- Mean free path
-
In physics, the mean free path is the average distance covered by a moving particle (such as an atom, a molecule, a photon) between successive impacts (collisions) [1] which modify its direction or energy or other particle properties.
Contents
Derivation
 Figure 1: Slab of target
Figure 1: Slab of targetImagine a beam of particles being shot through a target, and consider an infinitesimally thin slab of the target (Figure 1). The atoms (or particles) that might stop a beam particle are shown in red. The magnitude of mean free path depends on the characteristics of the system the particle is in:
Where
 is the mean free path, n is the number of target particles per unit volume, and σ is the effective cross sectional area for collision.
is the mean free path, n is the number of target particles per unit volume, and σ is the effective cross sectional area for collision.The area of the slab is L2 and its volume is L2dx. The typical number of stopping atoms in the slab is the concentration n times the volume, i.e., nL2dx. The probability that a beam particle will be stopped in that slab is the net area of the stopping atoms divided by the total area of the slab.
where σ is the area (or, more formally, the "scattering cross-section") of one atom.
The drop in beam intensity equals the incoming beam intensity multiplied by the probability of being stopped within the slab
- dI = − Inσdx
This is an ordinary differential equation
whose solution is known as Beer-Lambert law and has form
 , where x is the distance traveled by the beam through the target and I0 is the beam intensity before it entered the target; ℓ is called the mean free path because it equals the mean distance traveled by a beam particle before being stopped. To see this, note that the probability that a particle is absorbed between x and x + dx is given by
, where x is the distance traveled by the beam through the target and I0 is the beam intensity before it entered the target; ℓ is called the mean free path because it equals the mean distance traveled by a beam particle before being stopped. To see this, note that the probability that a particle is absorbed between x and x + dx is given byThus the expectation value (or average, or simply mean) of x is
Fraction of particles that were not stopped (attenuated) by the slab is called transmission
 where x is equal to the thickness of the slab x = dx.
where x is equal to the thickness of the slab x = dx.Mean free path in kinetic theory
In kinetic theory the mean free path of a particle, such as a molecule, is the average distance the particle travels between collisions with other moving particles. The formula
 still holds for a particle with a high velocity relative to the velocities of an ensemble of identical particles with random locations. If, on the other hand, the velocities of the identical particles have a Maxwell distribution, the following relationship applies:[2]
still holds for a particle with a high velocity relative to the velocities of an ensemble of identical particles with random locations. If, on the other hand, the velocities of the identical particles have a Maxwell distribution, the following relationship applies:[2]and it may be shown that:[3]
where kB is the Boltzmann constant, T is temperature, p is pressure, and d is the diameter of the gas particles.
Following table lists some typical values for air at different pressures.
Vacuum range Pressure in hPa (mbar) Molecules / cm3 Molecules / m3 Mean free path Ambient pressure 1013 2.7 × 1019 2.7 × 1025 68 nm[4] Low vacuum 300 – 1[dubious ] 1019 – 1016 1025 – 1022 0.1 – 100 μm Medium vacuum 1 – 10−3 1016 – 1013 1022 – 1019 0.1 – 100 mm High vacuum 10−3 – 10−7 1013 – 109 1019 – 1015 10 cm – 1 km Ultra high vacuum 10−7 – 10−12 109 – 104 1015 – 1010 1 km – 105 km Extremely high vacuum <10−12 <104 <1010 >105 km Mean free path in radiography
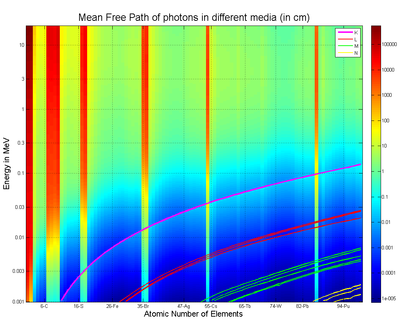 Mean free path for photons in energy range from 1 keV to 20 MeV for Elements Z = 1 to 100. Based on data from.[5] The discontinuities are due to low density of gas elements. Six bands correspond to neighborhoods of six noble gases. Also shown are locations of Compton edges.
Mean free path for photons in energy range from 1 keV to 20 MeV for Elements Z = 1 to 100. Based on data from.[5] The discontinuities are due to low density of gas elements. Six bands correspond to neighborhoods of six noble gases. Also shown are locations of Compton edges.
In gamma-ray radiography the mean free path of a pencil beam of mono-energetic photons, is the average distance a photon travels between collisions with atoms of the target material. It depends on material and energy of the photons:
where μ is linear attenuation coefficient, μ/ρ is mass attenuation coefficient and ρ is density of the material. Mass attenuation coefficient can be looked up or calculated for any material and energy combination using NIST databases [6] [7]
In X-ray radiography the calculation of mean free path is more complicated since photons are not mono-energetic, but have some distribution of energies called spectrum. As photons move through the target material they are attenuated with probabilities depending on their energy, as a result their distribution changes in process called Spectrum Hardening. Because of Spectrum Hardening mean free path of X-ray spectrum changes with distance.
