1660.
Humans and animals exposed to vacuum will lose consciousness after a few seconds and die of hypoxia within minutes, but the symptoms are not nearly as graphic as commonly depicted in media and popular culture. The reduction in pressure lowers the temperature at which blood and other body fluids boil, but the elastic pressure of blood vessels ensures that this boiling point remains above the internal body temperature of 37°C.[8] Although the blood will not boil, the formation of gas bubbles in bodily fluids at reduced pressures, known as ebullism, is still a concern. The steam may bloat the body to twice its normal size and slow circulation, but tissues are elastic and porous enough to prevent rupture.[9] Swelling and ebullism can be restrained by containment in a flight suit. Shuttle astronauts wear a fitted elastic garment called the Crew Altitude Protection Suit (CAPS) which prevents ebullism at pressures as low as 2 kPa (15 Torr).[10] Rapid boiling will cool the skin and create frost, particularly in the mouth, but this is not a significant hazard.
Animal experiments show that rapid and complete recovery is normal for exposures shorter than 90 seconds, while longer full-body exposures are fatal and resuscitation has never been successful.[11] There is only a limited amount of data available from human accidents, but it is consistent with animal data. Limbs may be exposed for much longer if breathing is not impaired.[12] Robert Boyle was the first to show in 1660 that vacuum is lethal to small animals.
During 1942, in one of a series of experiments on human subjects for the Luftwaffe, the Nazi regime experimented on prisoners in Dachau concentration camp by exposing them to low pressure.[13]
Cold or oxygen-rich atmospheres can sustain life at pressures much lower than atmospheric, as long as the density of oxygen is similar to that of standard sea-level atmosphere. The colder air temperatures found at altitudes of up to 3 km generally compensate for the lower pressures there.[12] Above this altitude, oxygen enrichment is necessary to prevent altitude sickness in humans that did not undergo prior acclimatization, and spacesuits are necessary to prevent ebullism above 19 km.[12] Most spacesuits use only 20 kPa (150 Torr) of pure oxygen, just enough to sustain full consciousness. This pressure is high enough to prevent ebullism, but simple evaporation of blood can still cause decompression sickness and gas embolisms if not managed.[citation needed]
Rapid decompression can be much more dangerous than vacuum exposure itself. Even if the victim does not hold his or her breath, venting through the windpipe may be too slow to prevent the fatal rupture of the delicate alveoli of the lungs.[12] Eardrums and sinuses may be ruptured by rapid decompression, soft tissues may bruise and seep blood, and the stress of shock will accelerate oxygen consumption leading to hypoxia.[14] Injuries caused by rapid decompression are called barotrauma. A pressure drop of 13 kPa (100 Torr), which produces no symptoms if it is gradual, may be fatal if it occurs suddenly.[12]
Some extremophile microrganisms, such as tardigrades, can survive vacuum for a period of days.
Historical interpretation
Historically, there has been much dispute over whether such a thing as a vacuum can exist. Ancient Greek philosophers did not like to admit the existence of a vacuum, asking themselves "how can 'nothing' be something?". Plato found the idea of a vacuum inconceivable. He believed that all physical things were instantiations of an abstract Platonic ideal, and he could not conceive of an "ideal" form of a vacuum. Similarly, Aristotle considered the creation of a vacuum impossible — nothing could not be something. Later Greek philosophers thought that a vacuum could exist outside the cosmos, but not within it. Hero of Alexandria was the first to challenge this belief in the first century AD, but his attempts to create an artificial vacuum failed.[15]
In the Roman city of Pompeii, a dual-action suction pump was found, proving that the ancient Romans had access to this kind of technology. Used for raising water, this pump had two cylinders, alternately operated by a walking-beam pump. In the suction phase, a lower valve opened, permitting the entry of water into the cylinder, while an upper valve remained closed. When the piston went down, the lower valve closed and the upper one opened.[16]
In the medieval Islamic world, the Muslim physicist and philosopher, Al-Farabi (Alpharabius, 872-950), conducted a small experiment concerning the existence of vacuum, in which he investigated handheld plungers in water.[17][unreliable source?] He concluded that air's volume can expand to fill available space, and he suggested that the concept of perfect vacuum was incoherent.[18] However, the Muslim physicist Ibn al-Haytham (Alhazen, 965-1039) and the Mu'tazili theologians disagreed with Aristotle and Al-Farabi, and they supported the existence of a void. Using geometry, Ibn al-Haytham mathematically demonstrated that place (al-makan) is the imagined three-dimensional void between the inner surfaces of a containing body.[19] Abū Rayhān al-Bīrūnī also states that "there is no observable evidence that rules out the possibility of vacuum".[20] The suction pump was described in 1206 by the Muslim engineer and inventor, Al-Jazari.[citation needed] The suction pump later appeared in Europe from the 15th century.[21][22][23] Taqi al-Din's six-cylinder 'Monobloc' pump, invented in 1551, could also create a partial vacuum.[citation needed]
