- Distal renal tubular acidosis
-
Distal renal tubular acidosis Classification and external resources OMIM 179800 MeSH D000141 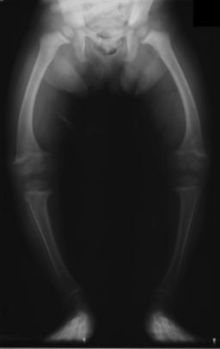 Radiograph of a rickets sufferer, a complication of both distal and proximal RTA.
Radiograph of a rickets sufferer, a complication of both distal and proximal RTA.
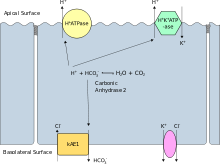
Distal RTA (dRTA) is the classical form of RTA, being the first described. Distal RTA is characterized by a failure of acid secretion by the alpha intercalated cells of the cortical collecting duct of the distal nephron. This failure of acid secretion may be due to a number of causes, and it leads to an inability to acidify the urine to a pH of less than 5.3. Because renal excretion is the primary means of eliminating acid from the body, there is consequently a tendency towards acidemia. This leads to the clinical features of dRTA;.[1]
- Normal anion gap metabolic acidosis/acidemia
- Hypokalemia
- Urinary stone formation (related to alkaline urine, hypercalciuria, and low urinary citrate).[2]
- Nephrocalcinosis (deposition of calcium in the substance of the kidney)
- Bone demineralisation (causing rickets in children and osteomalacia in adults)
The acidosis is variable, and one may have dRTA with alpha intercalated cell failure without necessarily being acidemic, this is termed incomplete dRTA. The diagnosis of dRTA can be made by the observation of a urinary pH of greater than 5.3 in the face of a systemic acidemia (usually taken to be a serum bicarbonate of 20 mmol/l or less). In the case of an incomplete dRTA, failure to acidify the urine following an oral acid loading challenge is often used as a test. The test usually performed is the short ammonium chloride test,[3] in which ammonium chloride capsules are used as the acid load. More recently, an alternative test using furosemide and fludrocortisone has been described.[4]
The symptoms and sequelae of dRTA are variable and ranging from being completely asymptomatic, through loin pain and hematuria from kidney stones to failure to thrive and severe rickets in childhood forms as well as possible renal failure and even death.
dRTA commonly leads to sodium loss and volume contraction, which causes a compensatory increase in blood levels of aldosterone.[5] Aldosterone causes increased resorption of sodium and loss of potassium in the collecting duct of the kidney, so these increased aldosterone levels cause the hypokalemia which is a common symptom of dRTA.[5]
Interestingly, dRTA has been proposed as a possible diagnosis for the unknown malady plaguing Tiny Tim in Charles Dickens' A Christmas Carol.[6][7]
Causes
- Autoimmune disease. Classically Sjögren's syndrome, but it is also associated with systemic lupus erythematosus, rheumatoid arthritis and even hypergammaglobulinemia. Hypokalaemia is often severe in these cases.[8]
- Hereditary causes include mutations of Band 3[9] the basolateral bicarbonate transporter of the intercalated cell, which may transmit in an autosomal dominant fashion in western European cases, or in an autosomal recessive fashion in South East Asian cases. The South East Asian cases are associated with more severe hypokalemia.[10] Other Hereditary causes include mutations of subunits of the apical proton pump vH+-ATPase,[11] which transmit in an autosomal recessive fashion, and may be associated with sensorineural deafness.[12]
- Liver cirrhosis.
- Nephrocalcinosis. While it is a consequence of dRTA, it can also be a cause; related to calcium-induced damage of the cortical collecting duct.
- Renal transplantation.
- Sickle cell anemia.
- Toxins, including ifosfamide,[13] toluene,[14] lithium carbonate[15] and amphotericin B.[16]
- Chronic urinary tract obstruction.
- Possible side effect of IV-administered bisphosphonates, such as zoledronate (~10%)
Treatment
This is relatively straightforward. It involves correction of the acidemia with oral sodium bicarbonate or sodium citrate. This will correct the acidemia and reverse bone demineralisation. Hypokalemia and urinary stone formation and nephrocalcinosis can be treated with potassium citrate tablets which not only replace potassium but also inhibit calcium excretion and thus do not exacerbate stone disease as sodium bicarbonate or citrate may do.[17]
References
- ^ Laing CM, Toye AM, Capasso G, Unwin RJ (2005). "Renal tubular acidosis: developments in our understanding of the molecular basis". Int. J. Biochem. Cell Biol. 37 (6): 1151–61. doi:10.1016/j.biocel.2005.01.002. PMID 15778079.
- ^ Buckalew VM Jr (1989). "Nephrolithiasis in renal tubular acidosis". Journal of Urolology 141 (3 (part 2)): 731–737. PMID 2645431.
- ^ Wrong, O; Davies HEF (1959). "The Excretion of Acid in Renal Disease". QJM 28 (110): 259–313. PMID 13658353.
- ^ Walsh SB, Shirley DG, Wrong OM, Unwin RJ (2007). "Urinary acidification assessed by simultaneous furosemide and fludrocortisone treatment: an alternative to ammonium chloride". Kidney Int. 71 (12): 1310–6. doi:10.1038/sj.ki.5002220. PMID 17410104.