Sometimes people measure thickness of material in number of mean free paths. Material with thickness of one mean free path will attenuate 37% (1/e) of photons. This concept is closely related to Half-Value Layer (HVL) or material with thickness of one HVL will attenuate 50% of photons. Standard x-ray image is a transmission image, a minus log of it is sometimes referred as number of mean free paths image.
Mean free path in particle physics
In particle physics the concept of mean free path is not commonly used, replaced instead by the similar concept of attenuation length. In particular, for high-energy photons, which mostly interact by electron-positron pair production, the radiation length is used much like the mean free path in radiography.
Mean free path in nuclear physics
Independent particle models in nuclear physics require the undisturbed orbiting of nucleons within the nucleus before they interact with other nucleons. Blatt and Weisskopf, in their 1952 textbook "Theoretical Nuclear Physics" (p. 778) wrote "The effective mean free path of a nucleon in nuclear matter must be somewhat larger than the nuclear dimensions in order to allow the use of the independent particle model. This requirement seems to be in contradiction to the assumptions made in the theory... We are facing here one of the fundamental problems of nuclear structure physics which has yet to be solved." (quoted by Norman D. Cook in "Models of the Atomic Nucleus" Ed.2 (2010) Springer, in Chapter 5 "The Mean Free Path of Nucleons in Nuclei").[8]
Mean free path in optics
If one takes a suspension of non light absorbing particles of diameter d with a volume fraction Φ. The mean free path [9] of the photons is:
where Qs is scattering efficiency factor. Qs can be evaluated numerically for spherical particles thanks to the Mie theory calculation
Mean free path in acoustics
In an otherwise empty cavity, the mean free path of a single particle bouncing off the walls is:
where V is volume of the cavity and S is total inside surface area of cavity. This relation is used in the derivation of the Sabine equation in acoustics, using a geometrical approximation of sound propagation.[10]
Examples
A classic application of mean free path is to estimate the size of atoms or molecules. Another important application is in estimating the resistivity of a material from the mean free path of its electrons.
For example, for sound waves in an enclosure, the mean free path is the average distance the wave travels between reflections off the enclosure's walls.
In aerodynamics, the mean free path is in the same order of magnitude as the shockwave thickness at mach numbers greater than one.
See also
References
- ^ Author: Marion Brünglinghaus, ENS, European Nuclear Society. "Mean free path". Euronuclear.org. http://www.euronuclear.org/info/encyclopedia/m/mean-fee-path.htm. Retrieved 2011-11-08.
- ^ S. Chapman and T.G. Cowling, The mathematical theory of non-uniform gases, 3rd. edition, Cambridge University Press, 1990, ISBN 052140844X, p. 88
- ^ "Mean Free Path, Molecular Collisions". Hyperphysics.phy-astr.gsu.edu. http://hyperphysics.phy-astr.gsu.edu/hbase/kinetic/menfre.html. Retrieved 2011-11-08.
- ^ Jennings, S (1988). "The mean free path in air". Journal of Aerosol Science 19 (2): 159. doi:10.1016/0021-8502(88)90219-4.
- ^ "NIST: Note - X-Ray Form Factor and Attenuation Databases". Physics.nist.gov. 1998-03-10. http://physics.nist.gov/PhysRefData/XrayNoteB.html. Retrieved 2011-11-08.
- ^ Hubbell, J. H.; Seltzer, S. M.. "Tables of X-Ray Mass Attenuation Coefficients and Mass Energy-Absorption Coefficients". National Institute of Standards and Technology (NIST). http://physics.nist.gov/PhysRefData/XrayMassCoef/cover.html. Retrieved September 2007.
- ^ Berger, M.J.; J.H. Hubbell, S.M. Seltzer, J. Chang, J.S. Coursey, R. Sukumar, and D.S. Zucker. "XCOM: Photon Cross Sections Database". National Institute of Standards and Technology (NIST). http://physics.nist.gov/PhysRefData/Xcom/Text/XCOM.html. Retrieved September 2007.
- ^ Cook, Norman D.. Models of the Atomic Nucleus. Heidelberg: Springer. p. 324. ISBN 978-3-642-14736-4. http://www.res.kutc.kansai-u.ac.jp/~cook/NVSIndex.html.
- ^ Mengual, O; Meunier, G; Cayré, I; Puech, K; Snabre, P (1999). "TURBISCAN MA 2000: multiple light scattering measurement for concentrated emulsion and suspension instability analysis". Talanta 50 (2): 445–56. doi:10.1016/S0039-9140(99)00129-0. PMID 18967735.
- ^ Davis, D. and Patronis, E. "Sound System Engineering" (1997) Focal Press, ISBN 0240803051 p. 173
External links
- Gas Dynamics Toolbox Calculate mean free path for mixtures of gases using VHS model
Categories:
Wikimedia Foundation. 2010.