In medieval Europe, the Catholic Church regarded the idea of a vacuum as against nature or even heretical; the absence of anything implied the absence of God, and harkened back to the void prior to the creation story in the Book of Genesis.[24] Medieval thought experiments into the idea of a vacuum considered whether a vacuum was present, if only for an instant, between two flat plates when they were rapidly separated.[24] There was much discussion of whether the air moved in quickly enough as the plates were separated, or, as Walter Burley postulated, whether a 'celestial agent' prevented the vacuum arising. The commonly held view that nature abhorred a vacuum was called horror vacui. Speculation that even God could not create a vacuum if he wanted to was shut down[clarification needed] by the 1277 Paris condemnations of Bishop Etienne Tempier, which required there to be no restrictions on the powers of God, which led to the conclusion that God could create a vacuum if he so wished.[25] René Descartes also argued against the existence of a vacuum, arguing along the following lines: "Space is identical with extension, but extension is connected with bodies; thus there is no space without bodies and hence no empty space (vacuum)." In spite of this, opposition to the idea of a vacuum existing in nature continued into the Scientific Revolution, with scholars such as Paolo Casati taking an anti-vacuist position. Jean Buridan reported in the 14th century that teams of ten horses could not pull open bellows when the port was sealed, apparently because of horror vacui.[15]
The belief in horror vacui was overthrown in the 17th century. Water pump designs had improved by then to the point that they produced measurable vacuums, but this was not immediately understood. What was known was that suction pumps could not pull water beyond a certain height: 18 Florentine yards according to a measurement taken around 1635. (The conversion to metres is uncertain, but it would be about 9 or 10 metres.) This limit was a concern to irrigation projects, mine drainage, and decorative water fountains planned by the Duke of Tuscany, so the Duke commissioned Galileo to investigate the problem. Galileo advertised the puzzle to other scientists, including Gasparo Berti who replicated it by building the first water barometer in Rome in 1639.[26] Berti's barometer produced a vacuum above the water column, but he could not explain it. The breakthrough was made by Evangelista Torricelli in 1643. Building upon Galileo's notes, he built the first mercury barometer and wrote a convincing argument that the space at the top was a vacuum. The height of the column was then limited to the maximum weight that atmospheric pressure could support. Some people believe that although Torricelli's experiment was crucial, it was Blaise Pascal's experiments that proved the top space really contained vacuum.
In 1654, Otto von Guericke invented the first vacuum pump[27] and conducted his famous Magdeburg hemispheres experiment, showing that teams of horses could not separate two hemispheres from which the air had been (partially) evacuated. Robert Boyle improved Guericke's design and conducted experiments on the properties of vacuum. Robert Hooke also helped Boyle produce an air pump which helped to produce the vacuum. The study of vacuum then lapsed until 1850 when August Toepler invented the Toepler Pump. Then in 1855 Heinrich Geissler invented the mercury displacement pump and achieved a record vacuum of about 10 Pa (0.1 Torr). A number of electrical properties become observable at this vacuum level, and this renewed interest in vacuum. This, in turn, led to the development of the vacuum tube. Shortly after this Hermann Sprengel invented the Sprengel Pump in 1865.
While outer space has been likened to a vacuum, early theories of the nature of light relied upon the existence of an invisible, aetherial medium which would convey waves of light. (Isaac Newton relied on this idea to explain refraction and radiated heat).[28] This evolved into the luminiferous aether of the 19th century, but the idea was known to have significant shortcomings - specifically, that if the Earth were moving through a material medium, the medium would have to be both extremely tenuous (because the Earth is not detectably slowed in its orbit), and extremely rigid (because vibrations propagate so rapidly). An 1891 article by William Crookes noted: "the [freeing of] occluded gases into the vacuum of space".[29] Even up until 1912, astronomer Henry Pickering commented: "While the interstellar absorbing medium may be simply the ether, [it] is characteristic of a gas, and free gaseous molecules are certainly there".[30]
In 1887, the Michelson-Morley experiment, using an interferometer to attempt to detect the change in the speed of light caused by the Earth moving with respect to the aether, was a famous null result. Many misinterpreted the results, which neither proved nor disproved the existence of the aether, as showing that there really was no static, pervasive medium throughout space and through which the Earth moved as though through a wind.[31][32] As a simplification, one can assume there no aether, and no such entity is required for the propagation of light. Besides the various particles which comprise cosmic radiation, there is a cosmic background of photonic radiation (electromagnetic radiation), including the cosmic microwave background (CMB), the thermal remnant of the Big Bang at about 2.7 K. However, none of these findings affect the outcome of the Michelson-Morley experiment to any significant degree.
Einstein argued that physical objects are not located in space, but rather have a spatial extent. Seen this way, the concept of empty space loses its meaning.[33] Rather, space is an abstraction, based on the relationships between local objects. Nevertheless, the general theory of relativity admits a pervasive gravitational field, which, in Einstein's words,[34] may be regarded as an "aether", with properties varying from one location to another. One must take care, though, to not ascribe to it material properties such as velocity and so on.