- ^ a b Wein, Alan, J (2011). Campbell-walsh Urology Expert Consult. (10th ed. ed.). Philadelphia, PA: W B Saunders Co. pp. 1045. ISBN 978-1416069119. http://www.mdconsult.com/das/book/pdf/282455359-3/978-1-4160-6911-9/4-u1.0-B978-1-4160-6911-9..00038-4..DOCPDF.pdf?isbn=978-1-4160-6911-9&eid=4-u1.0-B978-1-4160-6911-9..00038-4..DOCPDF.
- ^ Lewis D (1992). "What was wrong with Tiny Tim?". Am J Dis Child 146 (12): 1403–7. PMID 1340779.
- ^ "What Ailed Tiny Tim". Time. 1992-12-28. http://www.time.com/time/magazine/article/0,9171,977391,00.html. Retrieved 2010-05-22.
- ^ Wrong OM, Feest TG, MacIver AG (1993). "Immune-related potassium-losing interstitial nephritis: a comparison with distal renal tubular acidosis". Q. J. Med. 86 (8): 513–34. PMID 8210309. http://qjmed.oxfordjournals.org/cgi/content/abstract/86/8/513.
- ^ Bruce LJ, Cope DL, Jones GK, et al. (1997). "Familial distal renal tubular acidosis is associated with mutations in the red cell anion exchanger (Band 3, AE1) gene". J. Clin. Invest. 100 (7): 1693–707. doi:10.1172/JCI119694. PMC 508352. PMID 9312167. http://www.jci.org/cgi/content/abstract/100/7/1693.
- ^ Bruce LJ, Wrong O, Toye AM, et al. (2000). "Band 3 mutations, renal tubular acidosis and South-East Asian ovalocytosis in Malaysia and Papua New Guinea: loss of up to 95% band 3 transport in red cells". Biochem. J. 350 Pt 1: 41–51. doi:10.1042/0264-6021:3500041. PMC 1221222. PMID 10926824. http://www.biochemj.org/bj/350/0041/bj3500041.htm.
- ^ Wagner, CA; Finberg, KE; Breton, S; Marshansky, V; Brown, D; Geibel, JP (October 2004). "Renal Vacuolar H+-ATPase". Physiological Reviews 84 (4): 1263–314. doi:10.1152/physrev.00045.2003. PMID 15383652. http://physrev.physiology.org/cgi/content/full/84/4/1263.
- ^ Karet FE, Finberg KE, Nelson RD, et al. (1999). "Mutations in the gene encoding B1 subunit of H+-ATPase cause renal tubular acidosis with sensorineural deafness". Nat. Genet. 21 (1): 84–90. doi:10.1038/5022. PMID 9916796. http://www.nature.com/ng/journal/v21/n1/abs/ng0199_84.html.
- ^ Skinner R, Pearson AD, English MW, et al. (1996). "Risk factors for ifosfamide nephrotoxicity in children". Lancet 348 (9027): 578–80. doi:10.1016/S0140-6736(96)03480-0. PMID 8774570. http://linkinghub.elsevier.com/retrieve/pii/S0140673696034800.
- ^ Batlle DC, Sabatini S, Kurtzman NA (1988). "On the mechanism of toluene-induced renal tubular acidosis". Nephron 49 (3): 210–8. doi:10.1159/000185057. PMID 3135502.
- ^ Boton R, Gaviria M, Batlle DC (1987). "Prevalence, pathogenesis, and treatment of renal dysfunction associated with chronic lithium therapy". Am. J. Kidney Dis. 10 (5): 329–45. PMID 3314489.
- ^ McCurdy DK, Frederic M, Elkinton JR (1968). "Renal tubular acidosis due to amphotericin B". N. Engl. J. Med. 278 (3): 124–30. doi:10.1056/NEJM196801182780302. PMID 5634966.
- ^ Morris RC, Sebastian A (2002). "Alkali therapy in renal tubular acidosis: who needs it?" (PDF). J. Am. Soc. Nephrol. 13 (8): 2186–8. doi:10.1097/01.ASN.0000027973.07189.00. PMID 12138154. http://jasn.asnjournals.org/cgi/reprint/13/8/2186.pdf.
Urinary system · Pathology · Urologic disease / Uropathy (N00–N39, 580–599) Abdominal Primarily
nephrotic.3 Mesangial proliferative · .4 Endocapillary proliferative .5/.6 Membranoproliferative/mesangiocapillaryBy conditionType III RPG/Pauci-immuneTubulopathy/
tubulitisAny/allAny/allGeneral syndromesOtherUreterPelvic UrethraUrethritis (Non-gonococcal urethritis) · Urethral syndrome · Urethral stricture/Meatal stenosis · Urethral caruncleAny/all Obstructive uropathy · Urinary tract infection · Retroperitoneal fibrosis · Urolithiasis (Bladder stone, Kidney stone, Renal colic) · Malacoplakia · Urinary incontinence (Stress, Urge, Overflow)Water-electrolyte imbalance and acid-base imbalance (E86–E87, 276) Volume status Electrolyte Acid-base Metabolic: High anion gap (Ketoacidosis/Diabetic ketoacidosis, Lactic) · Normal anion gap (Hyperchloremic, Renal tubular)BothCategories:- Kidney diseases
Wikimedia Foundation. 2010.