In 1930, Paul Dirac proposed a model of vacuum as an infinite sea of particles possessing negative energy, called the Dirac sea. This theory helped refine the predictions of his earlier formulated Dirac equation, and successfully predicted the existence of the positron, discovered two years later in 1932. Despite this early success, the idea was soon abandoned in favour of the more elegant quantum field theory.
The development of quantum mechanics has complicated the modern interpretation of vacuum by requiring indeterminacy. Niels Bohr and Werner Heisenberg's uncertainty principle and Copenhagen interpretation, formulated in 1927, predict a fundamental uncertainty in the instantaneous measurability of the position and momentum of any particle, and which, not unlike the gravitational field, questions the emptiness of space between particles. In the late 20th century, this principle was understood to also predict a fundamental uncertainty in the number of particles in a region of space, leading to predictions of virtual particles arising spontaneously out of the void. In other words, there is a lower bound on the vacuum, dictated by the lowest possible energy state of the quantized fields in any region of space.
In electromagnetism
In classical electromagnetism, free space or perfect vacuum is a standard reference medium for electromagnetic effects.[35][36]
In the theory of classical electromagnetism, free space has the following properties:
- Electromagnetic radiation travels without obstructions, at the speed of light.
- The superposition principle is always exactly true.[37] For example, the electric potential generated by two charges is the simple addition of the potentials generated by each charge in isolation. The value of the electric field at any point around these two charges is found by calculating the vector sum of the two electric fields from each of the charges acting alone.
- The permittivity and permeability are exactly ε0 and μ0 respectively (in SI units), or exactly 1 (in Gaussian units).
- The characteristic impedance (η) equals the impedance of free space Z0 ≈ 376.73 Ω.
In quantum mechanics
For more details on this topic, see vacuum state.In quantum mechanics and quantum field theory, the vacuum is defined as the state (i.e. solution to the equations of the theory) with the lowest possible energy (the ground state of the Hilbert space). This is a state with no matter particles (hence the name), and also no photons, no gravitons, etc. As described above, this state is impossible to achieve experimentally. (Even if every matter particle could somehow be removed from a volume, it would be impossible to eliminate all the blackbody photons.)
This hypothetical vacuum state often has interesting and complex properties. For example, it contains vacuum fluctuations (virtual particles that hop into and out of existence). It also, relatedly, has a finite energy, called vacuum energy. Vacuum fluctuations are an essential and ubiquitous part of quantum field theory. Some readily-apparent effects of vacuum fluctuations include the Casimir effect and Lamb shift.[25]
There can be more than one possible vacuum state. The starting and ending of cosmological inflation is thought to have arisen from transitions between different vacuum states. For theories obtained by quantization of a classical theory, each stationary point of the energy in the configuration space gives rise to a single vacuum. String theory is believed to have a huge number of vacua - the so-called string theory landscape.
In the superfluid vacuum theory the physical vacuum is described as the quantum superfluid which is essentially non-relativistic whereas the Lorentz symmetry is an approximate emerging symmetry valid only for the small fluctuations of the superfluid background. An observer who resides inside such vacuum and is capable of creating and/or measuring the small fluctuations would observe them as relativistic objects - unless their energy and momentum are sufficiently high (as compared to the background ones) to make the Lorentz-breaking corrections detectable. It was shown that the relativistic gravity arises as the small-amplitude collective excitation mode whereas the relativistic elementary particles can be described by the particle-like modes in the low-momentum limit.
Pumping and ambient air pressure
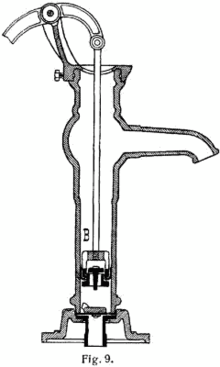 This shallow water well pump reduces atmospheric air pressure inside the pump chamber. Atmospheric pressure extends down into the well, and forces water up the pipe into the pump to balance the reduced pressure. Above-ground pump chambers are only effective to a depth of approximately 9 meters due to the water column weight balancing the atmospheric pressure.
This shallow water well pump reduces atmospheric air pressure inside the pump chamber. Atmospheric pressure extends down into the well, and forces water up the pipe into the pump to balance the reduced pressure. Above-ground pump chambers are only effective to a depth of approximately 9 meters due to the water column weight balancing the atmospheric pressure.
 Deep wells have the pump chamber down in the well close to the water surface, or in the water. A "sucker rod" extends from the handle down the center of the pipe deep into the well to operate the plunger. The pump handle acts as a heavy counterweight against both the sucker rod weight and the weight of the water column standing on the upper plunger up to ground level.
Deep wells have the pump chamber down in the well close to the water surface, or in the water. A "sucker rod" extends from the handle down the center of the pipe deep into the well to operate the plunger. The pump handle acts as a heavy counterweight against both the sucker rod weight and the weight of the water column standing on the upper plunger up to ground level. Main article: Vacuum pump
Main article: Vacuum pumpFluids cannot generally be pulled, so a vacuum cannot be created by suction. Suction can spread and dilute a vacuum by letting a higher pressure push fluids into it, but the vacuum has to be created first before suction can occur. The easiest way to create an artificial vacuum is to expand the volume of a container. For example, the diaphragm muscle expands the chest cavity, which causes the volume of the lungs to increase. This expansion reduces the pressure and creates a partial vacuum, which is soon filled by air pushed in by atmospheric pressure.
To continue evacuating a chamber indefinitely without requiring infinite growth, a compartment of the vacuum can be repeatedly closed off, exhausted, and expanded again. This is the principle behind positive displacement pumps, like the manual water pump for example. Inside the pump, a mechanism expands a small sealed cavity to create a vacuum. Because of the pressure differential, some fluid from the chamber (or the well, in our example) is pushed into the pump's small cavity. The pump's cavity is then sealed from the chamber, opened to the atmosphere, and squeezed back to a minute size.
 A cutaway view of a turbomolecular pump, a momentum transfer pump used to achieve high vacuum
A cutaway view of a turbomolecular pump, a momentum transfer pump used to achieve high vacuum
The above explanation is merely a simple introduction to vacuum pumping, and is not representative of the entire range of pumps in use. Many variations of the positive displacement pump have been developed, and many other pump designs rely on fundamentally different principles. Momentum transfer pumps, which bear some similarities to dynamic pumps used at higher pressures, can achieve much higher quality vacuums than positive displacement pumps. Entrapment pumps can capture gases in a solid or absorbed state, often with no moving parts, no seals and no vibration. None of these pumps are universal; each type has important performance limitations. They all share a difficulty in pumping low molecular weight gases, especially hydrogen, helium, and neon.
The lowest pressure that can be attained in a system is also dependent on many things other than the nature of the pumps. Multiple pumps may be connected in series, called stages, to achieve higher vacuums. The choice of seals, chamber geometry, materials, and pump-down procedures will all have an impact. Collectively, these are called vacuum technique. And sometimes, the final pressure is not the only relevant characteristic. Pumping systems differ in oil contamination, vibration, preferential pumping of certain gases, pump-down speeds, intermittent duty cycle, reliability, or tolerance to high leakage rates.
In ultra high vacuum systems, some very "odd" leakage paths and outgassing sources must be considered. The water absorption of aluminium and palladium becomes an unacceptable source of outgassing, and even the adsorptivity of hard metals such as stainless steel or titanium must be considered. Some oils and greases will boil off in extreme vacuums. The permeability of the metallic chamber walls may have to be considered, and the grain direction of the metallic flanges should be parallel to the flange face.
The lowest pressures currently achievable in laboratory are about 10−13 torr (13 pPa).[38] However, pressures as low as 5×10−17 Torr (6.7 fPa) have been indirectly measured in a 4 K cryogenic vacuum system.[3] This corresponds to ≈100 particles/cm3.
Outgassing
Main article: OutgassingEvaporation and sublimation into a vacuum is called outgassing. All materials, solid or liquid, have a small vapour pressure, and their outgassing becomes important when the vacuum pressure falls below this vapour pressure. In man-made systems, outgassing has the same effect as a leak and can limit the achievable vacuum. Outgassing products may condense on nearby colder surfaces, which can be troublesome if they obscure optical instruments or react with other materials. This is of great concern to space missions, where an obscured telescope or solar cell can ruin an expensive mission.
The most prevalent outgassing product in man-made vacuum systems is water absorbed by chamber materials. It can be reduced by desiccating or baking the chamber, and removing absorbent materials. Outgassed water can condense in the oil of rotary vane pumps and reduce their net speed drastically if gas ballasting is not used. High vacuum systems must be clean and free of organic matter to minimize outgassing.
Ultra-high vacuum systems are usually baked, preferably under vacuum, to temporarily raise the vapour pressure of all outgassing materials and boil them off. Once the bulk of the outgassing materials are boiled off and evacuated, the system may be cooled to lower vapour pressures and minimize residual outgassing during actual operation. Some systems are cooled well below room temperature by liquid nitrogen to shut down residual outgassing and simultaneously cryopump the system.
Quality
The quality of a vacuum is indicated by the amount of matter remaining in the system, so that a high quality vacuum is one with very little matter left in it. Vacuum is primarily measured by its absolute pressure, but a complete characterization requires further parameters, such as temperature and chemical composition. One of the most important parameters is the mean free path (MFP) of residual gases, which indicates the average distance that molecules will travel between collisions with each other. As the gas density decreases, the MFP increases, and when the MFP is longer than the chamber, pump, spacecraft, or other objects present, the continuum assumptions of fluid mechanics do not apply. This vacuum state is called high vacuum, and the study of fluid flows in this regime is called particle gas dynamics. The MFP of air at atmospheric pressure is very short, 70 nm, but at 100 mPa (~1×10−3 Torr) the MFP of room temperature air is roughly 100 mm, which is on the order of everyday objects such as vacuum tubes. The Crookes radiometer turns when the MFP is larger than the size of the vanes.
Vacuum quality is subdivided into ranges according to the technology required to achieve it or measure it. These ranges do not have universally agreed definitions, but a typical distribution is as follows:[39][40]
pressure (Torr) pressure (Pa) Atmospheric pressure 760 101.3 kPa Low vacuum 760 to 25 100 kPa to 3 kPa Medium vacuum 25 to 1×10−3 3 kPa to 100 mPa High vacuum 1×10−3 to 1×10−9 100 mPa to 100 nPa Ultra high vacuum 1×10−9 to 1×10−12 100 nPa to 100 pPa Extremely high vacuum <1×10−12 <100 pPa Outer Space 1×10−6 to <3×10−17 100 µPa to <3fPa Perfect vacuum 0 0 Pa - Atmospheric pressure is variable but standardized at 101.325 kPa (760 Torr)
- Low vacuum, also called rough vacuum or coarse vacuum, is vacuum that can be achieved or measured with rudimentary equipment such as a vacuum cleaner and a liquid column manometer.
- Medium vacuum is vacuum that can be achieved with a single pump, but the pressure is too low to measure with a liquid or mechanical manometer. It can be measured with a McLeod gauge, thermal gauge or a capacitive gauge.
- High vacuum is vacuum where the MFP of residual gases is longer than the size of the chamber or of the object under test. High vacuum usually requires multi-stage pumping and ion gauge measurement. Some texts differentiate between high vacuum and very high vacuum.
- Ultra high vacuum requires baking the chamber to remove trace gases, and other special procedures. British and German standards define ultra high vacuum as pressures below 10−6 Pa (10−8 Torr).[41][42]
- Deep space is generally much more empty than any artificial vacuum. It may or may not meet the definition of high vacuum above, depending on what region of space and astronomical bodies are being considered. For example, the MFP of interplanetary space is smaller than the size of the solar system, but larger than small planets and moons. As a result, solar winds exhibit continuum flow on the scale of the solar system, but must be considered as a bombardment of particles with respect to the Earth and Moon.
- Perfect vacuum is an ideal state of no particles at all. It cannot be achieved in a laboratory, although there may be small volumes which, for a brief moment, happen to have no particles of matter in them. Even if all particles of matter were removed, there would still be photons and gravitons, as well as dark energy, virtual particles, and other aspects of the quantum vacuum.
- Hard vacuum and Soft vacuum are terms that are defined with a dividing line defined differently by different sources, such as 5 psia,[43] one Torr,[44] or 0.1 Torr[45] the common denominator being that a hard vacuum is a higher vacuum than a soft one.
Examples
pressure (Pa) pressure (Torr) mean free path molecules per cm3 Vacuum cleaner approximately 80 kPa 600 70 nm 1019 liquid ring vacuum pump approximately 3.2 kPa 24 1.75 μm 1018 freeze drying 100 to 10 Pa 1 to 0.1 100 μm to 1 mm 1016 to 1015 rotary vane pump 100 Pa to 100 mPa 1 to 10−3 100 μm to 10 cm 1016 to 1013 Incandescent light bulb 10 to 1 Pa 0.1 to 0.01 1 mm to 1 cm 1015 to 1014 Thermos bottle 1 to 0.01 Pa[1] 10−2 to 10−4 1 cm to 1 m 1014 to 1012 Earth thermosphere 1 Pa to 100 nPa 10−2 to 10−9 1 cm to 100 km 1014 to 107 Vacuum tube 10 µPa to 10 nPa 10−7 to 10−10 1 to 1,000 km 109 to 106 Cryopumped MBE chamber 100 nPa to 1 nPa 10−9 to 10−11 100 to 10,000 km 107 to 105 Pressure on the Moon approximately 1 nPa 10−11 10,000 km 4×105 [46] Interplanetary space 10 [1] Interstellar space 1 [47] Intergalactic space 10−6 [1] Measurement
Main article: Pressure measurementRelative versus absolute measurement
Vacuum is measured in units of pressure, typically as a subtraction relative to ambient atmospheric pressure on Earth. But the amount of relative measurable vacuum varies with local conditions. On the surface of Jupiter, where ground level atmospheric pressure is much higher than on Earth, much higher relative vacuum readings would be possible. On the surface of the moon with almost no atmosphere, it would be extremely difficult to create a measurable vacuum relative to the local environment.
Similarly, much higher than normal relative vacuum readings are possible deep in the Earth's ocean. A submarine maintaining an internal pressure of 1 atmosphere submerged to a depth of 10 atmospheres (98 meters; a 9.8 meter column of seawater has the equivalent weight of 1 atm) is effectively a vacuum chamber keeping out the crushing exterior water pressures, though the 1 atm inside the submarine would not normally be considered a vacuum.
Therefore to properly understand the following discussions of vacuum measurement, it is important that the reader assumes the relative measurements are being done on Earth at sea level, at exactly 1 atmosphere of ambient atmospheric pressure.
Vacuum measurements relative to 1 atm
The SI unit of pressure is the pascal (symbol Pa), but vacuum is usually measured in torrs, named for Torricelli, an early Italian physicist (1608–1647). A torr is equal to the displacement of a millimeter of mercury (mmHg) in a manometer with 1 torr equaling 133.3223684 pascals above absolute zero pressure. Vacuum is often also measured using inches of mercury on the barometric scale or as a percentage of atmospheric pressure in bars or atmospheres. Low vacuum is often measured in inches of mercury (inHg), millimeters of mercury (mmHg) or kilopascals (kPa) below atmospheric pressure. "Below atmospheric" means that the absolute pressure is equal to the current atmospheric pressure (e.g. 29.92 inHg) minus the vacuum pressure in the same units. Thus a vacuum of 26 inHg is equivalent to an absolute pressure of 4 inHg (29.92 inHg − 26 inHg).
In other words, most low vacuum gauges that read, for example, −28 inHg at full vacuum are actually reporting 2 inHg, or 50.79 Torr. Many inexpensive low vacuum gauges have a margin of error and may report a vacuum of −30 inHg, or 0 Torr but in practice this generally requires a two stage rotary vane or other medium type of vacuum pump to go much beyond (lower than) 25 torr.
Many devices are used to measure the pressure in a vacuum, depending on what range of vacuum is needed.[48]
Hydrostatic gauges (such as the mercury column manometer) consist of a vertical column of liquid in a tube whose ends are exposed to different pressures. The column will rise or fall until its weight is in equilibrium with the pressure differential between the two ends of the tube. The simplest design is a closed-end U-shaped tube, one side of which is connected to the region of interest. Any fluid can be used, but mercury is preferred for its high density and low vapour pressure. Simple hydrostatic gauges can measure pressures ranging from 1 torr (100 Pa) to above atmospheric. An important variation is the McLeod gauge which isolates a known volume of vacuum and compresses it to multiply the height variation of the liquid column. The McLeod gauge can measure vacuums as high as 10−6 torr (0.1 mPa), which is the lowest direct measurement of pressure that is possible with current technology. Other vacuum gauges can measure lower pressures, but only indirectly by measurement of other pressure-controlled properties. These indirect measurements must be calibrated via a direct measurement, most commonly a McLeod gauge.[49]
Mechanical or elastic gauges depend on a Bourdon tube, diaphragm, or capsule, usually made of metal, which will change shape in response to the pressure of the region in question. A variation on this idea is the capacitance manometer, in which the diaphragm makes up a part of a capacitor. A change in pressure leads to the flexure of the diaphragm, which results in a change in capacitance. These gauges are effective from 10+3 torr to 10−4 torr, and beyond.
Thermal conductivity gauges rely on the fact that the ability of a gas to conduct heat decreases with pressure. In this type of gauge, a wire filament is heated by running current through it. A thermocouple or Resistance Temperature Detector (RTD) can then be used to measure the temperature of the filament. This temperature is dependent on the rate at which the filament loses heat to the surrounding gas, and therefore on the thermal conductivity. A common variant is the Pirani gauge which uses a single platimum filament as both the heated element and RTD. These gauges are accurate from 10 torr to 10−3 torr, but they are sensitive to the chemical composition of the gases being measured.
Ion gauges are used in ultrahigh vacuum. They come in two types: hot cathode and cold cathode. In the hot cathode version an electrically heated filament produces an electron beam. The electrons travel through the gauge and ionize gas molecules around them. The resulting ions are collected at a negative electrode. The current depends on the number of ions, which depends on the pressure in the gauge. Hot cathode gauges are accurate from 10−3 torr to 10−10 torr. The principle behind cold cathode version is the same, except that electrons are produced in a discharge created by a high voltage electrical discharge. Cold cathode gauges are accurate from 10−2 torr to 10−9 torr. Ionization gauge calibration is very sensitive to construction geometry, chemical composition of gases being measured, corrosion and surface deposits. Their calibration can be invalidated by activation at atmospheric pressure or low vacuum. The composition of gases at high vacuums will usually be unpredictable, so a mass spectrometer must be used in conjunction with the ionization gauge for accurate measurement.[50]
See also
- Joining materials
- Decay of the vacuum (Pair production)
- False vacuum
- Helium mass spectrometer - technical instrumentation to detect a vacuum leak
- Engine vacuum
- Pneumatic tube - transport system using vacuum or pressure to move containers in tubes
- Rarefaction - reduction of a medium's density
- Suction - creation of a partial vacuum
- Vacuum angle
- Vacuum cementing - natural process of solidifying homogeneous "dust" in vacuum
- Vacuum deposition - process of depositing atoms and molecules in a sub-atmospheric pressure environment
- Vacuum engineering
- Vacuum flange
Notes
- ^ a b c d Chambers, Austin (2004). Modern Vacuum Physics. Boca Raton: CRC Press. ISBN 0-8493-2438-6. OCLC 55000526.
- ^ Campbell, Jeff (2005). Speed cleaning. p. 97. ISBN 1594862745. http://books.google.com/books?id=hqegeIz9dyQC&pg=PA97. Note that 1 inch of water is ≈0.0025 atm.
- ^ a b Gabrielse, G., et. al. (1990). "Thousandfold Improvement in Measured Antiproton Mass". Phys. Rev. Lett. 65 (11): 1317–1320. Bibcode 1990PhRvL..65.1317G. doi:10.1103/PhysRevLett.65.1317. PMID 10042233.
- ^ a b Tadokoro, M. (1968). "A Study of the Local Group by Use of the Virial Theorem". Publications of the Astronomical Society of Japan 20: 230. Bibcode 1968PASJ...20..230T. This source estimates a density of 7×10−29 g/cm3 for the Local Group. An atomic mass unit is 1.66×10−24 g, for roughly 40 atoms per cubic meter.
- ^ How to Make an Experimental Geissler Tube, Popular Science monthly, February 1919, Unnumbered page, Scanned by Google Books: http://books.google.com/books?id=7igDAAAAMBAJ&pg=PT3
- ^ "What words in the English language contain two u's in a row?", Oxford Dictionaries Online, http://oxforddictionaries.com/page/twousinarow, retrieved 2011-10-23
- ^ Squire, Tom (September 27, 2000), "U.S. Standard Atmosphere, 1976", Thermal Protection Systems Expert and Material Properties Database (NASA), http://tpsx.arc.nasa.gov/cgi-perl/alt.pl, retrieved 2011-10-23
- ^ "Human Exposure to Vacuum". http://www.sff.net/people/Geoffrey.Landis/vacuum.html. Retrieved 2006-03-25.
- ^ Billings, Charles E. (1973). "Barometric Pressure". In edited by James F. Parker and Vita R. West. Bioastronautics Data Book (Second ed.). NASA. NASA SP-3006.
- ^ Webb P. (1968). "The Space Activity Suit: An Elastic Leotard for Extravehicular Activity". Aerospace Medicine 39: 376–383. PMID 4872696.
- ^ Cooke JP, RW Bancroft (1966). "Some Cardiovascular Responses in Anesthetized Dogs During Repeated Decompressions to a Near-Vacuum". Aerospace Medicine 37: 1148–1152. PMID 5972265.
- ^ a b c d e Harding, Richard M. (1989). Survival in Space: Medical Problems of Manned Spaceflight. London: Routledge. ISBN 0-415-00253-2. OCLC 18744945..
- ^ Höhentodversuche im KZ Dachau Seite 15-20
- ^ Czarnik, Tamarack R.. "EBULLISM AT 1 MILLION FEET: Surviving Rapid/Explosive Decompression". http://www.sff.net/people/Geoffrey.Landis/ebullism.html. Retrieved 2006-03-25.
- ^ a b Genz, Henning (1994). Nothingness, the Science of Empty Space (translated from German by Karin Heusch ed.). New York: Perseus Book Publishing (published 1999). ISBN 978-0-7382-0610-3. OCLC 48836264..
- ^ Institute and Museum of the History of Science. Pompeii: Nature, Science, and Technology in a Roman Town[1]
- ^ Zahoor, Akram (2000). Muslim History: 570-1950 C.E.. Gaithersburg, MD: AZP (ZMD Corporation). ISBN 9780970238900.[self-published source?]
- ^ Arabic and Islamic Natural Philosophy and Natural Science, Stanford Encyclopedia of Philosophy
- ^ El-Bizri, Nader (2007). "In Defence of the Sovereignty of Philosophy: Al-Baghdadi's Critique of Ibn al-Haytham's Geometrisation of Place". Arabic Sciences and Philosophy (Cambridge University Press) 17: 57–80. doi:10.1017/S0957423907000367.
- ^ Dallal, Ahmad (2001-2002). "The Interplay of Science and Theology in the Fourteenth-century Kalam". From Medieval to Modern in the Islamic World, Sawyer Seminar at the University of Chicago. http://humanities.uchicago.edu/orgs/institute/sawyer/archive/islam/dallal.html. Retrieved 2008-02-02.
- ^ Donald Routledge Hill, "Mechanical Engineering in the Medieval Near East", Scientific American, May 1991, pp. 64-69 (cf. Donald Routledge Hill, Mechanical Engineering)
- ^ Ahmad Y Hassan. "The Origin of the Suction Pump: Al-Jazari 1206 A.D.". http://www.history-science-technology.com/Notes/Notes%202.htm. Retrieved 2008-07-16.
- ^ Donald Routledge Hill (1996), A History of Engineering in Classical and Medieval Times, Routledge, pp. 143 & 150-2.
- ^ a b Edward Grant (1981). Much ado about nothing: theories of space and vacuum from the Middle Ages to the scientific revolution. Cambridge University Press. ISBN 9780521229838. http://books.google.com/books?id=SidBQyFmgpsC.
- ^ a b Barrow, John D. (2000). The book of nothing : vacuums, voids, and the latest ideas about the origins of the universe (1st American ed.). New York: Pantheon Books. ISBN 0-09-928845-1. OCLC 46600561.
- ^ "The World's Largest Barometer". http://www.denmark.com.au/en/Worlds+Largest+Barometer/default.htm. Retrieved 2008-04-30.
- ^ Encyclopedia Britannica:Otto von Guericke
- ^ R. H. Patterson, Ess. Hist. & Art 10 1862
- ^ William Crookes, The Chemical News and Journal of Industrial Science; with which is Incorporated the "Chemical Gazette." (1932)
- ^ Pickering, W. H. (1912). "Solar system, the motion of the, relatively to the intersteller absorbing medium". Monthly Notices of the Royal Astronomical Society 72: 740. Bibcode 1912MNRAS..72..740P.
- ^ Michelson-Morley: Detecting The Ether Wind Experiment
- ^ Michelson-Morley Interometer Results
- ^ French Wikipedia article on Vacuum, citing appendix 5 of Relativity - the Special and General Theory, translated to French by Robert Lawson, 1961. (Please replace this with a more direct reference.)
- ^ Einstein, A., Naturwissenschaften 6, 697-702 (1918)
- ^ Werner S. Weiglhofer and Akhlesh Lakhtakia (2003). "§ 4.1 The classical vacuum as reference medium". Introduction to complex mediums for optics and electromagnetics. SPIE Press. pp. 28, 34, 65. ISBN 9780819449474. http://books.google.com/?id=QtIP_Lr3gngC&pg=PA34.
- ^ Tom G. MacKay (2008). "Electromagnetic Fields in Linear Bianisotropic Mediums". In Emil Wolf. Progress in Optics, Volume 51. Elsevier. p. 143. ISBN 9780444520388. http://books.google.com/books?id=lCm9Q18P8cMC&pg=PA143.
- ^ Chattopadhyay, D. and Rakshit, P.C. (2004). Elements of Physics: vol. 1. New Age International. p. 577. ISBN 8122415385. http://books.google.com/books?id=tvkoopJMQQ8C&pg=PA577.
- ^ Ishimaru, H (1989). "Ultimate Pressure of the Order of 10-13 torr in an Aluminum Alloy Vacuum Chamber". J. Vac. Sci. Technol. 7 (3-II): 2439–2442. doi:10.1116/1.575916.
- ^ American Vacuum Society. "Glossary". AVS Reference Guide. http://www.aip.org/avsguide/refguide/glossary.html. Retrieved 2006-03-15.
- ^ National Physical Laboratory, UK. "FAQ on Pressure and Vacuum". http://www.npl.co.uk/pressure/faqs/vacuum.html. Retrieved 2006-03-25.
- ^ BS 2951: Glossary of Terms Used in Vacuum Technology. Part I. Terms of General Application. British Standards Institution, London, 1969.
- ^ DIN 28400: Vakuumtechnik Bennenungen und Definitionen, 1972.
- ^ "Vacuum Measurements". Pressure Measurement Division. Setra Systems, Inc. 1998. http://www.setra.com/tra/app/app_vac.htm. Retrieved 2010-04-08.
- ^ "A look at vacuum pumps 14-9". eMedicine. McNally Institute. http://www.mcnallyinstitute.com/14-html/14-09.htm. Retrieved 2010-04-08.
- ^ "1500 Torr Diaphragm Transmitter" (PDF). Vacuum Transmitters for Diaphragm & Pirani Sensors 24 VDC Power. Vacuum Research Corporation. 2003-07-26. http://www.vacuumresearch.com/partsnmans/pdfs/24vdcman.pdf. Retrieved 2010-04-08.
- ^ Öpik, E. J. (May 1962). "The Lunar Atmosphere". Planetary and Space Science (Elsevier) 9 (5): 211–244. Bibcode 1962P&SS....9..211O. doi:10.1016/0032-0633(62)90149-6. ISSN 0032-0633..
- ^ University of New Hampshire Experimental Space Plasma Group. "What is the Interstellar Medium". The Interstellar Medium, an online tutorial. http://www-ssg.sr.unh.edu/ism/what1.html. Retrieved 2006-03-15.
- ^ John H., Moore; Christopher Davis, Michael A. Coplan and Sandra Greer (2002). Building Scientific Apparatus. Boulder, CO: Westview Press. ISBN 0-8133-4007-1. OCLC 50287675.
- ^ Beckwith, Thomas G.; Roy D. Marangoni and John H. Lienhard V (1993). "Measurement of Low Pressures". Mechanical Measurements (Fifth ed.). Reading, MA: Addison-Wesley. pp. 591–595. ISBN 0-201-56947-7.
- ^ Robert M. Besançon, ed (1990). "Vacuum Techniques" (3rd edition ed.). Van Nostrand Reinhold, New York. pp. 1278–1284. ISBN 0-442-00522-9.
External links
- VIDEO on the nature of vacuum by Canadian astrophysicist Doctor P
- The Foundations of Vacuum Coating Technology
- American Vacuum Society
- Journal of Vacuum Science and Technology A
- Journal of Vacuum Science and Technology B
- FAQ on explosive decompression and vacuum exposure.
- Discussion of the effects on humans of exposure to hard vacuum.
- Vacuum Energy in High Energy Physics
- Vacuum, Production of Space
- "Much Ado About Nothing" by Professor John D. Barrow, Gresham College
- Free pdf copy of The Structured Vacuum - thinking about nothing by Johann Rafelski and Berndt Muller (1985) ISBN 3-87144-889-3.
Wikimedia Foundation. 2010.




